Advanced Synthesis of 1,4-Dialkoxy Anthracene: A Scalable Route for Optical Materials
Advanced Synthesis of 1,4-Dialkoxy Anthracene: A Scalable Route for Optical Materials
The development of efficient synthetic routes for functionalized anthracene derivatives is critical for the advancement of optical materials and security technologies. Patent CN102850193A discloses a novel preparation method for 1,4-dialkoxy anthracene compounds, which serve as potent fluorescent agents suitable for anti-counterfeiting applications. This technology represents a significant departure from traditional methodologies by replacing hazardous Lewis acids with benign boric acid catalysts, thereby enhancing process safety and environmental compliance. The core innovation lies in a three-step sequence that transforms readily available commodity chemicals into high-value fluorescent intermediates with controlled regioselectivity. By leveraging this proprietary knowledge, manufacturers can achieve superior purity profiles essential for electronic and optical grade materials. The following analysis details the technical merits and commercial viability of this synthetic strategy for global supply chains.
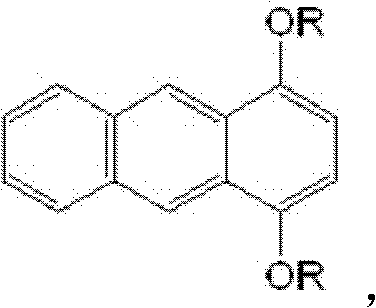
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of anthraquinone derivatives often relies on the Friedel-Crafts acylation using anhydrous aluminum chloride (AlCl3) as a catalyst. This conventional approach presents severe operational challenges, primarily due to the stringent requirement for absolutely anhydrous reaction conditions to prevent catalyst deactivation. Furthermore, the reaction generates substantial amounts of corrosive hydrogen chloride gas and creates complex aluminum-containing sludge that is difficult and costly to treat. The exothermic nature of the AlCl3-catalyzed reaction also poses significant safety risks during scale-up, requiring specialized equipment to manage heat dissipation effectively. Additionally, the workup procedure typically involves quenching with ice water, which generates large volumes of acidic wastewater, complicating environmental compliance and increasing disposal costs. These factors collectively contribute to higher production costs and reduced process reliability for manufacturers relying on legacy technologies.
The Novel Approach
In contrast, the method described in CN102850193A utilizes boric acid (H3BO3) as a catalyst for the initial cyclization step, fundamentally altering the reaction landscape. This substitution allows the reaction to proceed under less stringent moisture controls, significantly simplifying reactor preparation and operation. The use of boric acid minimizes the generation of toxic fumes and corrosive byproducts, creating a safer working environment for plant personnel. Moreover, the subsequent reduction step employs a zinc/acetic acid (Zn/HAc) system, which is known for its mildness and selectivity compared to harsher reducing agents. This novel approach not only improves the overall yield stability but also streamlines the purification process, as the byproducts are easier to separate from the desired anthracene backbone. The result is a more robust and economically viable manufacturing process that aligns with modern green chemistry principles.
Mechanistic Insights into Boric Acid-Catalyzed Cyclization and Reduction
The mechanistic pathway begins with the condensation of phthalic anhydride and hydroquinone in the presence of boric acid and sulfuric acid at elevated temperatures ranging from 165°C to 180°C. In this step, boric acid acts as a mild Lewis acid promoter, facilitating the electrophilic aromatic substitution and subsequent intramolecular cyclization to form 1,4-dihydroxy-9,10-anthraquinone. The presence of sulfuric acid likely serves as a dehydrating agent to drive the equilibrium towards the formation of the quinone ring system. This specific catalytic combination ensures high regioselectivity, favoring the 1,4-substitution pattern which is crucial for the fluorescence properties of the final product. The reaction kinetics are well-controlled within the specified temperature window, preventing over-oxidation or polymerization side reactions that often plague high-temperature organic syntheses.
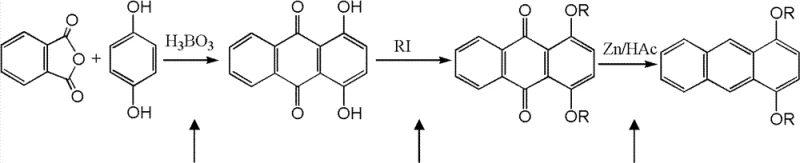
Following the formation of the anthraquinone core, the process involves an alkylation step where the hydroxyl groups are converted to alkoxy groups using alkyl halides or dialkyl sulfates in a basic medium. The final transformation is a reductive aromatization using zinc dust in glacial acetic acid. In this reduction mechanism, zinc serves as the electron donor, reducing the carbonyl groups of the anthraquinone to methylene groups while restoring the aromaticity of the central ring. The acetic acid provides the necessary protons for the reduction and solubilizes the intermediate species. This Zn/HAc system is particularly advantageous because it avoids the use of high-pressure hydrogenation equipment, allowing the reaction to be performed under atmospheric pressure at reflux temperatures of 120°C to 150°C. The selectivity of this reduction is high, preserving the alkoxy substituents while efficiently converting the quinone moiety to the anthracene structure.
