Advanced Ligand-Assisted Zirconium Oxychloride Catalysis for Efficient Industrial Amide Production
The formation of the amide bond remains one of the most fundamental and critical transformations in the synthesis of active pharmaceutical ingredients (APIs) and complex fine chemicals. As detailed in patent CN103274958A, a groundbreaking methodology has been established utilizing ligand-assisted zirconium oxychloride catalysis to convert esters directly into amides. This technology represents a significant paradigm shift from traditional stoichiometric approaches, offering a robust pathway for the production of high-purity pharmaceutical intermediates. The core innovation lies in the utilization of zirconium oxychloride (ZrOCl₂·xH₂O) combined with specific organic ligands, creating a catalytic system that is not only highly efficient but also remarkably tolerant to moisture and air compared to conventional reagents. For R&D directors and process chemists, this offers a compelling alternative to harsh conditions, enabling the synthesis of complex molecular architectures with improved atom economy and reduced environmental impact.
Historically, the direct aminolysis of esters to form amides has been plagued by significant kinetic barriers, often necessitating the use of aggressive reagents or extreme conditions. Conventional methods frequently rely on strong alkali metal catalysts, such as sodium methoxide or lithium aluminum hydride, which require rigorous anhydrous environments and often result in the generation of stoichiometric amounts of alcohol byproducts that complicate downstream purification. Furthermore, the use of pyrophoric reagents like butyllithium or Grignard reagents introduces severe safety hazards and limits the scope of substrates due to poor functional group tolerance. These legacy processes often demand cryogenic temperatures and extended reaction times, leading to inflated operational costs and reduced throughput in a manufacturing setting. The inherent instability of many traditional metal amide reagents further exacerbates supply chain vulnerabilities, as they require specialized storage and handling protocols that are difficult to maintain on a multi-ton scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on traditional metal amides, such as tin, aluminum, titanium, or lithium amides, presents a multifaceted challenge for modern chemical manufacturing. While these reagents can facilitate amidation at lower temperatures, they are prohibitively expensive and demand absolute exclusion of moisture, necessitating the use of inert atmosphere gloveboxes or specialized reactor setups that increase capital expenditure. The sensitivity of these reagents means that even trace amounts of water can quench the reaction, leading to inconsistent yields and batch-to-batch variability that is unacceptable in regulated pharmaceutical production. Additionally, the generation of metal-containing waste streams poses significant environmental disposal challenges, requiring complex treatment protocols to meet increasingly stringent regulatory standards. The economic burden of disposing of heavy metal residues, combined with the high cost of the reagents themselves, creates a substantial barrier to cost-effective production, particularly for commodity-grade intermediates where margin compression is a constant pressure.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent leverages the unique Lewis acidic properties of zirconium oxychloride, stabilized by auxiliary ligands, to activate the ester carbonyl towards nucleophilic attack by amines. This catalytic system operates effectively under reflux conditions in common organic solvents like toluene or n-heptane, eliminating the need for cryogenic cooling or absolute anhydrous conditions. The addition of ligands, ranging from monodentate oxygen donors to bidentate nitrogen-oxygen chelators like 8-hydroxyquinoline, fine-tunes the electronic environment of the zirconium center, enhancing its catalytic activity and selectivity. This method allows for the direct conversion of a wide variety of esters, including those with aryl, arylmethyl, and long-chain alkyl substituents, into their corresponding amides with high efficiency. By avoiding the use of stoichiometric strong bases, the process minimizes side reactions and simplifies the workup procedure to a straightforward solvent removal and recrystallization step.
