Scalable Synthesis of 3H-1,2-Dithiole 2,2-Dioxides for Advanced Pharmaceutical Intermediates
Introduction to Next-Generation Sulfur-Sulfur Bond Construction
The development of efficient methodologies for constructing sulfur-sulfur bonds remains a critical frontier in modern organic synthesis, particularly for the production of high-value pharmaceutical intermediates and agrochemical active ingredients. Patent CN111484476B discloses a groundbreaking approach to synthesizing 3-hydro-1,2-dithio-2,2-dioxide derivatives, a structural motif prevalent in bioactive molecules such as the antioxidant Lipoic acid and the antitumor agent Leinamycin. This technology leverages the unique reactivity of sulfur-containing ylides reacting with alkylsulfonyl chlorides to form the target heterocyclic core with exceptional efficiency. By utilizing N,N-diisopropylethylamine (DIPEA) as a base in dichloromethane, the process achieves rapid cyclization at room temperature, eliminating the need for energy-intensive heating or cryogenic cooling. For R&D directors and process chemists, this represents a significant leap forward in accessing complex sulfur-rich scaffolds that were previously difficult to prepare with high purity and selectivity.
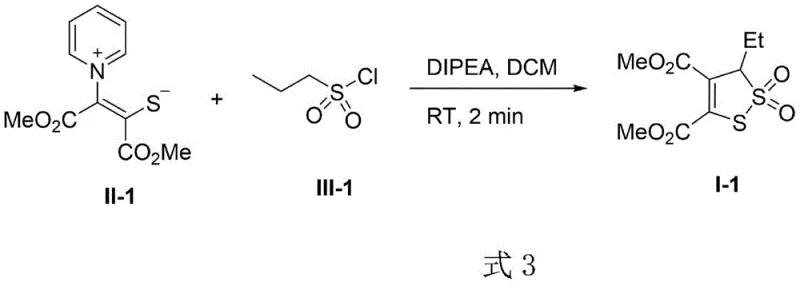
The strategic importance of this invention lies in its ability to bypass the limitations of classical disulfide synthesis, offering a direct route to functionalized 1,2-dithiole 2,2-dioxides. These compounds serve as versatile building blocks for drug discovery programs targeting oxidative stress pathways and enzyme inhibition. The patent details a robust protocol where the sulfur-sulfur bond is formed in a single step from readily available precursors, ensuring that the supply chain for these critical intermediates remains resilient and cost-effective. As the demand for sulfur-containing APIs continues to grow, mastering this specific cyclization chemistry provides a competitive advantage in bringing new therapeutic candidates to clinical trials faster and more reliably.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of asymmetric disulfide bonds has been plagued by significant synthetic challenges that hinder scalability and purity profiles. Traditional methods often rely on the oxidation of thiols, which introduces severe handling issues due to the notorious foul odor and toxicity associated with low molecular weight thiols. Furthermore, oxidative coupling frequently lacks selectivity, leading to the formation of symmetrical disulfide byproducts that are difficult to separate from the desired asymmetric target, thereby compromising the overall yield and requiring extensive purification resources. Alternative strategies involving metal-catalyzed cross-coupling or the use of elemental sulfur often necessitate harsh reaction conditions, expensive transition metal catalysts, and rigorous exclusion of moisture and oxygen. These factors collectively increase the cost of goods sold (COGS) and create bottlenecks in the manufacturing workflow, making it difficult for procurement teams to secure consistent, high-quality supplies of sulfur-rich intermediates for commercial production.
The Novel Approach
In stark contrast, the methodology described in CN111484476B utilizes a sulfur-containing ylide and a sulfonyl chloride to construct the sulfur-sulfur bond under remarkably mild and operationally simple conditions. This novel approach completely avoids the use of malodorous thiols and eliminates the need for precious metal catalysts, thereby simplifying the impurity profile and reducing the burden on downstream purification processes. The reaction proceeds rapidly at room temperature (25°C) within a timeframe of merely 2 to 10 minutes, demonstrating exceptional kinetic efficiency that is rarely seen in heterocyclic synthesis. Moreover, the process does not require inert gas protection, allowing reactions to be conducted in standard glassware without specialized gloveboxes or nitrogen lines. This dramatic simplification of the experimental setup translates directly into lower capital expenditure for manufacturing facilities and enhanced safety for operators, positioning this technology as a superior alternative for the industrial-scale production of complex sulfur heterocycles.
