Scalable Synthesis of Whitening Ascorbic Acid Derivatives for Global Cosmetic Markets
Scalable Synthesis of Whitening Ascorbic Acid Derivatives for Global Cosmetic Markets
The global demand for stable and effective skin-whitening agents has driven significant innovation in the synthesis of ascorbic acid derivatives, specifically targeting compounds that offer superior bioavailability and stability compared to native Vitamin C. Patent CN109021014B introduces a groundbreaking methodology for synthesizing 2-O-(3-aminopropyl hydrogen phosphoryl)-ascorbic acid (CAS 220644-17-7), a potent molecule known for its ability to significantly reduce melanin content in pigment cells. This technical report analyzes the novel three-step continuous process detailed in the patent, which leverages inexpensive commodity chemicals such as acetonylidene protected ascorbic acid, N-Boc-3-aminopropanol, and phosphorus oxychloride. By integrating esterification and hydrolysis reactions into a streamlined workflow, this approach addresses critical bottlenecks in the manufacturing of high-value cosmetic intermediates. The method promises not only enhanced reaction yields but also a drastic reduction in operational complexity, making it an ideal candidate for reliable cosmetic ingredient supplier networks seeking to optimize their supply chains for next-generation skincare actives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for phosphorylated ascorbic acid derivatives often suffer from prohibitive costs and complex multi-step procedures that hinder efficient commercial scale-up of complex cosmetic actives. Conventional methodologies frequently rely on expensive phosphorylating agents or require harsh reaction conditions that degrade the sensitive ascorbic acid core, leading to poor selectivity and difficult purification challenges. Furthermore, legacy processes often involve the use of transition metal catalysts that necessitate rigorous and costly removal steps to meet the stringent purity specifications demanded by the pharmaceutical and personal care industries. The accumulation of toxic metal residues and the generation of substantial chemical waste pose significant environmental compliance hurdles, increasing the overall cost of goods sold. Additionally, the instability of intermediate species in older protocols often results in low overall yields, forcing manufacturers to process larger volumes of raw materials to achieve target output, thereby inflating logistics and storage expenses.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a highly efficient strategy that transforms cheap and easily obtained raw materials into the target high-purity ascorbic acid derivative through a controlled sequence of esterification and hydrolysis. The process initiates with the activation of N-Boc-3-aminopropanol using phosphorus oxychloride, followed by the coupling with acetonylidene protected ascorbic acid, ensuring that the reactive sites are selectively targeted without compromising the molecular integrity of the vitamin scaffold. 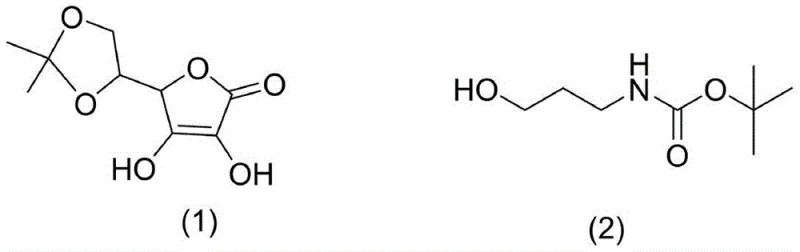 This strategic selection of starting materials allows for a continuous reaction flow that minimizes intermediate handling and exposure to degradative factors. The mild reaction conditions, ranging from zero degrees centigrade to fifty degrees centigrade, preserve the chiral center and prevent the formation of unwanted oxidation by-products. By eliminating the need for exotic catalysts and simplifying the workup procedure to a straightforward solvent evaporation and recrystallization, this method achieves cost reduction in skincare active manufacturing that is unparalleled by previous techniques.
This strategic selection of starting materials allows for a continuous reaction flow that minimizes intermediate handling and exposure to degradative factors. The mild reaction conditions, ranging from zero degrees centigrade to fifty degrees centigrade, preserve the chiral center and prevent the formation of unwanted oxidation by-products. By eliminating the need for exotic catalysts and simplifying the workup procedure to a straightforward solvent evaporation and recrystallization, this method achieves cost reduction in skincare active manufacturing that is unparalleled by previous techniques.
