Advanced Copper-Catalyzed Synthesis of 6-Methylisoindolo[2,1-a]quinoxaline Derivatives for Pharmaceutical Applications
Introduction to Novel Isoindoloquinoxaline Synthesis
The field of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical industry, particularly for complex nitrogen-containing heterocycles. Patent CN114437072B introduces a groundbreaking method for the synthesis of 6-methylisoindolo[2,1-a]quinoxaline compounds, a scaffold known for its significant biological activities including anticancer, antimalarial, and antibacterial properties. This technology represents a paradigm shift from traditional, hazardous synthetic routes to a more sustainable, copper-catalyzed cyclization process. By leveraging an in situ generated copper catalyst system with specialized oxazoline ligands, this method achieves high reaction activity and excellent yields under remarkably mild conditions. For R&D directors and procurement specialists, this innovation offers a reliable pathway to access high-purity pharmaceutical intermediates that were previously difficult or expensive to manufacture.
![General structural formula of 6-methylisoindolo[2,1-a]quinoxaline compounds showing variable substituents R1-R4](/insights/img/6-methylisoindolo-quinoxaline-synthesis-pharma-supplier-20260309042719-01.png)
The core innovation lies in the efficient construction of the tetracyclic isoindoloquinoxaline skeleton through the cyclization of propargyl compounds and diamine compounds. Unlike legacy methods that struggled with low efficiency and toxic reagents, this new approach ensures robust performance across a wide range of substrates. The ability to introduce diverse functional groups at the R1, R2, R3, and R4 positions allows for the rapid generation of compound libraries for drug discovery programs. This flexibility is crucial for medicinal chemists aiming to optimize lead compounds for potency and selectivity. Furthermore, the operational simplicity of the reaction—requiring standard laboratory equipment and common solvents—makes it an attractive candidate for immediate technology transfer and commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoindolo[2,1-a]quinoxaline derivatives has been plagued by significant technical and safety challenges that hindered their widespread adoption in drug development. Conventional literature describes methods involving the reflux of alkynylbenzaldehyde and o-phenylenediamine in nitrobenzene, a process that not only requires extremely harsh thermal conditions but also results in disappointingly low yields after prolonged reaction times of up to two days. Another existing route relies on the use of ortho-dialdehydes and ortho-diynes to generate intermediates, but critically, this pathway necessitates the use of potassium cyanide (KCN), a highly toxic and regulated substance that poses severe safety risks and environmental disposal issues. Additionally, some methods involve long-term heating with formic acid to construct the tetracyclic core, which lacks atom economy and often leads to complex mixtures requiring difficult purification. These limitations create bottlenecks in the supply chain, increasing costs and delaying project timelines for pharmaceutical companies.
The Novel Approach
In stark contrast, the method disclosed in CN114437072B offers a streamlined, catalytic solution that effectively bypasses these historical obstacles. The new protocol utilizes a copper-catalyzed cyclization reaction between readily available propargyl compounds and diamine compounds, eliminating the need for toxic cyanides or harsh nitrobenzene solvents. The reaction proceeds efficiently in polar or nonpolar solvents such as methanol, ethanol, or toluene, with temperatures ranging from a mild 25°C to 80°C. This dramatic reduction in energy consumption and hazard potential translates directly into cost reduction in pharmaceutical intermediate manufacturing. The general reaction scheme illustrates the elegant convergence of the two building blocks into the target heterocycle with high atom economy.
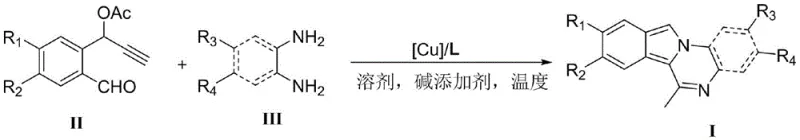
Furthermore, the novel approach demonstrates exceptional versatility, accommodating a broad spectrum of substituents on both the alkyne and diamine partners. Whether introducing electron-withdrawing halogens or electron-donating alkoxy groups, the catalytic system maintains high activity, ensuring consistent quality across different analogues. This reliability is a key factor for supply chain heads who require predictable outcomes when scaling processes from gram to kilogram quantities. By replacing multi-step, low-yielding sequences with a direct, one-pot cyclization, this method significantly simplifies the manufacturing workflow, reducing the overall carbon footprint and waste generation associated with the production of these valuable bioactive scaffolds.
Mechanistic Insights into Copper-Catalyzed Cyclization
The success of this synthesis hinges on the precise interplay between the copper catalyst and the chiral or achiral oxazoline ligands. The catalytic cycle likely initiates with the coordination of the copper salt, such as Cu(OAc)2·H2O or CuCl, with the bidentate oxazoline ligand to form an active metal-ligand complex in situ. This complex activates the terminal alkyne of the propargyl substrate, facilitating nucleophilic attack by the amine groups of the diamine component. The specific geometry imposed by the ligand, as seen in structures like L-1 through L-4, plays a critical role in stabilizing the transition states and enhancing the reaction rate. The use of bases like diisopropylethylamine (iPr2NEt) further assists in deprotonation steps necessary for the cyclization cascade to proceed smoothly. Understanding this mechanism allows chemists to fine-tune reaction parameters for optimal performance.
