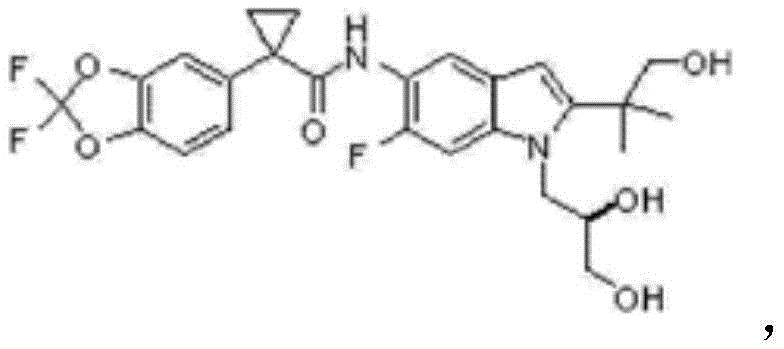
Optimizing Tezacaftor Intermediate II Production: A Novel Copper-Catalyzed Strategy for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways that balance efficiency with regulatory compliance, particularly for complex cystic fibrosis treatments like Tezacaftor. Patent CN110437125B introduces a transformative preparation method for Tezacaftor Intermediate II (CAS 1294504-67-8), addressing critical bottlenecks in the existing supply chain. This innovation shifts the paradigm from traditional palladium-dependent cyclization to a more economical and environmentally benign copper-catalyzed system. For global procurement teams and R&D directors, this patent represents a pivotal opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials without the baggage of heavy metal contamination. The technical breakthrough lies not just in the substitution of metals, but in the holistic optimization of reaction conditions that enhance both yield and operational safety. By leveraging this novel approach, manufacturers can achieve substantial cost reduction in API manufacturing while maintaining the stringent quality standards required for final drug substance production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
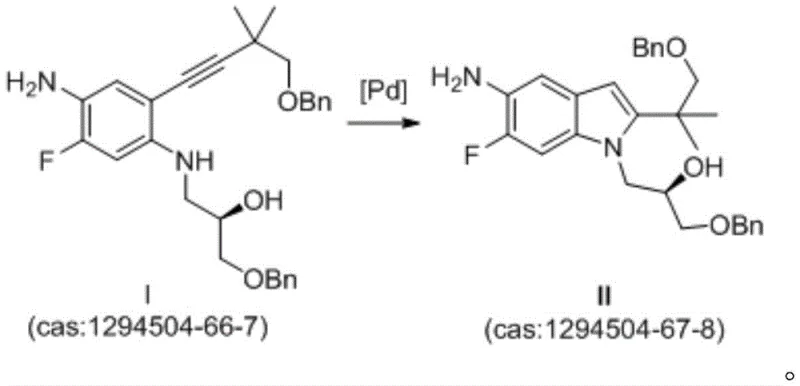
Prior art methodologies, such as those disclosed in patent CN103038214B, rely heavily on palladium catalysts to facilitate the crucial cyclization step required to form the indole core of the intermediate. While chemically feasible, these palladium-mediated routes present significant commercial and technical drawbacks that hinder large-scale adoption. The primary concern is the exorbitant cost associated with palladium reagents, which directly inflates the cost of goods sold (COGS) for the final intermediate. Moreover, the removal of trace palladium residues from the final product is a notoriously difficult and expensive purification challenge, often requiring specialized scavengers or multiple recrystallization steps. This not only extends the reducing lead time for high-purity intermediates but also introduces variability in the final impurity profile. From a supply chain perspective, reliance on precious metals creates vulnerability to market price fluctuations and availability issues, making the production process less predictable and more risky for long-term contracts.
The Novel Approach
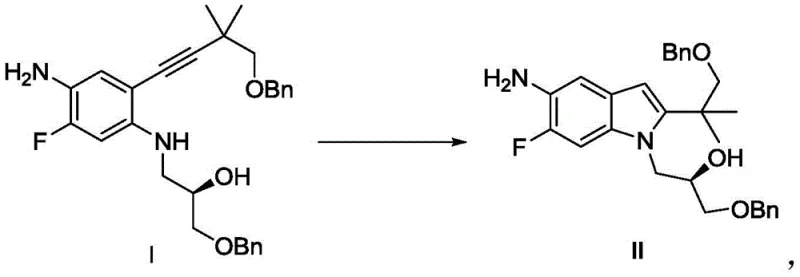
In stark contrast, the method described in CN110437125B utilizes a copper-based catalytic system, specifically employing Cuprous Iodide (CuI) in conjunction with N-methyl pyrrolidone (NMP). This strategic shift eliminates the dependency on palladium entirely, thereby removing the associated cost burden and purification complexities. The new route operates effectively in common organic solvents like chlorobenzene or toluene at moderate temperatures ranging from 100°C to 150°C. This simplicity translates directly into operational efficiency, as the reaction conditions are easier to control and scale. The absence of precious metals means that the downstream processing is significantly streamlined, allowing for faster turnover and higher throughput. For a reliable pharmaceutical intermediates supplier, adopting this copper-catalyzed protocol ensures a more stable and cost-effective production line, ultimately benefiting the entire value chain from raw material sourcing to final API delivery.
