Advanced Copper-Catalyzed Synthesis of 3-Iodo-4-Amino Maleimide Intermediates for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of 3-Iodo-4-Amino Maleimide Intermediates for Commercial Scale-Up
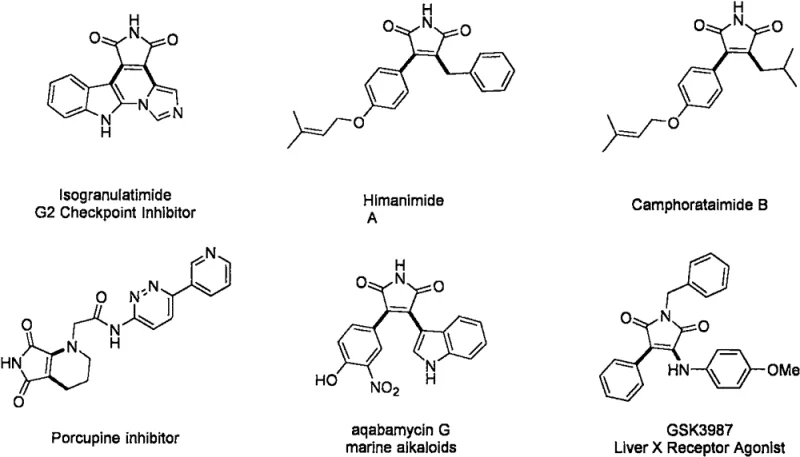
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN112724063A introduces a groundbreaking preparation method for 3-iodo-4-amido maleimide compounds, utilizing a transition metal copper-catalyzed radical tandem reaction. This technology addresses the critical need for constructing 3,4-bifunctional maleimides, which are privileged structures found in numerous marine natural alkaloids and potent antitumor active molecules. By leveraging an in-situ generated iodoamination reagent under oxygen conditions, this method achieves high product yields and exceptional purity, marking a significant advancement over conventional multi-step syntheses. For R&D teams and procurement strategists, this patent represents a viable pathway to access high-value intermediates like Isogranulatimide and Himanimide A derivatives with improved economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3,4-disubstituted maleimides has been fraught with synthetic challenges, often requiring multiple protection and deprotection steps or the use of unstable halogenating agents. Traditional amine halogenation reactions typically rely on relatively stable sulfonamide chlorides or amide chlorination reagents, which can introduce unwanted impurities and complicate the purification process. Furthermore, existing methods for introducing both amino and halo groups onto the maleimide core frequently suffer from poor regioselectivity and low atom economy. The reliance on expensive directing groups, such as 8-aminoquinoline in previous copper-catalyzed olefin functionalizations, adds significant cost and synthetic burden. These limitations hinder the rapid discovery and development of maleimide-based innovative drugs, creating a bottleneck for reliable pharmaceutical intermediate suppliers aiming to deliver cost reduction in API manufacturing.
The Novel Approach
The methodology described in CN112724063A revolutionizes this landscape by employing a direct radical tandem reaction that bypasses the need for pre-functionalized substrates. By using N-iodosuccinimide, a secondary amine, and an N-substituted maleimide as starting materials, the process generates the reactive iodoamination species in situ. This eliminates the handling of hazardous isolated reagents and streamlines the workflow into a single pot operation. The use of molecular oxygen as the oxidant further enhances the green chemistry profile of the reaction, reducing the environmental footprint associated with stoichiometric oxidants. This novel approach not only simplifies the synthetic route but also ensures high-purity pharmaceutical intermediate output, which is crucial for downstream biological testing and clinical applications.
Mechanistic Insights into Copper-Catalyzed Radical Tandem Reaction
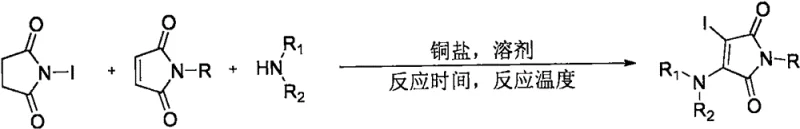
The core of this innovation lies in the unique mechanistic pathway facilitated by the transition metal copper catalyst, specifically cuprous chloride. Under oxygen conditions, the copper catalyst activates the N-iodosuccinimide and the secondary amine to generate a transient iodoamination radical species. This reactive intermediate then undergoes a radical addition to the electron-deficient double bond of the N-substituted maleimide. The subsequent oxidation and elimination steps, driven by the copper redox cycle and oxygen, restore the aromaticity or conjugation of the maleimide ring while installing the iodine and amino groups at the 3 and 4 positions respectively. This radical series reaction mechanism is highly efficient, avoiding the formation of stable by-products that typically plague ionic substitution pathways.
