Revolutionizing Quinoxalinedione Manufacturing: A Green Aqueous Oxidation Strategy for Global Supply Chains
Introduction to Patent CN110642798B
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign methodologies for constructing privileged heterocyclic scaffolds. Patent CN110642798B introduces a groundbreaking green synthesis protocol for N-substituted-1,4-dihydro-2,3-quinoxalinedione compounds, a core pharmacophore prevalent in anticancer and antibacterial drug discovery. This technology represents a paradigm shift from traditional harsh synthetic routes to a mild, aqueous-based oxidative strategy. By leveraging ammonium persulfate as a dual-function oxidant and oxygen donor within a specialized micellar system, the process achieves exceptional conversion rates while adhering to strict environmental compliance standards. For R&D directors and procurement specialists, this patent offers a viable pathway to reduce reliance on toxic chlorinated solvents and expensive coupling reagents, thereby optimizing the overall cost structure of API intermediate manufacturing.
The significance of this innovation lies in its ability to operate under air atmosphere at moderate temperatures, utilizing water as the bulk solvent. This approach not only mitigates the safety risks associated with volatile organic compounds but also simplifies the post-reaction workup procedures significantly. The utilization of DL-α-tocopheryl methoxy polyethylene glycol succinic acid (TPGS-750-M) creates a unique nano-reactor environment that enhances the solubility of organic substrates in water, facilitating efficient mass transfer and reaction kinetics. As global regulatory bodies tighten restrictions on solvent emissions and heavy metal residues, adopting such metal-free, water-based technologies becomes a strategic imperative for maintaining supply chain continuity and market access in regulated pharmaceutical markets.
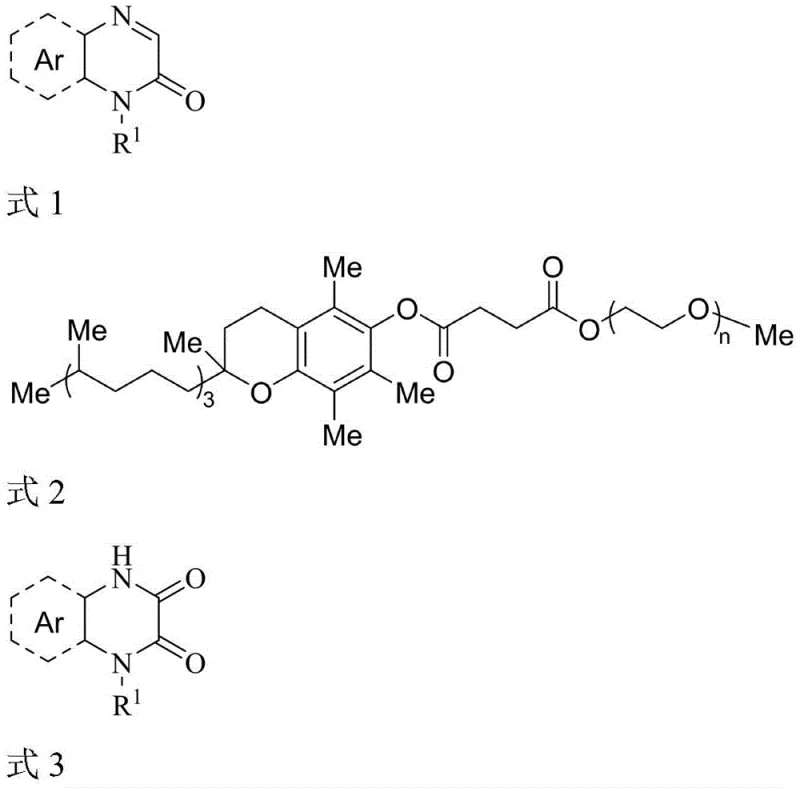
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted-1,4-dihydro-2,3-quinoxalinedione derivatives has relied heavily on the cycloaddition reaction between N-substituted o-phenylenediamines and oxalyl chloride or its derivatives. While chemically feasible, these conventional pathways are fraught with significant operational and economic drawbacks that hinder their scalability. The requirement for toxic chlorinated solvents such as o-dichlorobenzene or dichloromethane poses severe environmental hazards and necessitates costly waste treatment infrastructure. Furthermore, these reactions often demand harsh conditions, including high temperatures or the use of excessive strong bases as promoters, which can lead to side reactions and complicate the impurity profile of the final product.