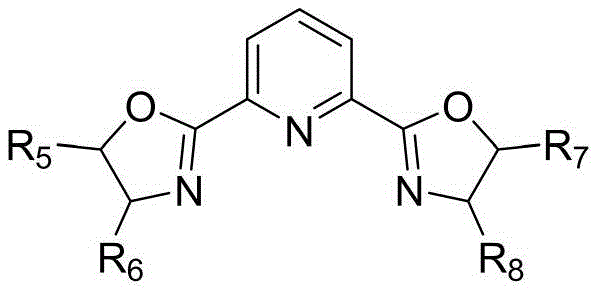
Impurity control is another critical aspect addressed by this mechanistic design. Traditional methods often produced complex mixtures due to non-selective polymerization of alkynes or side reactions with the solvent. The copper-ligand system described here exhibits high chemoselectivity, directing the reaction specifically towards the formation of the isoindoloquinoxaline ring system. The mild conditions prevent the decomposition of sensitive functional groups that might be present on the substrate, thereby preserving the integrity of the molecule and simplifying downstream purification. For R&D teams, this means a cleaner crude reaction profile and higher isolated purity, which is essential for meeting the stringent specifications required for clinical grade materials. The ability to suppress side reactions while maintaining high conversion rates is a testament to the sophistication of this catalytic design.
How to Synthesize 6-Methylisoindolo[2,1-a]quinoxaline Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward and follows a logical sequence of operations designed for maximum efficiency. The process begins with the preparation of the catalyst solution, followed by the addition of substrates and base, and concludes with a standard workup procedure. The patent details specific molar ratios and conditions that have been optimized to ensure reproducibility. For instance, a catalyst loading of 5 mol% is often sufficient to drive the reaction to completion within 12 hours. The detailed standardized synthesis steps below provide a clear roadmap for technical teams to replicate these results and adapt them to specific target molecules.
- Prepare the metallic copper catalyst by stirring copper salt and oxazoline ligand in a reaction medium under nitrogen protection for 1-2 hours.
- Dissolve the propargyl compound, diamine compound, and base additive in the reaction medium and add to the catalyst solution.
- Stir the mixture at 25-80°C for 1-24 hours, then concentrate, purify via silica gel chromatography, and dry to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers compelling advantages that address the core concerns of procurement managers and supply chain directors. The shift from hazardous, multi-step legacy processes to a direct, catalytic route fundamentally alters the cost structure and risk profile of producing these intermediates. By utilizing commodity chemicals as starting materials and avoiding expensive or regulated reagents, the overall cost of goods sold (COGS) can be significantly optimized. Moreover, the operational safety improvements reduce the regulatory burden and insurance costs associated with manufacturing, making the supply chain more resilient and sustainable in the long term.
- Cost Reduction in Manufacturing: The elimination of toxic reagents like KCN and harsh solvents like nitrobenzene removes the need for specialized containment equipment and costly waste treatment protocols. The use of inexpensive copper salts and simple ligands further drives down raw material costs. Additionally, the high yields achieved (up to 88% in optimized examples) mean less raw material is wasted per unit of product, directly improving the economic efficiency of the process. This logical deduction of cost savings makes the technology highly attractive for budget-conscious procurement strategies.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted propargyl alcohols and phenylenediamines, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvents, ensures that production is not easily disrupted by minor fluctuations in utility supplies or environmental conditions. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical customers who depend on just-in-time delivery models for their drug development pipelines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in standard solvents like methanol and ethanol which are easy to recover and recycle on a large scale. The absence of heavy metal contaminants (other than trace copper which is easily removed) and toxic byproducts simplifies the environmental compliance landscape. This facilitates smoother regulatory approvals for manufacturing sites and aligns with the growing industry demand for green chemistry practices, thereby future-proofing the supply chain against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this method for their own production needs. Understanding these nuances helps in making informed decisions about technology adoption and resource allocation.
Q: What are the advantages of this copper-catalyzed method over traditional synthesis routes?
A: Traditional methods often require harsh conditions like refluxing in nitrobenzene for days or using highly toxic reagents like KCN. This novel copper-catalyzed approach operates under mild conditions (25-80°C), uses readily available raw materials, avoids toxic cyanides, and achieves significantly higher yields up to 88%.
Q: What is the substrate scope for this cyclization reaction?
A: The method demonstrates wide substrate applicability. It tolerates various substituents on both the propargyl and diamine components, including halogens (Cl, Br, F), alkoxy groups (OMe), and fused ring systems, allowing for the synthesis of diverse 6-methylisoindolo[2,1-a]quinoxaline derivatives.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes inexpensive copper salts and simple ligands, operates in common solvents like methanol or ethanol, and involves straightforward workup procedures (concentration and chromatography), making it highly viable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methylisoindolo[2,1-a]quinoxaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis for the pharmaceutical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including inert atmosphere operations and precise temperature control, guaranteeing stringent purity specifications for every batch. With our rigorous QC labs and commitment to quality, we deliver high-purity pharmaceutical intermediates that meet the highest international standards.
We invite you to collaborate with us to leverage this advanced synthetic route for your drug development projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to clinical success while optimizing your manufacturing costs.