Mechanistic Insights into Ylide-Mediated S-S Bond Formation
The mechanistic pathway of this transformation involves a nucleophilic attack by the sulfur atom of the stabilized ylide onto the sulfur center of the sulfonyl chloride, initiating the formation of the critical sulfur-sulfur bond. The presence of electron-withdrawing groups (EWG) on the ylide carbon stabilizes the negative charge, enhancing the nucleophilicity of the adjacent sulfur anion while maintaining the structural integrity of the precursor. Upon addition of the base, N,N-diisopropylethylamine, the reaction mixture facilitates the displacement of the chloride ion, generating a reactive sulfenyl intermediate that undergoes rapid intramolecular cyclization. This cascade sequence effectively constructs the five-membered 1,2-dithiole ring system with the concomitant formation of the sulfone moiety (S=O bonds), resulting in the final 3-hydro-1,2-dithio-2,2-dioxide architecture. The elegance of this mechanism lies in its atom economy and the avoidance of high-energy intermediates that typically lead to decomposition or side reactions in traditional sulfur chemistry.
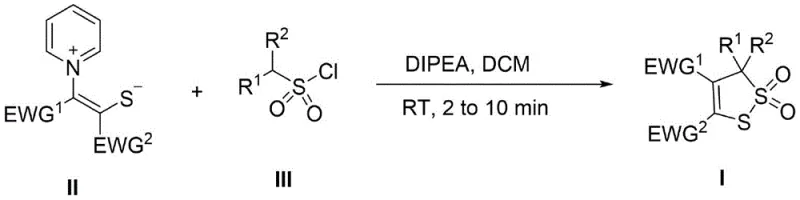
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the generation of symmetric disulfide contaminants that plague thiol oxidation routes. Since the two sulfur atoms originate from distinct chemical environments (the ylide and the sulfonyl chloride), the probability of homocoupling is inherently suppressed, leading to a cleaner reaction profile. The use of DIPEA as a non-nucleophilic base further ensures that side reactions such as ester hydrolysis or elimination are kept to a minimum, preserving the integrity of sensitive functional groups like esters and ketones present on the substrate. For quality control teams, this means that the crude product often requires minimal processing to meet stringent purity specifications, reducing the consumption of silica gel and solvents during chromatography. Understanding this mechanistic nuance allows process engineers to fine-tune stoichiometry and addition rates to maximize throughput while maintaining the high selectivity required for GMP manufacturing of pharmaceutical intermediates.
How to Synthesize 3H-1,2-Dithiole 2,2-Dioxides Efficiently
The practical implementation of this synthesis is designed for ease of execution, requiring only standard laboratory equipment and commercially available reagents to achieve high yields. The protocol dictates dissolving the sulfur-containing ylide and the alkylsulfonyl chloride in dichloromethane, followed by the addition of DIPEA, with the reaction reaching completion in minutes at ambient temperature. This straightforward procedure eliminates the need for complex temperature programming or extended reaction times, making it highly amenable to both small-scale discovery chemistry and large-scale pilot plant operations. For detailed operational parameters regarding stoichiometry, solvent concentration, and workup procedures, please refer to the standardized synthesis guide provided below which outlines the exact steps validated in the patent examples.
- Dissolve the sulfur-containing ylide (Compound II), alkylsulfonyl chloride (Compound III), and N,N-diisopropylethylamine (DIPEA) in dichloromethane solvent at room temperature.
- Monitor the reaction progress via thin-layer chromatography (TLC) until the starting material (Compound II) has completely disappeared, typically within 2 to 10 minutes.