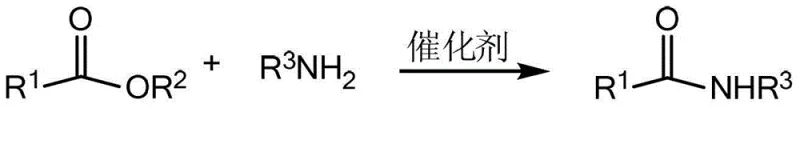
This transformative reaction pathway is visually represented by the general scheme where diverse ester substrates react with various amines to yield the target amide products. The versatility of this system is underscored by its ability to accommodate a broad spectrum of R groups, including aromatic rings, heterocycles like pyridine and thiophene, and aliphatic chains up to C18. This structural flexibility is crucial for medicinal chemists who require access to diverse chemical space for structure-activity relationship (SAR) studies. The use of zirconium, a relatively abundant and non-toxic transition metal, aligns perfectly with the principles of green chemistry, reducing the reliance on precious or toxic metals like palladium or tin. Consequently, this methodology not only improves the technical feasibility of amide synthesis but also enhances the sustainability profile of the manufacturing process, a key consideration for modern supply chain managers focused on environmental, social, and governance (ESG) goals.
Mechanistic Insights into Ligand-Assisted Zirconium Catalysis
The mechanistic underpinning of this reaction involves the coordination of the zirconium center to the carbonyl oxygen of the ester substrate, thereby increasing the electrophilicity of the carbonyl carbon. This Lewis acid activation lowers the energy barrier for the nucleophilic attack by the amine, facilitating the formation of the tetrahedral intermediate. The presence of the ligand plays a dual role: it stabilizes the zirconium species against hydrolysis and aggregation, maintaining the catalyst in an active monomeric or oligomeric form, and it modulates the steric environment around the metal center to favor the approach of the amine nucleophile. Unlike traditional Friedel-Crafts catalysts which might promote unwanted side reactions, the zirconium-ligand complex exhibits high chemoselectivity, preserving sensitive functional groups elsewhere in the molecule. This precise control over the reaction trajectory is essential for synthesizing complex pharmaceutical intermediates where the integrity of the molecular scaffold must be maintained throughout the synthetic sequence.
Furthermore, the impurity profile generated by this catalytic method is significantly cleaner compared to base-mediated processes. Since the reaction does not involve strong bases, there is minimal risk of epimerization at chiral centers alpha to the carbonyl, a common pitfall in peptide synthesis and chiral drug manufacturing. The byproduct of the reaction is the corresponding alcohol derived from the ester leaving group, which is typically volatile or easily separated during the solvent removal phase. This simplifies the purification process, often allowing for high-purity isolation via simple recrystallization rather than demanding column chromatography. For quality control teams, this translates to a more robust and reproducible process with a well-defined impurity spectrum, facilitating faster regulatory approval and release testing. The ability to run the reaction at reflux temperatures (typically 80°C to 110°C depending on the solvent) ensures sufficient thermal energy to drive the reaction to completion without degrading thermally labile products.
How to Synthesize Amide Compounds Efficiently
To implement this advanced catalytic protocol in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize yield and purity while ensuring safety. The process begins with the precise weighing of the ester and amine starting materials, which are then dissolved in a selected organic solvent such as n-heptane or toluene. The catalytic system, consisting of zirconium oxychloride octahydrate and the chosen ligand, is added to the reaction mixture, initiating the catalytic cycle upon heating. Detailed standardized operating procedures regarding mixing rates, temperature ramping, and monitoring techniques are critical for consistent results. The following guide outlines the essential steps for executing this synthesis effectively, ensuring that the full potential of this ligand-assisted zirconium catalysis is realized in practical applications.
- Mix the ester substrate and amine reactant in a suitable organic solvent such as toluene or n-heptane within a reaction vessel.
- Add the catalytic system comprising zirconium oxychloride (ZrOCl2·xH2O) and a specific ligand (e.g., 8-hydroxyquinoline or citric acid) to the mixture.
