Advanced Metal-Free Difluorobutoxylation for Scalable Pharmaceutical Intermediate Manufacturing
Introduction to Breakthrough Fluorination Technology
The landscape of fluorine chemistry is undergoing a significant transformation with the emergence of novel methodologies that prioritize sustainability and operational efficiency. A pivotal development in this domain is documented in patent CN114621137B, which discloses a groundbreaking method for the difluorobutoxylation of N-aryl-N-hydroxylamine compounds. This technology leverages the participation of tetrahydrofuran (THF) to achieve efficient functionalization without the need for transition metal catalysts. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this represents a critical advancement. The ability to introduce difluoroalkyl groups—a structural motif renowned for enhancing metabolic stability and membrane permeability in drug candidates—using mild, metal-free conditions addresses long-standing challenges in process chemistry. By utilizing bromodifluoromethyltrimethylsilane (TMSCF2Br) as a difluorocarbene precursor, this method circumvents the toxicity and cost associated with traditional reagents, positioning it as a highly attractive route for the synthesis of high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of difluoroalkyl groups into organic molecules has been fraught with significant technical and economic hurdles. Traditional difluoromethylation strategies often rely heavily on the use of toxic and hazardous reagents, such as chlorodifluoromethane (HCFC-22), which poses severe environmental and safety risks. Furthermore, many established protocols necessitate the use of expensive transition metal catalysts, particularly iridium complexes, to facilitate oxidative redox processes. These metal-catalyzed reactions not only inflate the raw material costs but also introduce complex downstream purification challenges, specifically the rigorous removal of trace heavy metals to meet stringent pharmaceutical regulatory standards. Additionally, conventional methods frequently demand strictly anhydrous and anaerobic conditions, requiring specialized equipment and inert gas manifolds that increase capital expenditure and operational complexity. The reliance on excessive amounts of strong bases further complicates waste stream management, making these processes less aligned with modern green chemistry principles and unsustainable for large-scale commercial manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in patent CN114621137B offers a paradigm shift towards greener and more economical synthesis. This novel approach utilizes tetrahydrofuran not merely as a solvent but as an active reactant that undergoes ring-opening to install the difluorobutoxy moiety. The reaction proceeds under remarkably mild conditions, typically between 0°C and 55°C, and critically, it does not require anhydrous or oxygen-free environments. This tolerance to ambient conditions drastically simplifies the operational workflow, allowing for the use of standard reactor setups without the need for costly inert atmosphere controls. The elimination of transition metal catalysts is a standout feature, removing the burden of metal scavenging steps and reducing the overall impurity profile of the final product. As illustrated in the general reaction scheme below, the transformation of N-aryl-N-hydroxylamine compounds (Formula I) into their difluorobutoxylated derivatives (Formula II) is achieved with high efficiency and broad substrate applicability.
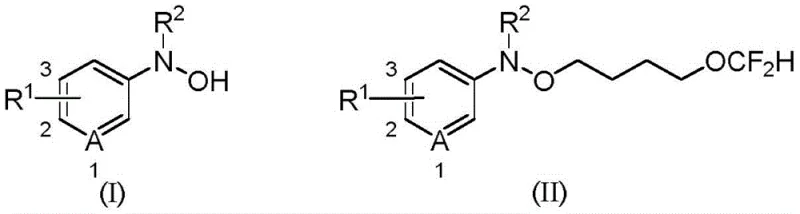
This streamlined process not only enhances safety by avoiding toxic gases but also significantly improves the atom economy and E-factor of the synthesis. The use of inorganic bases like potassium fluoride (KF) further underscores the cost-effectiveness and environmental compatibility of this route, making it an ideal candidate for the reliable production of pharmaceutical intermediates where purity and cost are paramount concerns.
Mechanistic Insights into TMSCF2Br-Mediated Ring Opening
To fully appreciate the robustness of this synthetic route, one must delve into the intricate mechanistic pathway that drives the difluorobutoxylation. The reaction is initiated by the activation of bromodifluoromethyltrimethylsilane (TMSCF2Br) by an inorganic base, such as potassium fluoride. This activation step generates the highly reactive difluorocarbene (:CF2) species in situ. Unlike free carbenes that might dimerize or insert indiscriminately, this difluorocarbene is immediately trapped by the oxygen atom of the tetrahydrofuran solvent. This interaction leads to the formation of a transient difluorooxonium ion intermediate, a key electrophilic species that dictates the regioselectivity of the subsequent transformation. The unique electronic properties of the fluorine atoms stabilize this intermediate just enough to allow for the next critical step without premature decomposition.
