Advanced Catalytic Route for High-Purity Pyrano[2,3-d]pyrimidone Derivatives Manufacturing
Advanced Catalytic Route for High-Purity Pyrano[2,3-d]pyrimidone Derivatives Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for heterocyclic compounds that serve as critical scaffolds in drug discovery, particularly for agents exhibiting antibacterial, anticancer, and anti-inflammatory properties. Patent CN110028518B introduces a groundbreaking methodology for the preparation of pyrano[2,3-d]pyrimidone derivatives, utilizing a novel catalytic system that addresses long-standing inefficiencies in traditional organic synthesis. This technology leverages a sulfonated molecular sieve catalyst within an ethanol-dimethylformamide mixed solvent system, facilitated by ultrasonic irradiation to achieve rapid conversion rates. By integrating a one-pot four-component reaction strategy, this approach not only streamlines the operational workflow but also significantly enhances the atom economy of the process. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates. The ability to recycle the catalyst system without complex regeneration steps offers a compelling value proposition for sustainable chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrano[2,3-d]pyrimidone derivatives has relied heavily on homogeneous acid catalysts such as concentrated sulfuric acid or acidic ionic liquids, both of which present significant logistical and environmental challenges. The use of concentrated sulfuric acid, while effective in driving the reaction, generates substantial quantities of hazardous waste acid that necessitates expensive neutralization and disposal protocols, thereby inflating the overall production costs. Furthermore, homogeneous catalysts are notoriously difficult to separate from the reaction mixture, often requiring extensive washing and purification steps that lead to product loss and increased solvent consumption. In the case of acidic ionic liquids, although they offer some recyclability, their high synthesis cost and potential toxicity pose barriers to widespread industrial adoption, especially when scaling up to multi-ton production levels. These conventional routes often suffer from poor atom economy and lengthy process flows, as they may require the pre-synthesis of intermediate pyran compounds before the final cyclization step can occur. Consequently, the cumulative effect of these inefficiencies results in higher carbon footprints and reduced competitiveness in the global supply chain for fine chemical intermediates.
The Novel Approach
In stark contrast, the method disclosed in patent CN110028518B employs a heterogeneous sulfonated molecular sieve catalyst that fundamentally alters the economic and environmental landscape of this synthesis. This innovative approach utilizes a one-pot condensation of aromatic aldehydes, malononitrile, 6-methyl-4-hydroxy-2-pyrone, and acetic anhydride, eliminating the need for isolated intermediate steps and drastically shortening the process timeline. The solid nature of the sulfonated molecular sieve allows for straightforward separation via simple hot filtration, enabling the catalyst to be recovered and reused directly in subsequent batches without any pretreatment. This recyclability is a game-changer for supply chain stability, as it mitigates the volatility associated with purchasing fresh catalysts for every production run. Moreover, the integration of ultrasonic assistance accelerates the reaction kinetics, ensuring high yields within minutes rather than hours, which translates to increased reactor throughput and energy savings. ![Reaction scheme showing the one-pot four-component synthesis of pyrano[2,3-d]pyrimidone derivatives catalyzed by sulfonated molecular sieve](/insights/img/pyrano-pyrimidone-synthesis-catalyst-pharma-supplier-20260304172735-01.webp)
Mechanistic Insights into Sulfonated Molecular Sieve Catalysis
The efficacy of this synthetic route is rooted in the unique structural properties of the sulfonated molecular sieve, specifically the MCM-41 type functionalized with propyl sulfonic acid groups. These sulfonic acid moieties act as strong Brønsted acid sites anchored on a high-surface-area mesoporous silica framework, providing ample active centers for the activation of carbonyl groups in the aromatic aldehyde and acetic anhydride. The reaction mechanism likely proceeds through a Knoevenagel condensation between the aldehyde and malononitrile, followed by a Michael addition of the 6-methyl-4-hydroxy-2-pyrone, and finally a cyclization facilitated by the acidic environment. The mesoporous structure of the catalyst ensures that reactants can diffuse freely to the active sites, minimizing steric hindrance and promoting uniform reaction progress throughout the mixture. 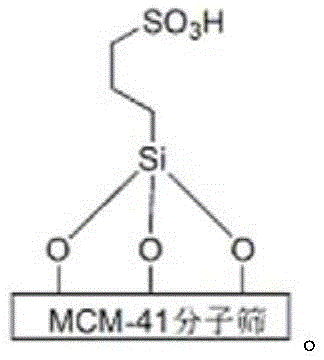
From an impurity control perspective, the mild yet efficient nature of this solid acid catalysis prevents the degradation of sensitive functional groups that often occurs under the harsh conditions of liquid mineral acids. The precise control over acidity provided by the grafted sulfonic groups reduces the incidence of polymerization side reactions, which are common pitfalls in the synthesis of complex heterocycles. Additionally, the use of an ethanol-DMF mixed solvent system optimizes the solubility of diverse aromatic aldehydes, ensuring a homogeneous reaction phase that further suppresses the formation of by-products. For quality assurance teams, this means the resulting crude product possesses a cleaner impurity profile, reducing the burden on downstream purification processes such as recrystallization or column chromatography. The stability of the catalyst over multiple cycles, as evidenced by consistent yield maintenance, indicates a robust catalytic cycle that resists leaching or deactivation, ensuring batch-to-batch consistency crucial for GMP manufacturing environments.