From an impurity control perspective, the specificity of the radical addition to the maleimide double bond ensures a clean reaction profile. The patent data highlights that the choice of solvent plays a pivotal role in stabilizing the radical intermediates and suppressing side reactions. While polar aprotic solvents like DMSO failed to produce the desired product, non-polar or weakly coordinating solvents like toluene provided an optimal environment for the radical cascade. This mechanistic understanding allows process chemists to fine-tune reaction parameters to minimize impurities such as di-iodinated species or polymerization products, ensuring that the final commercial scale-up of complex pharmaceutical intermediates meets stringent regulatory standards for purity and safety.
How to Synthesize 3-Iodo-4-Amino Maleimide Efficiently
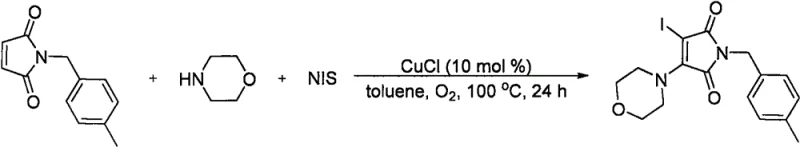
To implement this synthesis effectively, precise control over reaction stoichiometry and atmospheric conditions is required. The protocol dictates a molar ratio of 1:3:3 for the maleimide, N-iodosuccinimide, and secondary amine, ensuring complete conversion of the limiting reagent. The reaction is initiated at room temperature to allow for the formation of the catalytic complex before heating to 100°C to drive the radical propagation. Detailed standard operating procedures for this specific transformation are outlined below, providing a clear roadmap for laboratory and pilot plant execution.
- Charge a reaction tube with N-(4-methylbenzyl)maleimide, N-iodosuccinimide (3.0 equiv), cuprous chloride (10 mol%), and toluene solvent at room temperature.
- Add morpholine (3.0 equiv) to the mixture, perform evacuation-oxygen replacement three times, and stir at room temperature for 1 hour.
- Heat the reaction mixture to 100°C and stir for 23 hours under oxygen atmosphere, then cool, dilute with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed protocol offers substantial strategic benefits beyond mere technical feasibility. The shift towards using earth-abundant copper catalysts instead of precious metals like palladium or rhodium drastically reduces the raw material costs associated with catalytic loading. Furthermore, the simplicity of the workup procedure, which involves basic filtration and column chromatography without complex aqueous extractions or heavy metal scavenging steps, translates directly into reduced processing time and lower operational expenditures. This efficiency is critical for maintaining competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The utilization of cuprous chloride, an inexpensive and widely available commodity chemical, replaces costly noble metal catalysts, leading to significant savings in catalyst procurement. Additionally, the high yield of 80% reported in the preferred embodiments minimizes raw material waste, thereby optimizing the overall cost of goods sold. The elimination of specialized reagents like 8-aminoquinoline directing groups further lowers the input costs, making the process economically attractive for large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as N-iodosuccinimide and morpholine ensures a stable and resilient supply chain. Unlike proprietary reagents that may face sourcing bottlenecks, these bulk chemicals are produced by multiple vendors globally, reducing the risk of supply disruption. The robustness of the reaction conditions, tolerating standard laboratory glassware and heating mantles, means that the process can be easily transferred between different manufacturing sites without requiring specialized equipment investments.
- Scalability and Environmental Compliance: The use of toluene as a preferred solvent aligns with established industrial solvent recovery protocols, facilitating easy recycling and minimizing waste disposal costs. The reaction operates under an oxygen atmosphere rather than requiring high-pressure hydrogen or inert gases, simplifying the engineering controls needed for scale-up. This inherently safer design reduces the regulatory burden and insurance costs associated with hazardous chemical processing, supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, aimed at clarifying the operational parameters for potential licensees and manufacturing partners.
Q: What are the key advantages of this copper-catalyzed method over traditional halogenation?
A: Unlike traditional methods that often require harsh conditions or pre-functionalized reagents, this patent utilizes an in-situ generated iodoamination reagent under mild oxygen conditions, resulting in higher purity and simplified post-treatment.
Q: Which copper catalyst provides the optimal yield for this transformation?
A: Experimental data within the patent indicates that cuprous chloride (CuCl) is the superior catalyst, achieving an 80% yield, whereas other copper salts like copper acetate or bromide resulted in significantly lower yields or no reaction.
Q: Is this synthesis route scalable for industrial pharmaceutical production?
A: Yes, the process uses inexpensive and readily available raw materials such as N-iodosuccinimide and common organic solvents like toluene, combined with a cheap transition metal catalyst, making it highly suitable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Iodo-4-Amino Maleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology in accelerating the development of antitumor agents and kinase inhibitors. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-iodo-4-amino maleimide intermediate delivered meets the highest quality standards required for clinical trials and commercial drug launch.
We invite you to collaborate with our technical team to explore how this novel synthetic route can optimize your specific drug development pipeline. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume requirements. Please contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable supply of these critical building blocks for your next breakthrough therapy.
