Advanced Manufacturing of (S)-1-Phenyl-1,2,3,4-Tetrahydroisoquinoline for Global Pharma Supply Chains
The pharmaceutical industry's relentless pursuit of efficient, safe, and cost-effective synthetic routes for active pharmaceutical ingredients (APIs) and their key intermediates is vividly exemplified in the recent technological advancements regarding the synthesis of (S)-1-phenyl-1,2,3,4-tetrahydroisoquinoline. This specific chiral amine serves as the critical backbone for Solifenacin, a widely prescribed antispasmodic agent used in the treatment of overactive bladder, which has demonstrated substantial market value since its regulatory approval. A groundbreaking preparation method disclosed in patent CN111925266A introduces a paradigm shift away from hazardous phosphorus-based reagents towards a more sustainable and economically viable pathway. This report provides a deep technical analysis of this novel methodology, highlighting its strategic importance for R&D directors seeking robust impurity profiles, procurement managers aiming for raw material cost optimization, and supply chain leaders focused on operational continuity and environmental compliance.
By leveraging benzophenone and aminoacetaldehyde dimethyl acetal as primary feedstocks, the patented process circumvents the significant safety liabilities and waste disposal burdens associated with legacy technologies. The transition from dangerous chlorinating agents to standard mineral acids for the cyclization step represents a major engineering improvement, facilitating smoother technology transfer from laboratory to pilot and commercial scales. Furthermore, the implementation of L-mandelic acid for chiral resolution addresses historical bottlenecks related to low crystallization yields and prolonged processing times. For global stakeholders evaluating potential partners for reliable pharmaceutical intermediates supplier capabilities, understanding the mechanistic nuances and commercial implications of this specific patent is essential for securing a competitive edge in the urological therapeutic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1-phenyl-1,2,3,4-tetrahydroisoquinoline has relied heavily on the Bischler-Napieralski reaction or variations thereof, which typically involve the acylation of 2-phenethylamine followed by cyclodehydration. As illustrated in the reaction scheme below, traditional routes frequently employ phosphorus oxychloride (POCl3) in conjunction with phosphorus pentoxide (P2O5) or polyphosphoric acid to effect the ring closure. These reagents are notoriously hazardous, posing severe risks of thermal runaway and corrosive exposure to personnel, while simultaneously generating vast quantities of phosphorus-laden wastewater that requires complex and costly treatment protocols before discharge.
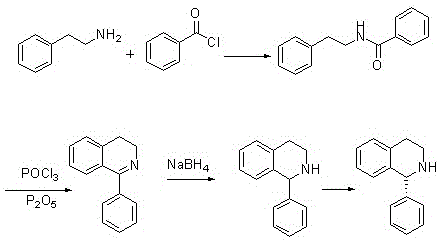
Alternative approaches found in literature, such as those utilizing trifluoromethanesulfonic anhydride (Tf2O) for activation, introduce different sets of operational challenges, including the necessity for cryogenic reaction conditions (e.g., -78°C) which are energy-intensive and difficult to maintain in large-scale reactors. Additionally, methods relying on Grignard reagents or palladium-catalyzed cross-couplings of halogenated isoquinolines demand strictly anhydrous and oxygen-free environments, significantly increasing the complexity of equipment requirements and the risk of batch failure due to moisture ingress. The subsequent chiral resolution steps using tartaric acid often suffer from poor crystallization behavior, leading to suboptimal yields and extended mother liquor recycling loops that hinder overall process throughput and economic efficiency.
The Novel Approach
In stark contrast to these legacy constraints, the innovative route described in patent CN111925266A utilizes a condensation-cyclization strategy that begins with the formation of a Schiff base from benzophenone and aminoacetaldehyde dimethyl acetal. This initial step proceeds smoothly under reflux conditions in solvents like toluene or chlorobenzene, utilizing a Dean-Stark trap to remove water and drive the equilibrium forward without the need for exotic catalysts. The resulting N-(diphenylmethylene)-2,2-dimethoxyethylamine intermediate is then subjected to acid-catalyzed cyclization using concentrated sulfuric acid or phosphoric acid at elevated temperatures, effectively constructing the isoquinoline core while avoiding the generation of toxic phosphorus chloride byproducts.