How to Synthesize Pyrano[2,3-d]pyrimidone Derivatives Efficiently
The operational simplicity of this patented method makes it highly attractive for process chemists looking to implement scalable solutions without extensive equipment modifications. The procedure involves dissolving the stoichiometric amounts of aromatic aldehyde, malononitrile, and 6-methyl-4-hydroxy-2-pyrone in the solvent mixture, followed by the addition of the catalyst and subjecting the mixture to ultrasonic radiation. After a brief induction period, acetic anhydride is introduced, and the reaction is driven to completion under reflux with continued ultrasonic assistance, typically requiring less than twenty minutes. The detailed standardized synthesis steps, including specific molar ratios and temperature controls optimized for maximum yield, are outlined below for technical reference.
- Dissolve aromatic aldehyde, malononitrile, and 6-methyl-4-hydroxy-2-pyrone in an ethanol-DMF mixed solvent with the sulfonated molecular sieve catalyst.
- Apply ultrasonic radiation for initial mixing, then add acetic anhydride and reflux under continued ultrasonic assistance for 13-21 minutes.
- Perform hot suction filtration to recover the catalyst, cool the filtrate to precipitate the product, and dry the residue to obtain the pure derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic technology offers profound strategic benefits that extend beyond mere chemical efficiency. The elimination of corrosive liquid acids and the implementation of a recyclable solid catalyst system directly address the growing regulatory pressures regarding waste disposal and environmental compliance. By adopting this method, organizations can significantly reduce their dependency on volatile raw material markets for consumable catalysts, thereby stabilizing long-term production costs. The streamlined workflow also minimizes the manpower and utility resources required for reaction monitoring and workup, contributing to a leaner operational model. Furthermore, the high purity of the crude product reduces the load on quality control laboratories and shortens the release time for finished goods, enhancing overall supply chain agility.
- Cost Reduction in Manufacturing: The implementation of a reusable heterogeneous catalyst eliminates the recurring expense of purchasing stoichiometric or excess amounts of homogeneous acids for every batch, leading to substantial direct material cost savings. Additionally, the simplified workup procedure, which relies on filtration rather than complex extraction and neutralization, drastically reduces solvent consumption and waste treatment fees. The energy efficiency gained from shorter reaction times driven by ultrasonic assistance further lowers the utility overhead associated with prolonged heating and stirring. These cumulative factors result in a significantly lower cost of goods sold (COGS), allowing for more competitive pricing in the global pharmaceutical intermediate market.
- Enhanced Supply Chain Reliability: The robustness of the sulfonated molecular sieve catalyst ensures consistent performance over multiple cycles, reducing the risk of production delays caused by catalyst failure or variability. Since the catalyst can be recovered and reused directly without regeneration, the supply chain is insulated from potential disruptions in the availability of specialized catalytic reagents. The use of common, commercially available starting materials such as aromatic aldehydes and malononitrile further secures the supply line against raw material shortages. This reliability is critical for maintaining continuous manufacturing schedules and meeting the stringent delivery commitments required by downstream API manufacturers.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction system facilitates easy scale-up from laboratory to pilot and commercial scales without the heat transfer and mixing limitations often encountered with viscous ionic liquids. The absence of heavy metal contaminants or corrosive liquid acids simplifies the environmental permitting process and aligns with green chemistry principles, enhancing the corporate sustainability profile. Waste generation is minimized through catalyst recycling and reduced solvent usage, ensuring compliance with increasingly strict environmental regulations across different jurisdictions. This scalability ensures that the technology can support the transition from clinical trial material supply to full-scale commercial production seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic synthesis route, derived directly from the patent specifications and experimental data. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines. The answers provided reflect the specific advantages of the sulfonated molecular sieve system over traditional methodologies.
Q: What are the primary advantages of using sulfonated molecular sieves over concentrated sulfuric acid?
A: Sulfonated molecular sieves act as heterogeneous solid acid catalysts that can be easily separated via filtration and recycled multiple times without significant loss of activity, whereas concentrated sulfuric acid generates large volumes of corrosive waste acid that requires complex neutralization and disposal procedures.
Q: How does ultrasonic assistance impact the reaction efficiency?
A: Ultrasonic irradiation enhances mass transfer and mixing at the molecular level, significantly reducing reaction times from hours to minutes and improving overall yields by preventing the formation of side products associated with prolonged heating.
Q: Is the catalyst system suitable for industrial scale-up?
A: Yes, the robustness of the sulfonated molecular sieve combined with the simple filtration recovery process makes this method highly scalable, addressing key supply chain concerns regarding catalyst cost and waste management in large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrano[2,3-d]pyrimidone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the pharmaceutical intermediate sector. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is executed with precision and safety. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging the catalytic efficiencies described in patent CN110028518B, we can offer our partners a supply solution that balances cost-effectiveness with uncompromising quality standards.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this green catalytic process. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain planning initiatives.