Following the formation of the oxonium ion, the N-aryl-N-hydroxylamine substrate, deprotonated by the base to form a nucleophilic anion, attacks the carbon atom adjacent to the oxygen in the tetrahydrofuran ring. This nucleophilic attack triggers the cleavage of the carbon-oxygen bond within the THF ring, resulting in a ring-opening event. This specific mechanistic sequence ensures that the difluoromethylene unit is seamlessly integrated into the butoxy chain attached to the nitrogen atom. The result is the formation of the target difluorobutoxylated N-aryl-N-hydroxylamine derivative with high fidelity. This mechanism explains the broad substrate tolerance observed in the patent examples, as the reaction relies on the nucleophilicity of the hydroxylamine rather than specific electronic properties of the aromatic ring, allowing for diverse substituents such as halogens, cyano groups, and esters to remain intact during the process.
How to Synthesize Difluorobutoxylated Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and mixing protocols to maximize yield and minimize side reactions. The patent outlines a straightforward procedure where the N-aryl-N-hydroxylamine starting material is combined with the inorganic base, water, and tetrahydrofuran prior to the addition of the difluorocarbene source. Maintaining the reaction temperature within the specified range of 0°C to 55°C is crucial for controlling the rate of carbene generation and ensuring selective ring opening. The preferred molar ratio of substrate to TMSCF2Br to base is approximately 1:3:4, which ensures complete conversion while suppressing potential decomposition pathways. Detailed standardized operating procedures for this transformation are provided below to assist technical teams in replicating these results.
- Mix the N-aryl-N-hydroxylamine substrate with inorganic base (preferably KF), water, and tetrahydrofuran in a reaction vessel.
- Add bromodifluoromethyltrimethylsilane (TMSCF2Br) to the mixture and stir at 0-55°C for 5-24 hours to allow ring-opening difluorobutoxylation.
- Perform post-treatment by concentrating the reaction mixture under reduced pressure and purifying via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend beyond simple chemical yield. The most significant advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependence on scarce and volatile precious metal catalysts like iridium, manufacturers can insulate their production lines from geopolitical supply shocks and price fluctuations associated with rare earth elements. Furthermore, the use of tetrahydrofuran as a dual-purpose solvent and reactant reduces the total volume of chemicals required, leading to substantial cost savings in material procurement and waste disposal. The operational simplicity of running reactions under ambient atmospheric conditions means that existing manufacturing infrastructure can be utilized without the need for expensive retrofits to handle inert gases or ultra-dry environments, thereby accelerating time-to-market for new intermediates.
- Cost Reduction in Manufacturing: The economic impact of removing transition metal catalysts cannot be overstated. Traditional metal-catalyzed fluorination often incurs high costs not just for the catalyst itself, but for the downstream processing required to remove metal residues to parts-per-million levels. This metal-free approach completely obviates the need for expensive scavenger resins or complex filtration steps, directly lowering the cost of goods sold (COGS). Additionally, the use of inexpensive inorganic bases like potassium fluoride instead of bulky organic bases or alkoxides further drives down raw material expenses. The high yields reported across various substrates indicate a robust process that minimizes batch failures and rework, contributing to overall financial efficiency in API manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are widely available from multiple global suppliers. Tetrahydrofuran and TMSCF2Br are established industrial chemicals with stable supply chains, unlike specialized ligands or custom-synthesized catalysts that may have single-source bottlenecks. The mild reaction conditions also reduce the risk of thermal runaways or hazardous incidents, ensuring consistent production schedules and reliable delivery timelines. This reliability is critical for maintaining continuous flow in the production of active pharmaceutical ingredients, where interruptions can have cascading effects on downstream formulation and distribution networks.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with increasingly stringent global regulations. The avoidance of toxic gases and heavy metals simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing site. The patent notes successful scale-up experiments, confirming that the kinetics and heat transfer characteristics of the reaction are manageable at larger volumes. This scalability ensures that the process can grow from kilogram-scale R&D batches to multi-ton commercial production without encountering unforeseen engineering hurdles, facilitating a smooth transition from development to full-scale manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this difluorobutoxylation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing synthetic pipelines for complex fluorinated molecules.
Q: What are the primary advantages of this difluorobutoxylation method over traditional approaches?
A: Unlike conventional methods that often require expensive transition metal catalysts like iridium or harsh anhydrous conditions, this patented process operates under mild, metal-free conditions using readily available tetrahydrofuran as both solvent and reactant.
Q: How does the reaction mechanism facilitate the introduction of the difluoro group?
A: The mechanism involves the in situ generation of difluorocarbene from TMSCF2Br, which reacts with tetrahydrofuran to form an oxonium ion intermediate that is subsequently attacked by the N-hydroxylamine nucleophile, resulting in ring opening and difluorobutoxylation.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method has been successfully scaled up in laboratory settings, demonstrating high yields and operational simplicity that translate well to commercial manufacturing environments without requiring specialized inert atmosphere equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Aryl-N-Hydroxylamine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the difluorobutoxylation technology described in patent CN114621137B for the next generation of pharmaceutical and agrochemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle fluorine chemistry with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to meet the exacting demands of global regulatory bodies. We are committed to delivering high-purity intermediates that empower your drug discovery and development programs.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific molecular targets. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's unique requirements. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and accelerate your path to market.