How to Synthesize 1,4-Dialkoxy Anthracene Efficiently
The synthesis of these high-value fluorescent agents requires precise control over reaction parameters to maximize yield and purity. The patented process outlines a clear protocol starting with the cyclization of raw materials, followed by etherification and final reduction. Operators must maintain strict temperature gradients, particularly during the initial high-temperature cyclization and the subsequent mild alkylation phases. The detailed标准化 synthesis steps provided below offer a roadmap for replicating the high yields reported in the patent documentation, ensuring consistent quality for commercial batches. Adherence to these parameters is essential for achieving the structural integrity required for optical applications.
- Cyclization of phthalic anhydride and hydroquinone using boric acid catalyst at 165-180°C to form 1,4-hydroxy-anthraquinone.
- Alkylation of the hydroxy-anthraquinone intermediate using alkyl halides or sulfates in the presence of base to form dialkoxy-anthraquinone.
- Reduction of the dialkoxy-anthraquinone using zinc dust and acetic acid under reflux conditions to yield the final 1,4-dialkoxy anthracene.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers distinct advantages by utilizing widely available commodity feedstocks such as phthalic anhydride and hydroquinone. The reliance on common chemicals mitigates supply chain risks associated with specialized or scarce reagents, ensuring continuous production capability even during market fluctuations. Furthermore, the elimination of anhydrous aluminum chloride removes the need for complex storage and handling infrastructure dedicated to moisture-sensitive hazardous materials. This simplification of raw material logistics translates directly into lower operational overhead and reduced capital expenditure for facility upgrades. The process inherently supports a more resilient supply chain model by decoupling production from volatile specialty chemical markets.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous catalysts with boric acid significantly lowers the direct material costs associated with the synthesis. By avoiding the generation of heavy metal sludge, the facility saves substantially on waste treatment and disposal fees, which are often a hidden cost driver in fine chemical manufacturing. The simplified workup procedures reduce labor hours and solvent consumption during purification, further driving down the cost of goods sold. Additionally, the ability to run the alkylation step at near-ambient temperatures (20°C-30°C) reduces energy consumption for heating and cooling compared to cryogenic or high-heat alternatives. These cumulative efficiencies create a leaner cost structure that enhances competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the boric acid catalytic system ensures high batch-to-batch consistency, which is critical for maintaining long-term contracts with downstream users in the security printing and optical industries. Since the raw materials are produced on a massive industrial scale globally, the risk of supply interruption is minimized, providing procurement managers with greater confidence in forecasting. The mild reaction conditions also extend the lifespan of reactor vessels and ancillary equipment by reducing corrosion rates, thereby decreasing maintenance downtime. This reliability allows supply chain heads to optimize inventory levels and reduce safety stock requirements without compromising delivery performance.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of extremely exothermic steps that are difficult to manage in large reactors. The use of zinc and acetic acid in the final step generates waste streams that are easier to neutralize and treat compared to those containing aluminum or heavy transition metals. This environmental compatibility facilitates faster regulatory approvals for new production lines in regions with strict emission standards. The ability to scale from pilot plants to multi-ton production without significant process re-engineering supports rapid market entry for new fluorescent product lines. Consequently, manufacturers can respond agilely to increasing demand for anti-counterfeiting solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 1,4-dialkoxy anthracene derivatives. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this chemistry into their existing portfolios. The responses highlight the balance between performance metrics and operational practicality.
Q: What are the advantages of using boric acid over aluminum chloride in anthraquinone synthesis?
A: Using boric acid eliminates the need for strictly anhydrous conditions required by aluminum chloride, significantly reducing reaction hazards and simplifying post-reaction workup by avoiding heavy metal sludge.
Q: What is the typical yield profile for this three-step synthesis?
A: The patent data indicates individual step yields of approximately 77.5% for cyclization, 68-69% for alkylation, and 64-65% for reduction, offering a robust pathway for commercial production.
Q: Can this process be scaled for industrial anti-counterfeiting applications?
A: Yes, the use of commodity raw materials like phthalic anhydride and hydroquinone, combined with mild alkylation conditions (20-30°C), makes the process highly scalable for producing fluorescent security markers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dialkoxy Anthracene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced catalytic technologies like the boric acid-mediated route to deliver superior optical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,4-dialkoxy anthracene meets the exacting standards required for high-performance fluorescent applications. Our commitment to process optimization allows us to offer competitive pricing without compromising on the quality or safety of the final product.
We invite you to collaborate with us to explore how this innovative synthesis can enhance your product lineup. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing strategies. Let us be your partner in securing a stable and cost-effective supply of critical fluorescent materials.
