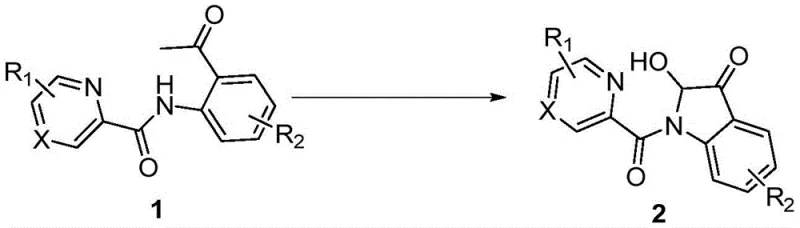
Advanced Copper-Catalyzed Synthesis of 2-Hydroxy-Indol-3-One Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN113278007B, published in May 2022, introduces a transformative synthetic strategy for producing 2-hydroxy-indol-3-one compounds, a privileged structural motif found in numerous bioactive natural products and drug candidates. This invention specifically utilizes N-(2-acetylphenyl)picolinamide derivatives as key starting materials, employing copper salts such as cuprous iodide as catalysts under an oxygen atmosphere. The significance of this technology lies in its ability to forge the indole-3-one core through a direct oxidative cyclization process that operates under remarkably mild conditions, typically between 80°C and 110°C. For R&D directors and process chemists, this represents a critical advancement over legacy methods, offering a pathway to access valuable intermediates like notoamide O and brevianamide analogues with superior atom economy and operational simplicity. The method eliminates the need for hazardous stoichiometric oxidants, aligning perfectly with modern green chemistry principles while maintaining high yields across a broad substrate scope.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-hydroxy-indol-3-one skeleton has been fraught with significant chemical and operational challenges that hinder efficient manufacturing. Early approaches, such as those reported by the Foote group, relied on the use of dimethyldioxirane (DMD) as a potent oxidant to convert 2-methyl-1H-indoles. However, this methodology is severely limited by extremely low yields, often falling below 10%, making it economically unviable for any scale beyond milligram-level discovery. Furthermore, DMD is inherently unstable and hazardous to handle on a large scale, posing serious safety risks in a production environment. Alternative strategies, like the Grignard-based approach described by the Dash group in 2019, require specialized pre-functionalized starting materials such as 3-hydroxy-indol-2-ones. These routes demand harsh reaction conditions and strict anhydrous environments, which drastically increase the complexity of the process and the cost of goods. The necessity for substrate pre-functionalization adds extra synthetic steps, reducing overall throughput and generating more waste, which is a major pain point for supply chain managers focused on sustainability and cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel copper-catalyzed protocol disclosed in CN113278007B offers a streamlined, one-step solution that fundamentally changes the economic landscape of producing these compounds. By leveraging the directing ability of the pyridine moiety in the N-(2-acetylphenyl)picolinamide substrate, the reaction facilitates a highly selective intramolecular C-H functionalization. This approach utilizes molecular oxygen from the air as the terminal oxidant, which is not only inexpensive but also generates water as the only byproduct, thereby maximizing atom economy. The reaction conditions are exceptionally mild, proceeding effectively at temperatures around 100°C in common polar aprotic solvents like DMSO or DMF. This simplicity translates directly into reduced operational costs and safer working conditions. Moreover, the method exhibits remarkable tolerance to various functional groups, allowing for the direct synthesis of diverse derivatives without the need for protecting group strategies. This capability significantly shortens the synthetic route, providing a reliable pharmaceutical intermediates supplier with a distinct competitive advantage in delivering complex molecules rapidly.