From a supply chain perspective, the reliance on oxalyl chloride introduces volatility, as it is a hazardous reagent requiring specialized handling and storage protocols. The post-reaction processing is equally burdensome, typically involving tedious acid-base neutralization steps to remove residual reagents and byproducts. This multi-step workup not only extends the production cycle time but also results in substantial material loss and increased consumption of auxiliary chemicals. For procurement managers, these inefficiencies translate into higher raw material costs and unpredictable lead times, making the traditional route less attractive for large-scale commercial production where margin compression is a constant concern.
The Novel Approach
In stark contrast, the methodology disclosed in CN110642798B utilizes a direct oxidative hydroxylation strategy that bypasses the need for dangerous acylating agents entirely. By employing ammonium persulfate in conjunction with the surfactant TPGS-750-M, the reaction proceeds smoothly in an aqueous medium under mild thermal conditions. This one-pot transformation converts N-substituted quinoxalin-2(1H)-one derivatives directly into the desired dione structures with high selectivity. The elimination of chlorinated solvents and strong bases drastically reduces the environmental footprint of the process, aligning perfectly with modern green chemistry principles and corporate sustainability goals.
The operational simplicity of this novel approach is a major advantage for industrial scale-up. The reaction can be conducted under an air atmosphere, removing the need for inert gas purging systems and reducing equipment complexity. Moreover, the aqueous micellar system allows for the recycling of the reaction medium; the patent data indicates that the TPGS-750-M solution can be reused for at least five cycles without compromising yield. This recyclability feature offers profound economic benefits by lowering the effective cost of the solvent system per kilogram of product produced. For manufacturing teams, this translates to a streamlined workflow with fewer unit operations, reduced energy consumption for solvent recovery, and a safer working environment free from noxious fumes.
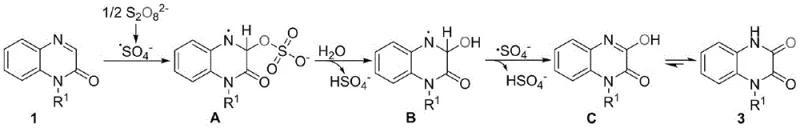
Mechanistic Insights into Ammonium Persulfate Mediated Oxidation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for diverse substrate scopes. The reaction is initiated by the thermal decomposition of ammonium persulfate, which generates sulfate anion radicals in situ. These highly reactive radical species attack the electron-rich double bond of the N-substituted quinoxalin-2(1H)-one substrate, forming a carbon-centered radical intermediate. This initial addition step is facilitated by the unique microenvironment provided by the TPGS-750-M micelles, which concentrate both the hydrophobic organic substrate and the hydrophilic oxidant at the interface, effectively overcoming the solubility barriers inherent in aqueous systems.
Following the radical addition, the intermediate undergoes a series of rapid transformations involving water molecules and further radical interactions. The sulfate radical intermediate reacts with water to introduce the hydroxyl functionality, followed by the elimination of bisulfate anions. This sequence ultimately leads to the formation of an unstable hydroxylated intermediate which spontaneously tautomerizes to yield the thermodynamically stable N-substituted-1,4-dihydro-2,3-quinoxalinedione product. The absence of transition metal catalysts is particularly noteworthy, as it eliminates the risk of heavy metal contamination in the final API, a critical quality attribute for pharmaceutical registration. This metal-free radical pathway ensures a clean impurity profile, simplifying the analytical validation and regulatory filing processes for downstream drug candidates.
How to Synthesize N-Substituted-1,4-Dihydro-2,3-Quinoxalinedione Efficiently
The practical implementation of this green synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the micellar catalyst solution, followed by the sequential addition of the substrate and the oxidant. The reaction is then heated to the optimal temperature range identified in the patent studies. Detailed standard operating procedures regarding stoichiometry, mixing rates, and specific workup parameters are essential for ensuring consistent batch-to-batch reproducibility. For a comprehensive guide on the exact molar ratios and isolation techniques validated in the patent examples, please refer to the standardized synthesis protocol outlined below.