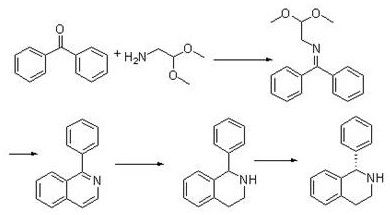
Following the formation of 1-phenylisoquinoline, the process employs catalytic hydrogenation using either Pd/C or Raney Nickel under moderate pressure to saturate the heterocyclic ring, yielding the racemic tetrahydroisoquinoline. The final and perhaps most commercially significant innovation lies in the chiral resolution step, where L-mandelic acid is utilized instead of the conventional tartaric acid. This specific choice of resolving agent promotes rapid and high-yielding crystallization of the desired (S)-enantiomer salt, which can then be easily liberated to provide the free base with high optical purity. This streamlined sequence not only enhances cost reduction in API manufacturing by utilizing cheaper starting materials but also drastically simplifies the downstream purification train, making it an ideal candidate for commercial scale-up of complex chiral amines.
Mechanistic Insights into Acid-Catalyzed Cyclization and Chiral Resolution
The core chemical transformation in this novel pathway is the acid-mediated cyclization of the Schiff base intermediate, which can be understood through the lens of electrophilic aromatic substitution mechanisms adapted for acetal-containing substrates. Upon exposure to strong mineral acids like sulfuric acid, the dimethoxy acetal moiety undergoes hydrolysis to reveal a reactive aldehyde or iminium species in situ, which acts as a potent electrophile. The electron-rich aromatic ring of the phenyl group attached to the imine nitrogen then attacks this electrophilic center, closing the six-membered ring to form the dihydroisoquinoline skeleton, which subsequently aromatizes to the stable 1-phenylisoquinoline product. This mechanism avoids the harsh dehydrating conditions of POCl3, relying instead on the intrinsic reactivity of the protonated acetal/imine system to drive ring closure with high atom economy.
Following the construction of the heterocyclic core, the stereochemical integrity of the final product is established through diastereomeric salt formation. The interaction between the basic nitrogen of the racemic 1-phenyl-1,2,3,4-tetrahydroisoquinoline and the carboxylic acid group of L-mandelic acid creates two distinct diastereomeric salts with different solubility profiles in the methanol/toluene solvent system. The (S)-enantiomer salt exhibits significantly lower solubility at reduced temperatures, precipitating out of the solution as high-purity crystals while the (R)-enantiomer remains largely in the mother liquor. This thermodynamic control over crystallization allows for the efficient separation of enantiomers without the need for expensive chiral chromatography or enzymatic kinetic resolution, ensuring that the final high-purity Solifenacin intermediate meets stringent regulatory specifications for optical purity.
How to Synthesize (S)-1-Phenyl-1,2,3,4-Tetrahydroisoquinoline Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the exothermic cyclization and the high-pressure hydrogenation steps, to ensure both safety and reproducibility. The initial condensation should be monitored via water collection to confirm complete conversion to the Schiff base, which can be telescoped directly into the next step to minimize handling losses. The cyclization reaction demands precise temperature control to prevent degradation of the sensitive imine intermediate while ensuring sufficient energy for ring closure, typically requiring heating to 150-160°C for several hours. Detailed standardized operating procedures for each unit operation, including quench protocols and crystallization cooling rates, are critical for maintaining batch-to-batch consistency in a GMP environment.
- Condense benzophenone with aminoacetaldehyde dimethyl acetal in toluene under reflux to form the Schiff base intermediate.
- Perform acid-catalyzed cyclization using concentrated sulfuric or phosphoric acid at elevated temperatures to generate 1-phenylisoquinoline.
- Reduce the isoquinoline ring using Pd/C or Raney Nickel under hydrogen pressure to obtain the racemic tetrahydroisoquinoline.