Mechanistic Insights into Cu-Catalyzed Oxidative Cyclization
The core of this technological breakthrough lies in the intricate interplay between the copper catalyst and the pyridine directing group, which orchestrates the selective formation of the indole ring. The mechanism initiates with the coordination of the copper species to the nitrogen atom of the pyridine ring and the carbonyl oxygen of the acetyl group, forming a stable chelate complex. This coordination brings the metal center into close proximity with the specific C-H bond on the aromatic ring that needs to be activated. Under the influence of the oxygen atmosphere, the copper catalyst undergoes a redox cycle, facilitating the abstraction of a hydrogen atom and the subsequent formation of a carbon-oxygen or carbon-nitrogen bond, depending on the specific pathway nuances. The pyridine directing group is crucial here, as it ensures regioselectivity, preventing unwanted side reactions on other parts of the molecule. This level of control is essential for maintaining high purity specifications, as it minimizes the formation of regioisomers that are difficult to separate. The oxidative nature of the reaction, driven by O2, regenerates the active copper species, allowing the catalytic cycle to continue with high turnover numbers. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as ligand effects or oxygen pressure, to further optimize yields for specific substrates.
Impurity control is another critical aspect where this mechanism offers distinct advantages over traditional oxidation methods. In conventional processes using strong chemical oxidants, over-oxidation is a common issue, leading to the degradation of the sensitive indole core or the formation of N-oxide byproducts. However, the copper-catalyzed system described here operates under kinetic control where the cyclization is faster than non-selective oxidation pathways. The mild temperature range of 80-110°C further suppresses thermal decomposition pathways that often plague high-temperature reactions. Additionally, the use of simple acid additives like acetic acid helps to stabilize the reaction intermediates and buffer the system, preventing the accumulation of basic byproducts that could catalyze decomposition. For quality control teams, this means a cleaner crude reaction profile, which simplifies downstream purification. The ability to tolerate halogens such as chlorine, bromine, and iodine without dehalogenation is particularly noteworthy, as these groups are often sensitive to metal-catalyzed conditions. This robustness ensures that the final high-purity 2-hydroxy-indol-3-one products retain their functional handles for subsequent derivatization, which is vital for medicinal chemistry campaigns.
How to Synthesize 2-Hydroxy-Indol-3-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize the benefits of the patented technology. The process begins with the precise weighing of the N-(2-acetylphenyl)picolinamide substrate, ensuring a molar purity that supports high conversion rates. The choice of copper salt is flexible, with cuprous iodide being the preferred catalyst, though cupric chloride and other copper sources are also effective, offering procurement teams flexibility in sourcing raw materials. The reaction is typically conducted in a sealed vessel capable of withstanding slight positive pressure from the oxygen atmosphere. Detailed standard operating procedures for mixing, heating, and workup are essential to ensure reproducibility and safety. The following guide outlines the standardized synthesis steps derived from the patent examples, serving as a foundational protocol for scaling this chemistry.
- Combine N-(2-acetylphenyl)picolinamide derivative, copper salt catalyst (e.g., CuI), and acid additive in a polar aprotic solvent like DMSO.
- Purge the reaction vessel with vacuum and backfill with oxygen, then heat the mixture to 80-110°C for 4-6 hours with stirring.
- Upon completion, cool the reaction, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered around cost efficiency and supply reliability. The shift from expensive, hazardous oxidants to molecular oxygen represents a drastic reduction in raw material costs. Oxygen is ubiquitous and inexpensive, eliminating the need for complex logistics associated with shipping and storing unstable chemical oxidants. Furthermore, the catalyst loading is relatively low, and copper salts are commodity chemicals with stable pricing, unlike precious metals such as palladium or rhodium which are subject to volatile market fluctuations. This stability allows for more accurate long-term budgeting and cost forecasting. The simplification of the synthetic route from multi-step sequences to a single pot operation also reduces labor costs and equipment occupancy time. By minimizing the number of unit operations, manufacturers can increase throughput without expanding facility footprint, leading to substantial cost savings in capital expenditure and operational overhead.
