Advanced Ruthenium-Catalyzed Synthesis of 1,2,3-Triazole Quinoxalinone Derivatives for Pharmaceutical Applications
Advanced Ruthenium-Catalyzed Synthesis of 1,2,3-Triazole Quinoxalinone Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds that serve as the core backbone for bioactive molecules. A recent technological breakthrough documented in patent CN114195792A introduces a highly efficient synthetic route for 1,2,3-triazole quinoxalinone derivatives, a class of compounds known for their potent biological activities including G-protein coupled receptor antagonism. This novel approach leverages visible-light photoredox catalysis to drive the cyclization process under exceptionally mild conditions, marking a significant departure from traditional thermal methods. By utilizing a ruthenium-based catalyst system activated by blue LED irradiation, the process achieves high conversion rates at room temperature, offering a sustainable and operationally simple alternative for the production of high-purity pharmaceutical intermediates. The strategic implementation of this technology addresses critical pain points in process chemistry, specifically regarding safety, energy consumption, and substrate versatility, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,3-triazole fused heterocyclic systems has relied heavily on multi-step sequences involving hazardous reagents and苛刻 reaction conditions that pose significant challenges for industrial scale-up. Traditional pathways often employ Sonogashira coupling followed by cyclization, or copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions which necessitate the use of sodium azide. Sodium azide is notoriously toxic and possesses explosive potential, creating severe safety liabilities and requiring specialized containment infrastructure that drives up capital expenditure. Furthermore, these conventional routes frequently suffer from narrow substrate scope, where the presence of sensitive functional groups can lead to decomposition or side reactions, resulting in lower overall yields and difficult purification processes. The reliance on stoichiometric amounts of heavy metal catalysts or harsh thermal conditions also complicates the removal of residual impurities, a critical factor when targeting regulatory compliance for active pharmaceutical ingredients.
The Novel Approach
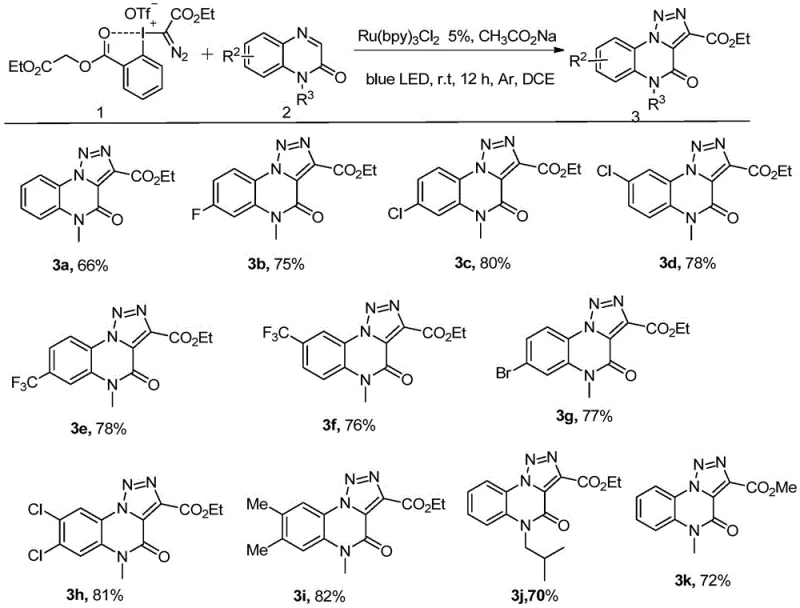
In stark contrast to these legacy methods, the patented technology utilizes a diazo trivalent iodine reagent in conjunction with a quinoxalinone derivative under the influence of a ruthenium photocatalyst and blue light. This innovative strategy enables a direct, one-pot cyclization that bypasses the need for explosive azides and reduces the step count significantly. The reaction proceeds smoothly at room temperature, eliminating the energy costs associated with heating and allowing for the preservation of thermally labile functional groups on the substrate. As illustrated in the general reaction scheme below, the transformation is clean and efficient, producing the target triazole quinoxalinone core with excellent regioselectivity. This method not only streamlines the synthetic workflow but also enhances the safety profile of the manufacturing process, aligning perfectly with modern green chemistry principles and the demands for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ru-Photocatalyzed Cyclization
The success of this transformation hinges on the unique photophysical properties of the ruthenium catalyst, typically tris(2,2'-bipyridyl)ruthenium dichloride, which acts as a potent photosensitizer upon irradiation with blue light. When exposed to photons in the visible spectrum, the ground state Ru(II) complex is excited to a long-lived metal-to-ligand charge transfer (MLCT) state, denoted as Ru(II)*. This excited species possesses sufficient redox potential to engage in single-electron transfer (SET) processes with the diazo trivalent iodine reagent, initiating the formation of reactive carbene or radical intermediates. These transient species subsequently undergo insertion or addition reactions with the nucleophilic sites on the quinoxalinone ring, driving the cyclization forward without the need for external oxidants or harsh activators. The catalytic cycle is closed by the regeneration of the ground state catalyst, ensuring that only a low loading of the precious metal is required to sustain the reaction, which is economically advantageous for large-scale operations.