- Prepare the reaction mixture by combining the N-substituted quinoxalin-2(1H)-one derivative with ammonium persulfate (2.5 equivalents) in a 2wt% aqueous solution of TPGS-750-M.
- Heat the mixture to 60°C under an air atmosphere and stir for approximately 12 hours to allow the radical oxidation and hydroxylation to proceed.
- Upon completion, extract the product using ethyl acetate, dry the organic layer with anhydrous sodium sulfate, and concentrate to obtain the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic advantages beyond mere technical feasibility. The shift from hazardous organic solvents to water-based media fundamentally alters the cost dynamics of production. By eliminating the need for expensive, regulated solvents like dichloromethane and the associated costs of solvent recovery or incineration, the overall variable cost of goods sold is significantly reduced. Furthermore, the use of commodity-grade ammonium persulfate as the oxidant ensures a stable and low-cost supply of key reagents, insulating the manufacturing process from the price volatility often seen with specialized coupling agents.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the recyclability of the aqueous surfactant system. Since the TPGS-750-M solution can be reused multiple times without significant degradation in performance, the effective cost of the reaction medium per unit of product drops dramatically over time. Additionally, the simplified workup procedure, which involves basic extraction and drying rather than complex chromatographic purification or neutralization, reduces labor hours and consumable usage. This lean manufacturing approach directly contributes to improved gross margins and a more competitive pricing structure for the final intermediates supplied to pharmaceutical clients.
- Enhanced Supply Chain Reliability: Relying on water as the primary solvent and readily available inorganic salts minimizes supply chain risks associated with the logistics of hazardous chemicals. Transporting and storing large quantities of flammable or toxic organic solvents requires specialized infrastructure and permits, which can be bottlenecks in global supply chains. By contrast, the materials used in this green synthesis are non-hazardous and widely accessible, ensuring uninterrupted production schedules. This reliability is critical for meeting the just-in-time delivery expectations of multinational pharmaceutical partners who require consistent volumes of high-quality intermediates to support their own clinical and commercial timelines.
- Scalability and Environmental Compliance: The inherent safety of the aqueous system facilitates easier scale-up from kilogram to tonnage production. The absence of exothermic risks associated with strong bases or reactive acyl chlorides allows for larger batch sizes without proportional increases in cooling capacity or safety containment measures. From a regulatory standpoint, the process generates significantly less hazardous waste, simplifying compliance with increasingly stringent environmental protection laws. This proactive alignment with green chemistry standards future-proofs the manufacturing site against potential regulatory crackdowns and enhances the corporate reputation of the supplier as a responsible partner in the sustainable development of new medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and expected outcomes. Understanding these details helps stakeholders make informed decisions about integrating this methodology into their existing manufacturing portfolios.
Q: What are the primary advantages of using TPGS-750-M in this synthesis?
A: TPGS-750-M acts as a designer surfactant that forms nanoreactors in water, enabling organic reactions to proceed efficiently without toxic organic solvents. It allows for the recycling of the aqueous medium multiple times without significant loss in yield, drastically reducing waste disposal costs.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process is highly scalable because it utilizes water as the primary solvent and avoids hazardous reagents like oxalyl chloride or chlorinated solvents. The mild reaction conditions (60°C, air atmosphere) simplify safety protocols and equipment requirements for commercial manufacturing.
Q: How does the purity of the product compare to traditional methods?
A: The method yields high-purity products directly after simple extraction and drying, eliminating the need for complex acid-base neutralization steps often required in traditional syntheses. This streamlined workup minimizes impurity formation and simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted-1,4-Dihydro-2,3-Quinoxalinedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry in driving the next generation of pharmaceutical manufacturing. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110642798B are successfully translated into robust industrial processes. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your quinoxalinedione requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this technology can optimize your supply chain economics. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate the development of your life-saving therapies through superior chemical manufacturing solutions.