- Heat the reaction mixture to reflux for 2 to 24 hours, then remove the solvent under reduced pressure and purify the crude product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this zirconium-catalyzed amidation technology offers profound advantages for procurement managers and supply chain leaders seeking to optimize cost structures and mitigate risk. The primary driver of cost reduction is the replacement of expensive, moisture-sensitive metal amides with commodity-grade zirconium oxychloride, which is available in bulk quantities at a fraction of the cost. This shift eliminates the need for specialized storage infrastructure, such as dry rooms or inert gas blanketing systems, thereby reducing facility overheads. Moreover, the simplified workup procedure, which avoids aqueous quenches and complex extractions, reduces solvent consumption and waste disposal fees, contributing to a leaner and more sustainable manufacturing operation. The overall process intensity is lowered, allowing for higher throughput in existing reactor fleets without the need for significant capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic reduction in raw material costs associated with the catalyst system. By utilizing zirconium oxychloride, a widely available industrial chemical, manufacturers can avoid the premium pricing of specialized organometallic reagents. Additionally, the elimination of cryogenic cooling requirements significantly lowers energy consumption, as reactions can be run at ambient pressure and reflux temperatures. The high atom economy of the direct ester-to-amide conversion minimizes raw material waste, ensuring that a greater proportion of input costs are converted into valuable product. This efficiency gain is compounded by the reduced need for extensive purification steps, which often account for a significant portion of the total manufacturing cost in fine chemical production.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the use of stable, shelf-stable reagents that do not degrade upon exposure to atmospheric moisture. Unlike pyrophoric reagents that require just-in-time delivery and specialized logistics, zirconium salts can be stocked in standard warehouses, buffering the production line against supply disruptions. The broad substrate scope of the reaction allows for the sourcing of diverse ester and amine building blocks from multiple global suppliers, reducing dependency on single-source vendors. This flexibility is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream customers. The robustness of the catalytic system also ensures consistent batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is straightforward due to the use of standard unit operations like reflux and distillation. The absence of hazardous reagents simplifies safety assessments and regulatory filings, accelerating the timeline for technology transfer to manufacturing sites. From an environmental standpoint, the process generates minimal hazardous waste, as the zirconium catalyst can potentially be recovered or disposed of with less stringent requirements than heavy metal alternatives. The use of common organic solvents facilitates recycling and recovery, further reducing the environmental footprint. This alignment with green chemistry principles enhances the corporate sustainability profile, a factor that is increasingly important for securing contracts with major pharmaceutical companies committed to responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zirconium-catalyzed amidation technology. These insights are derived directly from the patent data and practical experience in scaling similar catalytic processes. Understanding these nuances is vital for stakeholders evaluating the feasibility of adopting this method for their specific product portfolios. The answers provided reflect the balance between technical performance and operational practicality that defines modern chemical manufacturing.
Q: What are the advantages of using Zirconium Oxychloride over traditional metal amides?
A: Unlike traditional metal amides which require absolute anhydrous conditions and are expensive, zirconium oxychloride is moisture-stable, low-cost, and operates effectively under standard reflux conditions, significantly simplifying the operational requirements.
Q: Does this catalytic method tolerate sensitive functional groups?
A: Yes, the mild Lewis acidic nature of the zirconium-ligand complex allows for the conversion of esters to amides without affecting other sensitive functional groups that might degrade under strong basic or pyrophoric conditions.
Q: Is this process suitable for large-scale manufacturing?
A: Absolutely. The process utilizes common organic solvents, avoids cryogenic temperatures, and employs a simple workup procedure involving solvent removal and recrystallization, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of ligand-assisted zirconium catalysis in reshaping the landscape of amide synthesis. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative patent technology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this efficient catalytic method are fully realized at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of amide intermediate meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality and consistency makes us the preferred partner for companies seeking to secure their supply of critical building blocks.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific manufacturing needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating the tangible economic benefits of switching to this zirconium-catalyzed process. We encourage you to contact us to request specific COA data and route feasibility assessments for your projects. By leveraging our deep understanding of catalytic amidation and our robust supply chain capabilities, we can help you reduce lead time for high-purity pharmaceutical intermediates and achieve your strategic sourcing goals efficiently.