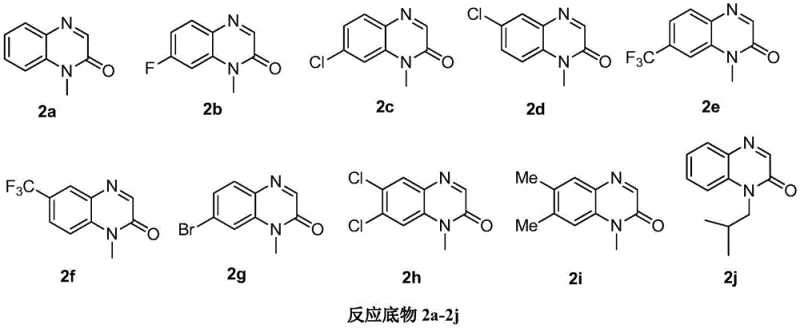
A critical advantage of this mechanistic pathway is its remarkable tolerance for diverse electronic environments on the aromatic ring, as evidenced by the broad substrate scope demonstrated in the patent data. The reaction accommodates both electron-deficient substrates, such as those bearing chloro, fluoro, bromo, and trifluoromethyl groups, and electron-rich variants with methyl substituents, delivering consistent yields across the series. This universality is visually represented by the array of successful substrates shown below, confirming that the electronic nature of the substituent does not inhibit the photocatalytic cycle. Such robustness is essential for process chemists who require a flexible platform capable of generating libraries of analogs for structure-activity relationship (SAR) studies without needing to re-optimize conditions for every new derivative. The ability to handle halogenated substrates is particularly valuable, as these groups serve as versatile handles for downstream cross-coupling reactions in medicinal chemistry campaigns.
How to Synthesize 1,2,3-Triazole Quinoxalinone Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the preparation of the diazo trivalent iodine reagent, which serves as the key carbon source for the triazole ring. The reagent is typically prepared in situ or pre-synthesized from 1-methoxy-1,2-benziodoxol-3(1H)-one and diazoacetates, ensuring high purity before introduction to the main reaction vessel. Once the reagents are combined with the ruthenium catalyst in a solvent like 1,2-dichloroethane, the mixture is subjected to blue LED irradiation while maintaining an inert atmosphere to prevent catalyst deactivation. The detailed standardized synthesis steps see the guide below for precise molar ratios and workup procedures that ensure maximum recovery of the high-purity product.
- Prepare the reaction mixture by combining the quinoxalinone derivative substrate and the diazo trivalent iodine reagent in a suitable organic solvent such as 1,2-dichloroethane.
- Add the ruthenium catalyst, specifically tris(2,2'-bipyridyl)ruthenium dichloride, to the reaction vessel under an inert atmosphere to prevent oxidation.
- Irradiate the mixture with blue LED light at room temperature for 10 to 15 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this photocatalytic methodology offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of hazardous reagents like sodium azide drastically simplifies the regulatory compliance landscape, reducing the administrative burden and insurance costs associated with handling explosive materials. Furthermore, the use of commercially available and inexpensive starting materials, such as substituted quinoxalinones and diazoacetates, ensures a stable and resilient supply chain that is less susceptible to market volatility compared to exotic custom building blocks. The mild reaction conditions also translate to lower energy consumption, as the process does not require cryogenic cooling or high-temperature heating, contributing to a reduced carbon footprint and lower utility costs per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction significantly reduces the number of unit operations required, thereby lowering labor costs and minimizing solvent usage throughout the production cycle. By avoiding the need for expensive transition metal ligands often required in palladium or copper-catalyzed cross-couplings, the raw material bill of materials is optimized, leading to direct cost savings. Additionally, the simplified post-reaction workup, which often involves straightforward column chromatography or crystallization, reduces the time and resources spent on purification, further enhancing the overall economic viability of the process for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, resulting in more consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous supply lines to downstream drug manufacturers, reducing the risk of production delays caused by failed batches or lengthy re-processing. The use of stable ruthenium catalysts and common organic solvents ensures that sourcing of critical inputs remains straightforward, mitigating the risk of supply disruptions that can occur with specialized or single-source reagents.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but advancements in flow chemistry and reactor design now make this technology highly scalable for industrial production. The absence of toxic azides and the use of catalytic amounts of ruthenium simplify waste treatment protocols, allowing for easier adherence to strict environmental regulations regarding heavy metal discharge and hazardous waste disposal. This environmental compatibility positions the technology as a future-proof solution for sustainable manufacturing, aligning with the increasing corporate social responsibility goals of major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed synthesis platform. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the practical aspects of adopting this technology. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: This method eliminates the need for sodium azide, a highly toxic and explosive reagent commonly used in traditional click chemistry approaches, thereby significantly enhancing operational safety and reducing hazardous waste disposal costs.
Q: What is the substrate scope for this ruthenium-catalyzed reaction?
A: The reaction demonstrates broad universality, successfully tolerating various substituents on the quinoxalinone ring including electron-withdrawing groups like fluorine, chlorine, bromine, and trifluoromethyl, as well as electron-donating methyl groups.
Q: Does this process require harsh reaction conditions or expensive equipment?
A: No, the process operates efficiently at room temperature under visible blue light irradiation, avoiding the need for high-pressure reactors or extreme thermal conditions, which simplifies the equipment requirements for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Triazole Quinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced photocatalytic technology in accelerating the development of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with specialized photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,3-triazole quinoxalinone derivative meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume needs. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