- Cost Reduction in Manufacturing: The elimination of stoichiometric hazardous oxidants and the use of cheap copper catalysts significantly lower the direct material costs associated with production. Traditional methods often require expensive reagents that generate large amounts of waste, increasing disposal costs. In contrast, this method produces water as a benign byproduct, reducing waste treatment expenses. The high yields reported, often exceeding 70-80%, mean that less starting material is wasted, improving the overall mass balance of the process. This efficiency translates directly into a lower cost per kilogram of the final API intermediate, providing a competitive edge in price-sensitive markets. Additionally, the avoidance of cryogenic conditions or ultra-high pressures reduces energy consumption, further contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and common reagents mitigates the risk of supply chain disruptions. Specialized reagents required by older methods often have single-source suppliers or long lead times, creating bottlenecks. In this new protocol, components like acetic acid, DMSO, and copper iodide are globally sourced commodities with robust supply networks. This diversity of supply sources ensures continuity of production even during regional shortages. The robustness of the reaction to moisture and air (other than the intentional oxygen feed) also simplifies storage and handling requirements, reducing the risk of batch failures due to reagent degradation. For supply chain planners, this reliability is crucial for maintaining consistent inventory levels and meeting delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for scalability. The use of oxygen gas can be easily managed with standard sparging or headspace pressurization techniques familiar to chemical engineers. The mild reaction temperatures reduce the thermal load on cooling systems and minimize the risk of runaway reactions. From an environmental perspective, the high atom economy and reduced waste generation align with increasingly stringent global regulations on chemical manufacturing. Reducing the E-factor (mass of waste per mass of product) simplifies the permitting process and lowers the environmental compliance burden. This makes the technology future-proof against tightening environmental laws, ensuring long-term viability for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in patent CN113278007B. They are intended to provide clarity for technical teams evaluating the feasibility of adopting this route for their specific projects. Understanding these nuances is key to leveraging the full potential of the copper-catalyzed cyclization strategy.
Q: What are the advantages of this copper-catalyzed method over traditional oxidation routes?
A: Unlike traditional methods using hazardous oxidants like dimethyldioxirane (DMD) which yield less than 10%, or Grignard reagents requiring harsh conditions, this copper-catalyzed protocol utilizes molecular oxygen as a green oxidant. It achieves significantly higher yields (up to 80%) under mild temperatures (80-110°C) and avoids the need for pre-functionalized substrates, enhancing both safety and atom economy.
Q: Does this synthesis method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent substrate applicability. The patent data confirms successful cyclization with various substituents including electron-donating groups like methyl and ethyl, as well as electron-withdrawing halogens such as chlorine, bromine, iodine, and fluorine. This versatility allows for the synthesis of a wide library of derivatives for SAR studies without needing to protect sensitive functional groups.
Q: Is this process suitable for large-scale commercial production?
A: The process is highly amenable to scale-up due to its operational simplicity and use of inexpensive reagents. The reaction proceeds in a one-pot manner using common solvents like DMSO and readily available copper salts. The mild reaction conditions and the use of oxygen gas rather than stoichiometric hazardous oxidants simplify the engineering controls required for commercial manufacturing, reducing lead time for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-Indol-3-One Supplier
The technological potential of this copper-catalyzed synthesis is immense, offering a pathway to high-value heterocyclic building blocks that are essential for modern drug discovery. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant is seamless. Our team of expert process chemists is well-versed in optimizing copper-catalyzed reactions to meet stringent purity specifications required by global regulatory bodies. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our 2-hydroxy-indol-3-one intermediates meet the highest quality standards. Our commitment to technical excellence ensures that we can handle the complexities of this chemistry, from catalyst removal to final crystallization, delivering a product that is ready for the next stage of synthesis.
We invite potential partners to engage with us to explore how this innovative synthesis route can benefit their specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how switching to this method can reduce your overall manufacturing expenses. We encourage you to contact our technical procurement team to discuss your specific requirements. We are prepared to provide specific COA data for our reference standards and conduct comprehensive route feasibility assessments tailored to your project timelines. Let us collaborate to bring your next generation of therapeutic agents to market faster and more efficiently.