Mechanistic Insights into CuI/NMP-Catalyzed Cyclization
The core of this technological advancement lies in the unique interaction between the copper salt and the nitrogen-containing ligand, N-methyl pyrrolidone. In the cyclization of Compound I to Intermediate II, the copper species acts as a Lewis acid activator, facilitating the intramolecular nucleophilic attack of the amine nitrogen onto the alkyne moiety. The presence of NMP is critical, as it likely stabilizes the copper intermediate and enhances the solubility of the inorganic base, creating a homogeneous reaction environment that promotes efficient turnover. Unlike palladium cycles which often require phosphine ligands that are air-sensitive and costly, this copper system is robust and tolerant to various reaction conditions. The mechanism avoids the formation of complex organometallic side products that are typical in Pd-catalyzed couplings, resulting in a cleaner reaction profile. This mechanistic clarity allows process chemists to fine-tune parameters such as base strength and temperature with greater precision, ensuring consistent batch-to-batch reproducibility which is essential for commercial scale-up of complex pharmaceutical intermediates.
Furthermore, the impurity control mechanism inherent in this copper-catalyzed route is superior to its predecessors. Traditional methods often suffer from the formation of homocoupling byproducts or incomplete cyclization due to catalyst deactivation. The CuI/NMP system maintains catalytic activity over extended periods, ensuring near-complete consumption of the starting material as evidenced by HPLC analysis in the patent examples. The resulting crude product contains significantly fewer metal-associated impurities, simplifying the crystallization process. By utilizing a base like sodium hydroxide or cesium carbonate in conjunction with the copper catalyst, the reaction efficiently drives the equilibrium towards the desired indole product. This high level of selectivity minimizes the generation of hard-to-remove structural analogs, thereby enhancing the overall purity of the high-purity Tezacaftor intermediate before it even reaches the final isolation stage.
How to Synthesize Tezacaftor Intermediate II Efficiently
The synthesis of this critical building block is streamlined into a logical sequence that prioritizes yield and ease of operation. The process begins with the preparation of the alkyne precursor, Compound I, via a Sonogashira-type coupling, followed by the key copper-mediated cyclization. The patent details specific molar ratios and solvent choices that maximize efficiency, such as using a 1:0.1 to 1:3 ratio of substrate to copper salt. Detailed standardized synthetic steps see the guide below.
- Prepare Compound I by reacting Compound A and Compound B using a palladium/copper co-catalyst system in acetonitrile under reflux conditions.
- Subject the crude Compound I to cyclization using Cuprous Iodide (CuI) and N-methyl pyrrolidone (NMP) in chlorobenzene at elevated temperatures (100-150°C).
- Perform post-reaction workup involving filtration, aqueous washing, and crystallization from ethyl acetate/n-heptane to isolate the final pure intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed methodology offers tangible strategic benefits beyond mere technical curiosity. The elimination of palladium represents a direct reduction in raw material expenditure, which is a significant factor in the overall cost structure of specialty intermediates. Additionally, the simplified purification workflow reduces the consumption of solvents and auxiliary materials, further driving down operational expenses. This efficiency gain allows suppliers to offer more competitive pricing without compromising on quality margins. Moreover, the robustness of the copper system enhances supply continuity, as it is less susceptible to the disruptions often seen with precious metal supply chains. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturers who depend on timely deliveries.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with readily available copper salts results in a drastic decrease in catalyst costs. Since copper is abundant and inexpensive compared to precious metals, the direct material cost savings are substantial. Furthermore, the removal of the need for specialized palladium scavengers or extensive metal removal protocols reduces the cost of downstream processing. This cumulative effect leads to a leaner manufacturing process where resources are allocated more efficiently, ultimately translating into significant cost savings for the buyer. The economic advantage is compounded by the higher yields achieved, meaning less raw material is wasted per unit of product produced.
- Enhanced Supply Chain Reliability: Relying on copper instead of palladium mitigates the risk of supply chain disruptions caused by geopolitical instability or mining constraints associated with precious metals. Copper is a globally traded commodity with a stable supply network, ensuring that production can continue uninterrupted even during market volatility. This stability allows for more accurate forecasting and inventory planning, reducing the need for excessive safety stock. For the supply chain head, this means a more predictable lead time and a lower risk of production stoppages due to material shortages. The ability to source catalyst materials locally or from diverse suppliers adds an extra layer of resilience to the procurement strategy.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents like chlorobenzene and toluene that are easily handled in large-scale reactors. The absence of toxic heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. This eco-friendly profile reduces the regulatory burden and potential liability associated with hazardous waste management. Easier scale-up means that production volumes can be increased rapidly to meet surging demand without the need for extensive process re-validation. The combination of operational simplicity and environmental safety makes this route highly attractive for long-term commercial manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: Why is the copper-catalyzed method superior to the traditional palladium route for this intermediate?
A: The copper-catalyzed method eliminates the need for expensive palladium reagents, significantly reducing raw material costs. Furthermore, it avoids the risk of toxic palladium residue in the final API, simplifying the purification process and ensuring stricter compliance with heavy metal limits.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization requires a specific catalytic system comprising Cuprous Iodide (CuI) and N-methyl pyrrolidone (NMP). The reaction is typically conducted in chlorobenzene or toluene at temperatures between 100°C and 150°C for 10 to 60 hours to ensure complete conversion.
Q: How does this process impact the overall yield and purity?
A: By optimizing the catalyst system, this method achieves a significant promotion in reaction yield compared to prior art. The process consistently delivers high-purity product (over 98% purity) after simple crystallization, reducing the need for complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tezacaftor Intermediate II Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for life-saving medications like Tezacaftor. Our technical team has thoroughly analyzed the innovations presented in CN110437125B and integrated similar advanced copper-catalyzed strategies into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the highest international standards. We are committed to providing a stable supply of high-quality intermediates that support your drug development and commercialization timelines.
We invite you to collaborate with us to optimize your supply chain for Tezacaftor intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a wealth of chemical expertise and a production capacity designed to support your growth. Let us help you secure a competitive edge in the market through superior chemistry and reliable service.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