Mechanistic Insights into Phosphorylation Esterification
The core of this synthesis lies in the precise control of the phosphorylation mechanism, which proceeds through a well-defined dichlorophosphate intermediate to ensure high regioselectivity at the 2-O position of the ascorbic acid ring. In the first stage, the hydroxyl group of N-Boc-3-aminopropanol acts as a nucleophile attacking the phosphorus atom of phosphorus oxychloride at low temperatures, effectively displacing chloride ions to form a reactive dichlorophosphate species. 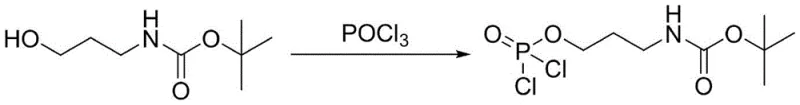 This step is critical as it activates the amino-propyl chain for subsequent coupling while the Boc group protects the amine functionality from unwanted side reactions with the acidic phosphorylating agent. The reaction is carefully monitored to ensure complete conversion before proceeding, as any unreacted alcohol could lead to impurities in the final product. The use of dichloromethane as a solvent provides an ideal medium for solubilizing both the organic amine and the inorganic phosphoryl chloride, facilitating homogeneous reaction kinetics.
This step is critical as it activates the amino-propyl chain for subsequent coupling while the Boc group protects the amine functionality from unwanted side reactions with the acidic phosphorylating agent. The reaction is carefully monitored to ensure complete conversion before proceeding, as any unreacted alcohol could lead to impurities in the final product. The use of dichloromethane as a solvent provides an ideal medium for solubilizing both the organic amine and the inorganic phosphoryl chloride, facilitating homogeneous reaction kinetics.
Following the formation of the activated phosphate, the second stage involves the nucleophilic attack by the enolic hydroxyl group of the protected ascorbic acid on the phosphorus center of the dichlorophosphate intermediate. 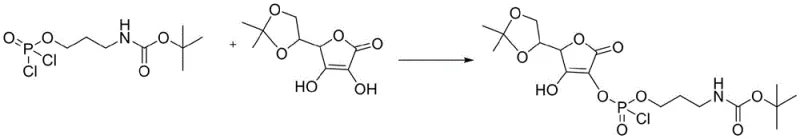 This esterification step replaces the remaining chlorine atoms on the phosphorus with the ascorbyl oxygen, creating the crucial phosphoester bond that links the vitamin C moiety to the aminopropyl tail. The reaction conditions are maintained at room temperature to allow sufficient energy for the coupling without inducing thermal decomposition of the sensitive lactone ring. Finally, the third stage employs a hydrolysis mechanism where water acts as both a solvent and a reagent to cleave the acetonylidene protecting group and the remaining P-Cl bond.
This esterification step replaces the remaining chlorine atoms on the phosphorus with the ascorbyl oxygen, creating the crucial phosphoester bond that links the vitamin C moiety to the aminopropyl tail. The reaction conditions are maintained at room temperature to allow sufficient energy for the coupling without inducing thermal decomposition of the sensitive lactone ring. Finally, the third stage employs a hydrolysis mechanism where water acts as both a solvent and a reagent to cleave the acetonylidene protecting group and the remaining P-Cl bond. 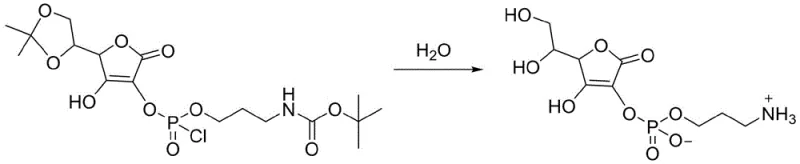 This simultaneous deprotection and hydrolysis at 50°C reveals the free hydroxyl groups on the sugar ring and converts the phosphate monoester into its final hydrogen phosphoryl form, yielding the biologically active target molecule with high stereochemical fidelity.
This simultaneous deprotection and hydrolysis at 50°C reveals the free hydroxyl groups on the sugar ring and converts the phosphate monoester into its final hydrogen phosphoryl form, yielding the biologically active target molecule with high stereochemical fidelity.
How to Synthesize 2-O-(3-aminopropyl hydrogen phosphoryl)-ascorbic acid Efficiently
To implement this synthesis effectively, operators must adhere to strict temperature controls and addition rates to manage the exothermic nature of the phosphorylation reactions. The process begins with the dropwise addition of the amine solution into the phosphorus oxychloride mixture at zero centigrade, followed by a slow warm-up to room temperature over five hours to ensure complete formation of the dichlorophosphate. Subsequently, the protected ascorbic acid solution is introduced under similar cooling conditions to control the rate of esterification, preventing localized overheating that could degrade the product. The final hydrolysis step requires the removal of organic solvents prior to the addition of water, ensuring a clean phase transition for the deprotection reaction. For a comprehensive guide on the exact molar ratios, solvent volumes, and recrystallization parameters, please refer to the standardized protocol below.
