Scalable Synthesis of Modified Fluorouracil Derivatives for Advanced Oligonucleotide Manufacturing
Scalable Synthesis of Modified Fluorouracil Derivatives for Advanced Oligonucleotide Manufacturing
In the rapidly evolving landscape of antiviral drug discovery and diagnostic probe development, the demand for high-purity, structurally precise nucleoside analogues has never been more critical. Patent CN102532227A introduces a groundbreaking methodology for the synthesis of 4-(2,4,6-trimethylphenoxy)-5-fluorouracil derivatives, specifically targeting the needs of researchers focused on HIV-1 virus DNA deamination detection. This technical disclosure represents a significant leap forward in nucleoside chemistry, offering a streamlined pathway that bypasses the historical bottlenecks of moisture sensitivity and low yield often associated with traditional triazole-mediated substitutions. For global procurement leaders and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this patented route is essential for securing a stable supply chain of complex oligonucleotide building blocks.
The core innovation lies in the strategic manipulation of the uracil ring's 4-position. Historically, introducing bulky aryl groups at this position required harsh conditions or unstable intermediates that compromised the integrity of the sugar moiety. The disclosed method utilizes a mild yet effective activation strategy using phosphorus oxychloride, followed by a clean nucleophilic attack by 2,4,6-trimethylphenol. This approach not only preserves the stereochemical integrity of the 2'-deoxyribose sugar but also ensures that the fluorine atom at the 5-position remains intact, a crucial feature for the biological activity of the final oligonucleotide therapeutic or diagnostic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for modifying the 4-position of uracil derivatives have long been plagued by the reliance on triazole intermediates. As noted in the background art of the patent, previous methods often employed triazole reagents to activate the uracil ring prior to substitution. However, triazoles are notoriously hygroscopic and prone to degradation upon exposure to atmospheric moisture, leading to inconsistent reaction outcomes and variable batch-to-batch quality. Furthermore, the removal of triazole byproducts often requires extensive purification steps, such as repeated column chromatography, which drastically reduces overall throughput and increases the cost of goods sold (COGS). In an industrial setting, the instability of these intermediates poses significant safety and logistical challenges, making the scale-up of such processes risky and economically unviable for commercial manufacturing.
The Novel Approach
The methodology presented in CN102532227A fundamentally reengineers this transformation by eliminating the triazole step entirely. Instead, it employs a direct activation of the 4-position using phosphorus oxychloride in the presence of a base, creating a highly reactive chloro-intermediate in situ. This intermediate is immediately trapped by 2,4,6-trimethylphenol, resulting in the desired 4-aryl ether linkage with high efficiency. This novel approach offers distinct advantages in terms of operational simplicity and robustness. The reaction conditions are mild, typically ranging from -5°C to 35°C, which minimizes the risk of side reactions such as glycosidic bond cleavage or epimerization. By simplifying the synthetic sequence and utilizing stable, commercially available reagents, this method significantly enhances the feasibility of cost reduction in pharmaceutical intermediate manufacturing, making it an attractive option for large-scale production facilities.
Mechanistic Insights into Phosphorus-Mediated Nucleophilic Substitution
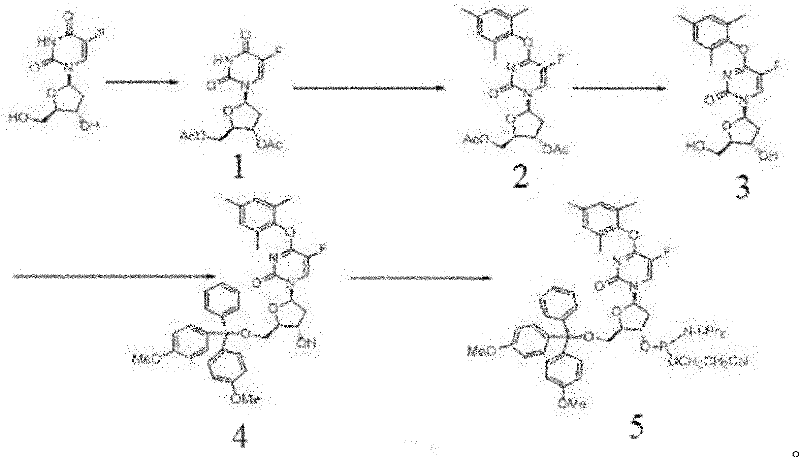
To fully appreciate the technical superiority of this route, one must delve into the mechanistic details of the phosphorus-mediated activation. The process begins with the protection of the 3' and 5' hydroxyl groups of 5-fluoro-2'-deoxyuridine using acetic anhydride and a catalytic amount of 4-dimethylaminopyridine (DMAP). This acetylation step is crucial as it prevents unwanted phosphorylation or substitution at the sugar hydroxyls during the subsequent activation of the nucleobase. Once the sugar is protected, the uracil ring is activated by phosphorus oxychloride (POCl3). The oxygen at the 4-position attacks the phosphorus atom, displacing a chloride ion and forming a reactive imidoyl chloride species. This species is highly electrophilic at the C4 carbon, rendering it susceptible to nucleophilic attack.
