Advanced Synthesis of 4-Ethyl-5-Aminopyrimidine: A Strategic Breakthrough for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Synthesis of 4-Ethyl-5-Aminopyrimidine: A Strategic Breakthrough for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes for heterocyclic building blocks, particularly pyrimidine derivatives which serve as critical scaffolds for antiviral and antitumor agents. Patent CN113185465A introduces a transformative preparation method for 4-ethyl-5-aminopyrimidine, addressing long-standing challenges in regioselectivity and purification efficiency. This innovation leverages a sophisticated two-step sequence involving a palladium-catalyzed Stille coupling followed by a mild catalytic hydrogenation, effectively bypassing the limitations of traditional alkylation methods. By shifting from direct alkyl halide substitution to a vinyl-coupling strategy, the process ensures high structural fidelity and minimizes the formation of unwanted isomers that typically plague pyrimidine functionalization. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more predictable and cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of alkyl groups onto the pyrimidine ring has been fraught with significant chemical hurdles, primarily stemming from the electronic nature of the heterocycle. Conventional alkylation strategies often require harsh reaction conditions and multi-step functional group conversions that introduce substantial risk and complexity into the manufacturing workflow. A major pain point in these traditional routes is the tendency to generate structural isomers during the synthesis reaction, which creates a nightmare for downstream purification teams. The presence of these isomers not only drastically lowers the overall yield of the desired target molecule but also necessitates expensive and time-consuming chromatographic separations to meet stringent purity specifications required for API production. Furthermore, many existing methods involve dangerous reagents or extreme temperatures that complicate safety protocols and increase the barrier to safe commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a strategic disconnection that transforms the problem from a difficult alkylation into a highly selective cross-coupling reaction. By employing tributylvinyltin as a coupling partner with 4-bromo-5-aminopyrimidine, the synthesis achieves precise control over the substitution pattern, ensuring that the carbon chain is installed exclusively at the 4-position. This methodology eliminates the formation of isomeric byproducts at the source, thereby simplifying the purification landscape and significantly enhancing the overall process efficiency. The subsequent conversion of the vinyl group to an ethyl group via catalytic hydrogenation is performed under exceptionally mild conditions, utilizing standard palladium on carbon catalysts at room temperature and atmospheric pressure. This gentle approach preserves the integrity of the sensitive aminopyrimidine core while delivering the target ethyl derivative with remarkable consistency and high yield.
Mechanistic Insights into Pd-Catalyzed Stille Coupling and Hydrogenation
The core of this synthetic innovation lies in the mechanistic elegance of the palladium-catalyzed Stille coupling, which facilitates the formation of the carbon-carbon bond between the electron-deficient pyrimidine ring and the vinyl stannane. The reaction initiates with the oxidative addition of the palladium(0) species, generated in situ from bis(triphenylphosphine)palladium dichloride, into the carbon-bromine bond of the 4-bromo-5-aminopyrimidine substrate. This is followed by a transmetallation step where the vinyl group is transferred from the tin atom to the palladium center, a process that is highly tolerant of the amino functionality present on the ring. The catalytic cycle concludes with reductive elimination, releasing the 4-vinyl-5-aminopyrimidine product and regenerating the active palladium catalyst. This mechanism is superior because it operates efficiently in polar aprotic solvents like acetonitrile at moderate temperatures of 70-80°C, avoiding the thermal degradation often seen in more aggressive alkylation protocols.
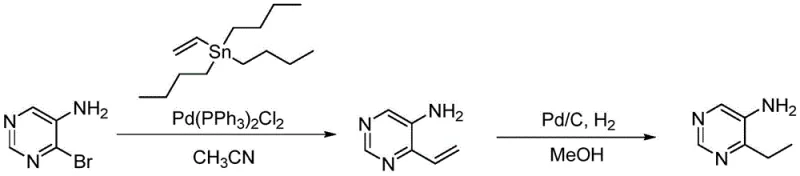
Following the coupling event, the purification and hydrogenation stages are meticulously designed to maximize impurity control and product recovery. The crude vinyl intermediate is purified using silica gel column chromatography with a specific dichloromethane and methanol eluent system, which effectively removes tin byproducts and unreacted starting materials before the final reduction step. The hydrogenation mechanism involves the adsorption of the vinyl double bond onto the surface of the palladium-carbon catalyst, where it reacts with molecular hydrogen to form the saturated ethyl chain. Crucially, the workup procedure employs a pH-swing extraction strategy, adjusting the aqueous phase to acidic conditions to remove non-basic impurities, followed by basification to extract the product into an organic solvent. This rigorous control over the chemical environment ensures that the final 4-ethyl-5-aminopyrimidine meets high-purity standards essential for downstream drug development applications.