- Dropwise add N-Boc-3-aminopropanol solution into phosphorus oxychloride at 0°C, then warm to room temperature to form the dichlorophosphate intermediate.
- Cool the system to 0°C and slowly add acetonylidene protected ascorbic acid solution, reacting to room temperature to obtain the monochlorophosphate intermediate.
- Remove organic solvent, add water, and react at 50°C for 5 hours to hydrolyze the protecting groups and yield the final pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthesis route offers transformative benefits by leveraging widely available commodity chemicals that are not subject to the supply volatility often seen with specialized fine chemical reagents. The reliance on acetonylidene protected ascorbic acid and N-Boc-3-aminopropanol ensures a stable supply chain, as these materials are produced in large volumes for various industries, mitigating the risk of raw material shortages. Furthermore, the elimination of expensive transition metal catalysts removes the need for costly scavenging resins and extensive filtration steps, directly lowering the bill of materials and processing time. The simplified workup procedure, which involves basic solvent evaporation and recrystallization, reduces the demand for specialized waste treatment facilities and lowers the environmental compliance burden associated with heavy metal disposal. This streamlined approach translates into substantial cost savings and enhanced supply chain reliability for manufacturers aiming to secure a competitive edge in the global market for functional skincare ingredients.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of low-cost starting materials and the high efficiency of the reaction sequence, which minimizes raw material waste. By avoiding the use of precious metal catalysts and complex purification columns, the operational expenditure is significantly reduced, allowing for more competitive pricing of the final active ingredient. The high yield reported in the patent data indicates that less feedstock is required per unit of output, further driving down the variable costs associated with production. Additionally, the ability to perform the reaction in common solvents like dichloromethane and water reduces solvent procurement costs and simplifies solvent recovery systems.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent production schedules, as the reaction conditions are mild and easy to control, reducing the likelihood of batch failures due to thermal runaway or sensitivity issues. The use of stable intermediates and the continuous nature of the process minimize the need for long-term storage of hazardous materials, thereby improving facility safety and throughput. Sourcing of raw materials is simplified due to their commodity status, reducing lead times and dependency on single-source suppliers for exotic reagents. This reliability is crucial for maintaining uninterrupted production lines in the fast-paced cosmetic and pharmaceutical sectors.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the straightforward transition from laboratory to potential industrial scales without requiring high-pressure or cryogenic equipment. The minimal generation of hazardous waste, particularly the absence of heavy metal contaminants, aligns with increasingly strict global environmental regulations and sustainability goals. Water is utilized as a green solvent in the final step, reducing the overall organic solvent load and facilitating easier wastewater treatment. This environmental friendliness enhances the corporate social responsibility profile of the manufacturer and ensures long-term regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this specific ascorbic acid derivative, based on the detailed disclosures within the patent literature. Understanding these aspects is vital for R&D teams evaluating the feasibility of integrating this molecule into new formulations or for procurement officers assessing supplier capabilities. The answers provided reflect the specific advantages of the phosphorylation method described, focusing on purity, yield, and process safety.
Q: What are the key advantages of this phosphorylation method over traditional synthesis?
A: This method utilizes inexpensive commodity raw materials like acetonylidene protected ascorbic acid and avoids complex transition metal catalysts. The process operates under mild conditions (0°C to 50°C) and achieves a significant yield improvement compared to conventional routes, while drastically reducing production costs.
Q: How does this process ensure high purity for cosmetic applications?
A: The synthesis employs a continuous one-pot strategy that minimizes intermediate isolation losses and exposure to contaminants. The final recrystallization step using methanol and water effectively removes residual solvents and by-products, ensuring the high-purity specifications required for sensitive skincare formulations.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability with simple operational controls and minimal environmental pollution. The use of common solvents like dichloromethane and water, along with readily available reagents, facilitates easy scale-up from laboratory to multi-ton commercial production without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-O-(3-aminopropyl hydrogen phosphoryl)-ascorbic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this novel synthesis route to revolutionize the availability of high-performance whitening agents in the global cosmetic market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of this critical intermediate. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-O-(3-aminopropyl hydrogen phosphoryl)-ascorbic acid meets the highest standards for safety and efficacy. We are committed to leveraging our technical expertise to optimize this process further, ensuring maximum yield and minimal environmental impact for our partners.
We invite forward-thinking companies to collaborate with us to unlock the full commercial potential of this advanced skincare active. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this cost-effective and high-quality ingredient into your product portfolio.