- Resolve the racemate using L-mandelic acid in a methanol/toluene system to isolate the target (S)-enantiomer with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this benzophenone-based route offers profound advantages in terms of raw material security and total cost of ownership. By shifting away from specialized halogenated heterocycles and hazardous phosphorylating agents, manufacturers can source feedstocks from a broader base of commodity chemical suppliers, thereby mitigating the risk of supply disruptions caused by regulatory crackdowns on hazardous material transport. The elimination of phosphorus oxychloride not only removes a significant safety hazard from the plant floor but also drastically reduces the volume of hazardous waste requiring specialized disposal, leading to substantial operational expenditure savings in waste management and environmental compliance reporting.
- Cost Reduction in Manufacturing: The utilization of benzophenone and aminoacetaldehyde dimethyl acetal represents a significant departure from expensive custom-synthesized building blocks, as these are high-volume commodity chemicals with stable pricing structures. The replacement of tartaric acid with L-mandelic acid further drives down costs by improving the recovery yield of the desired isomer, meaning less raw material is wasted in the mother liquor and fewer recrystallization cycles are needed to achieve target purity. Additionally, the ability to use Raney Nickel as an alternative to Palladium on Carbon for the hydrogenation step provides flexibility in catalyst procurement, allowing procurement teams to optimize spend based on fluctuating precious metal markets without compromising reaction efficiency.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against moisture and oxygen during the initial stages reduces the dependency on highly specialized, dry-box-equipped facilities, enabling production across a wider range of manufacturing sites globally. The simplified workup procedures, which avoid the complex neutralization and extraction steps associated with phosphorus waste, shorten the overall cycle time per batch, allowing for increased asset utilization and faster response to market demand spikes. This operational agility is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their key inputs without delay.
- Scalability and Environmental Compliance: The absence of corrosive gases and the use of standard mineral acids make this process inherently safer to scale from pilot plants to multi-ton commercial reactors, minimizing the engineering controls required for containment. The reduction in phosphorus-containing effluent aligns perfectly with increasingly stringent global environmental regulations, future-proofing the supply chain against potential bans or taxes on hazardous chemical discharges. This green chemistry approach not only enhances the corporate sustainability profile of the manufacturer but also ensures long-term regulatory continuity, securing the supply line for critical urological medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway, derived directly from the comparative data and experimental examples provided in the patent literature. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this technology into their existing supply networks and quality systems.
Q: How does this new method improve safety compared to traditional POCl3 routes?
A: The novel process eliminates the use of phosphorus oxychloride (POCl3) and phosphorus pentoxide (P2O5), which are highly corrosive and generate significant phosphorus-containing wastewater. By utilizing concentrated sulfuric or phosphoric acid for cyclization, the process significantly reduces hazardous waste generation and operational risks associated with handling aggressive chlorinating agents.
Q: Why is L-mandelic acid preferred over tartaric acid for resolution?
A: According to the patent data, L-mandelic acid offers superior crystallization kinetics compared to traditional tartaric acid. This results in faster crystal growth, higher recovery yields of the desired (S)-enantiomer, and a shortened overall production cycle, thereby enhancing manufacturing efficiency and reducing solvent consumption per unit of product.
Q: What are the scalability advantages of using benzophenone as a starting material?
A: Benzophenone and aminoacetaldehyde dimethyl acetal are commodity chemicals that are readily available in bulk quantities at low cost. Unlike specialized halogenated isoquinolines required for Grignard or coupling routes, these feedstocks do not require stringent anhydrous or oxygen-free conditions for the initial steps, making the process inherently more robust for multi-ton commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1-Phenyl-1,2,3,4-Tetrahydroisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is paramount for the long-term viability of pharmaceutical supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the benzophenone route are fully realized in practical, GMP-compliant manufacturing. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of (S)-1-phenyl-1,2,3,4-tetrahydroisoquinoline meets the exacting standards required for Solifenacin synthesis.
We invite global partners to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener methodology for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply of this critical chiral intermediate remains secure, cost-effective, and compliant with the highest industry standards.