The introduction of 2,4,6-trimethylphenol (mesitol) serves as the nucleophile in this substitution. The steric bulk of the two ortho-methyl groups on the phenol ring provides a unique electronic and steric environment that stabilizes the resulting ether linkage while preventing further unwanted reactions at the phenolic oxygen. The reaction proceeds through a tetrahedral intermediate which collapses to expel the phosphate species, restoring the aromaticity of the uracil ring and establishing the C-O-C bond. Following this key transformation, the acetyl protecting groups are removed under mild basic conditions using methanolamine, regenerating the free hydroxyls necessary for subsequent oligonucleotide synthesis. This precise control over reactivity ensures that the final product possesses the high-purity specifications required for sensitive diagnostic applications.
How to Synthesize 4-(2,4,6-Trimethylphenoxy)-5-Fluorouracil Derivative Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions and precise temperature control to maximize yield and minimize impurities. The process is divided into five distinct stages: protection, activation/substitution, deprotection, 5'-protection, and phosphitylation. Each stage builds upon the previous one, requiring careful monitoring of reaction progress via TLC or HPLC to ensure complete conversion before proceeding. The detailed standardized synthesis steps below outline the specific reagent ratios, solvent choices, and workup procedures necessary to achieve the high yields reported in the patent data, serving as a critical reference for process chemists aiming to replicate this route.
- Protect the hydroxyl groups of 5-fluoro-2'-deoxyuridine using acetic anhydride and DMAP to form the diacetyl intermediate.
- Activate the 4-position of the uracil ring using phosphorus oxychloride, followed by nucleophilic substitution with 2,4,6-trimethylphenol.
- Remove acetyl protecting groups, protect the 5'-hydroxyl with DMTr chloride, and finally convert to the phosphoramidite using standard reagents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from legacy triazole-based methods to this novel phosphorus-mediated route offers substantial strategic benefits beyond mere technical elegance. The elimination of moisture-sensitive reagents translates directly into reduced waste and lower handling costs, as specialized drying equipment and inert atmosphere gloveboxes are less critical for the bulk of the synthesis. Furthermore, the use of commodity chemicals like acetic anhydride, phosphorus oxychloride, and mesitol ensures a resilient supply chain that is not dependent on niche, high-cost specialty reagents. This stability in raw material sourcing is paramount for maintaining continuous production schedules and mitigating the risk of supply disruptions in the global market for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis significantly lowers the operational expenditure associated with purification. By avoiding the formation of difficult-to-remove triazole byproducts, the need for extensive chromatographic purification is reduced, allowing for more efficient crystallization or extraction protocols. Additionally, the higher overall yield achieved through this direct substitution method means that less starting material is required to produce the same amount of final product, effectively driving down the unit cost. The ability to perform reactions at near-ambient temperatures also reduces energy consumption compared to processes requiring cryogenic cooling or high-temperature reflux, contributing to a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a key metric for supply chain reliability. Since the reagents used are stable and widely available from multiple global suppliers, the risk of single-source dependency is minimized. This diversification of the supply base allows procurement teams to negotiate better terms and secure long-term contracts with confidence. Moreover, the simplified workflow reduces the total cycle time from raw material intake to finished goods, enabling faster response times to fluctuating market demands and ensuring that critical research projects are not delayed by material shortages.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route is inherently safer and easier to scale. The avoidance of potentially explosive or highly toxic triazole derivatives reduces the regulatory burden and safety risks associated with large-scale operations. The solvents employed, primarily dichloromethane and pyridine, are well-understood in industrial settings with established recovery and recycling protocols. This alignment with green chemistry principles not only facilitates smoother regulatory approvals but also enhances the corporate sustainability profile of the manufacturing entity, making it a preferred partner for environmentally conscious multinational corporations seeking commercial scale-up of complex polymer additives and pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this specific fluorouracil derivative. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and performance of the described methodology. Understanding these details is vital for technical teams evaluating the integration of this intermediate into their existing oligonucleotide synthesis workflows.
Q: What is the primary advantage of this synthesis route over traditional triazole methods?
A: The primary advantage is the avoidance of moisture-sensitive triazole intermediates. By directly substituting the 4-position with 2,4,6-trimethylphenoxy using phosphorus oxychloride, the process becomes more robust, yields higher purity, and eliminates the instability issues associated with triazole reagents.
Q: Is this intermediate suitable for large-scale oligonucleotide production?
A: Yes, the patent explicitly states the method is suitable for large-scale production. The use of common solvents like dichloromethane and pyridine, along with stable reagents like acetic anhydride and phosphorus oxychloride, facilitates easy scale-up from laboratory to industrial tonnage.
Q: What are the critical quality control parameters for this derivative?
A: Critical parameters include the purity of the final phosphoramidite (verified by NMR and HPLC), the absence of residual phosphorus oxychloride, and the correct stereochemistry at the phosphorus center. The patent details specific NMR shifts (1H, 13C, 19F, 31P) that serve as definitive identity markers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(2,4,6-Trimethylphenoxy)-5-Fluorouracil Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of next-generation antiviral therapeutics relies heavily on the availability of superior quality building blocks. Our team of expert process chemists has extensively analyzed the route disclosed in CN102532227A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering this complex nucleoside analogue with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for clinical and diagnostic applications. Our state-of-the-art facilities are equipped to handle the specific reactivity profiles of phosphoramidite chemistry, ensuring product stability and integrity throughout the manufacturing and shipping process.
We invite R&D directors and procurement specialists to collaborate with us to optimize their supply chains for modified nucleosides. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-efficient chemical partner in the industry.