How to Synthesize 4-Ethyl-5-Aminopyrimidine Efficiently
The execution of this synthesis requires strict adherence to inert atmosphere techniques and precise stoichiometric control to replicate the high yields reported in the patent literature. Operators must ensure that the acetonitrile solvent is thoroughly dried and dehydrated prior to use, as moisture can inhibit the palladium catalyst and reduce the efficiency of the transmetallation step. The reaction mixture should be subjected to multiple vacuum-nitrogen cycles to eliminate oxygen, which could otherwise oxidize the phosphine ligands or the tin reagent. Detailed standardized operating procedures regarding the addition rates of the catalyst and the temperature ramping profiles are critical for maintaining batch-to-batch consistency. For a comprehensive guide on the specific molar ratios, solvent volumes, and workup parameters required to achieve optimal results, please refer to the technical protocol outlined below.
- Perform Stille coupling between 4-bromo-5-aminopyrimidine and tributylvinyltin using Pd(PPh3)2Cl2 catalyst in dry acetonitrile at 70-80°C to form 4-vinyl-5-aminopyrimidine.
- Purify the crude vinyl intermediate using silica gel column chromatography with a dichloromethane/methanol eluent system to achieve high purity before hydrogenation.
- Conduct catalytic hydrogenation of the vinyl intermediate using 10% Pd/C in methanol or ethanol at room temperature and normal pressure, followed by pH-controlled extraction to isolate the final ethyl product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented methodology offers profound advantages by fundamentally simplifying the manufacturing topology and reducing reliance on exotic reagents. The shift to a Stille coupling strategy allows manufacturers to utilize widely available commodity chemicals like tributylvinyltin and standard palladium catalysts, thereby mitigating the risk of raw material shortages that often plague specialized alkylating agents. The operational simplicity of the process, characterized by ambient pressure hydrogenation and moderate heating requirements, means that production can be executed in standard glass-lined or stainless steel reactors without the need for costly high-pressure autoclaves. This compatibility with existing infrastructure significantly lowers the barrier to entry for contract manufacturing organizations and accelerates the timeline from pilot plant to full-scale commercial production.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the elimination of complex separation processes associated with isomeric mixtures. By achieving high regioselectivity, the process drastically reduces the volume of solvents and silica gel required for purification, leading to substantial savings in waste disposal and raw material costs. Furthermore, the high yields observed in both the coupling and hydrogenation steps minimize the loss of valuable intermediates, ensuring that the overall cost of goods sold remains competitive even at large production scales. The ability to recover and recycle solvents like acetonitrile and tetrahydrofuran further enhances the financial viability of the process for long-term supply contracts.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to improved supply continuity and reduced lead times for high-purity pharmaceutical intermediates. Since the reaction does not rely on cryogenic temperatures or ultra-high pressures, the risk of batch failure due to equipment malfunction or thermal runaway is significantly minimized. The use of stable reagents that do not require special storage conditions beyond standard inert atmosphere handling simplifies logistics and inventory management. This reliability allows procurement managers to forecast demand with greater confidence and secure consistent supply streams for critical drug development programs without fearing unexpected production delays.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is favorable due to the avoidance of hazardous alkylating agents and the generation of minimal toxic byproducts. The mild nature of the hydrogenation step reduces energy consumption compared to high-temperature alternatives, aligning with modern green chemistry principles and corporate sustainability goals. Scalability is further supported by the straightforward workup procedure, which relies on liquid-liquid extraction and crystallization rather than preparative HPLC, making it ideally suited for multi-kilogram and ton-scale operations. This combination of safety, efficiency, and environmental stewardship makes the process highly attractive for regulatory filings and long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route for industrial applications. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy and relevance for process chemists. Understanding these nuances is essential for successfully transferring this technology from the laboratory bench to the manufacturing floor while maintaining product quality.
Q: Why is the Stille coupling route preferred over direct alkylation for synthesizing 4-ethyl-5-aminopyrimidine?
A: Direct alkylation of pyrimidine rings often suffers from poor regioselectivity, leading to difficult-to-separate isomers and low yields. The Stille coupling route described in patent CN113185465A utilizes a vinyl tin reagent which couples selectively at the 4-position, avoiding isomer formation entirely and ensuring a single product configuration that is easy to purify.
Q: What are the expected yield and purity profiles for this synthesis method?
A: According to the experimental data provided in the patent, the initial Stille coupling step achieves a yield of approximately 93% with 95% purity. The subsequent hydrogenation step yields the final 4-ethyl-5-aminopyrimidine with yields ranging from 72% to 89% and purity levels consistently above 96%, demonstrating a robust and efficient process suitable for industrial application.
Q: Is this synthesis method scalable for commercial production of pharmaceutical intermediates?
A: Yes, the method is highly scalable due to its mild reaction conditions. The coupling occurs at moderate temperatures (70-80°C) and the hydrogenation proceeds at room temperature and normal pressure, eliminating the need for specialized high-pressure reactors. This significantly reduces capital expenditure and operational risks, facilitating easy realization of industrialization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Ethyl-5-Aminopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for advanced pharmaceutical intermediates like 4-ethyl-5-aminopyrimidine. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and timeliness. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality assurance means that we can deliver materials that are ready for immediate use in sensitive downstream reactions, minimizing your internal testing burden and accelerating your drug development timelines.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific application requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our optimized processes can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in pyrimidine chemistry can become a strategic asset for your organization.