- Remove the organic solvent under reduced pressure and purify the crude residue using silica gel column chromatography with a petroleum ether and ethyl acetate mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits that directly impact the bottom line and operational resilience. The elimination of precious metal catalysts removes a major cost driver and supply risk, as the prices of metals like palladium or rhodium can be volatile and their availability subject to geopolitical constraints. Furthermore, the ability to run the reaction at room temperature without inert gas protection significantly reduces energy consumption and infrastructure requirements, allowing for production in facilities that may not have specialized nitrogen manifolds or cryogenic capabilities. This flexibility enhances supply chain continuity by enabling manufacturing across a broader range of sites, reducing the risk of disruption due to facility-specific limitations. The simplified post-treatment process, which involves basic solvent removal and silica gel chromatography, minimizes waste generation and shortens the overall production cycle time, leading to faster turnaround for customer orders.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commodity-grade starting materials such as sulfonyl chlorides and simple amines, which are readily sourced from global chemical suppliers. By avoiding the use of expensive transition metal catalysts, the process eliminates the costly and time-consuming step of heavy metal scavenging, which is often mandatory for pharmaceutical intermediates to meet regulatory limits. Additionally, the high reaction efficiency and short reaction time mean that reactor occupancy is minimized, allowing for higher throughput per unit of time and better utilization of existing manufacturing assets. These factors combine to deliver a significantly lower cost of goods compared to traditional multi-step or metal-catalyzed routes, providing a competitive pricing advantage in the marketplace.
- Enhanced Supply Chain Reliability: The robustness of this chemistry ensures a stable and predictable supply of critical sulfur-containing intermediates, which is essential for maintaining uninterrupted production of downstream APIs. Since the reagents are stable and do not require special storage conditions like extreme cold or strict moisture exclusion, inventory management becomes simpler and less prone to spoilage or degradation. The tolerance of the reaction to ambient conditions also means that production is less susceptible to delays caused by utility failures or equipment maintenance related to inert gas systems. This reliability allows supply chain planners to optimize inventory levels and reduce safety stock requirements, freeing up working capital while ensuring that customer demand is met consistently and on time.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is straightforward due to the absence of exothermic hazards or gas evolution that typically complicate batch reactor operations. The mild conditions and lack of toxic thiol reagents improve the environmental, health, and safety (EHS) profile of the manufacturing process, reducing the burden on waste treatment facilities and lowering the risk of operator exposure. This alignment with green chemistry principles not only facilitates regulatory approval but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies. The ease of purification via standard chromatography further ensures that the process can be adapted to continuous flow or large batch modes without compromising product quality or environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfur-sulfur bond construction technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for process development teams. Understanding these specifics is crucial for evaluating the feasibility of integrating this route into your existing manufacturing portfolio.
Q: What are the primary advantages of this sulfur-sulfur bond construction method over traditional thiol oxidation?
A: Unlike traditional thiol oxidation which often suffers from poor selectivity (producing symmetric disulfides) and unpleasant odors, this novel method utilizes stable sulfur-containing ylides and commercially available sulfonyl chlorides. It achieves high chemoselectivity for asymmetric disulfide structures under mild conditions without the need for malodorous thiols or harsh oxidants.
Q: Does this synthesis require inert gas protection or specialized equipment?
A: No, one of the most significant operational benefits of this patented process is that it does not require inert gas protection such as nitrogen or argon. The reaction proceeds efficiently in an open flask at room temperature (25°C), significantly simplifying the equipment requirements and reducing operational complexity for large-scale manufacturing.
Q: What is the typical reaction time and yield for this transformation?
A: The reaction is exceptionally fast, typically completing within 2 to 10 minutes at room temperature. Experimental data demonstrates consistently high yields, with specific examples achieving up to 99% conversion, making it highly suitable for time-sensitive production schedules and cost-effective manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3H-1,2-Dithiole 2,2-Dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel synthesis route for the production of high-purity 3H-1,2-Dithiole 2,2-Dioxide derivatives and are fully equipped to support your commercialization goals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence means that we can navigate the complexities of sulfur chemistry with expertise, delivering products that are free from problematic impurities and ready for the next stage of synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative technology can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free, room-temperature process. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds and to request route feasibility assessments for your custom targets. Let us be your trusted partner in advancing your sulfur-containing drug candidates from the lab bench to the market with speed and efficiency.
