Advanced Manufacturing of Rivaroxaban Intermediates: A Technical Breakthrough for Scalable API Production
Advanced Manufacturing of Rivaroxaban Intermediates: A Technical Breakthrough for Scalable API Production
The global demand for direct Factor Xa inhibitors continues to surge, driven by the critical need for effective anticoagulant therapies in managing cardiovascular disorders such as deep vein thrombosis and pulmonary embolism. At the forefront of this therapeutic class is Rivaroxaban, a molecule whose complex stereochemistry and functional group density present significant challenges for process chemists aiming for cost-effective manufacturing. Patent CN103864773A introduces a transformative synthetic strategy that addresses these bottlenecks by utilizing a novel intermediate, designated as Formula (5). This technical insight report analyzes the mechanistic elegance and commercial viability of this route, offering a compelling value proposition for R&D directors and procurement leaders seeking a reliable pharmaceutical intermediates supplier. By shifting away from traditional, reagent-intensive pathways, this method leverages acylation modification to achieve high yields and operational simplicity.
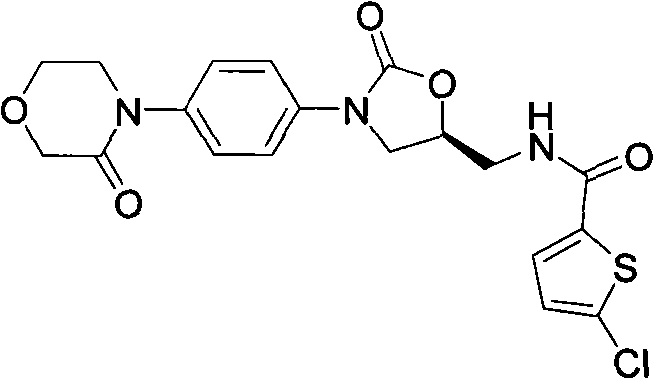
Rivaroxaban, chemically known as 5-chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-1,3-oxazolidin-5-yl}methyl)-2-thiophenecarboxamide, represents a pinnacle of modern medicinal chemistry. However, its commercial success is heavily dependent on the efficiency of its supply chain. The patent data reveals that while previous methods relied on costly cyclization agents and harsh conditions, the new approach streamlines the synthesis through a protected intermediate strategy. This not only enhances the overall throughput but also ensures consistent quality, a paramount concern for high-purity rivaroxaban intermediate sourcing. The ability to produce this critical scaffold with reduced environmental impact and lower material costs positions this technology as a cornerstone for next-generation API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rivaroxaban has been plagued by several intrinsic inefficiencies that hinder large-scale production. Early routes, such as those disclosed in WO2005/068456 and CN1262551, often necessitated the use of carbonyldiimidazole (CDI) for ring closure. While chemically effective, CDI is an expensive reagent that generates imidazole byproducts, complicating downstream purification and increasing waste disposal costs. Furthermore, these legacy processes frequently employed toxic solvents like toluene and required corrosive reagents such as acetic anhydride and hydrogen bromide under reflux conditions. The reliance on chiral column chromatography for enantiomeric separation, as seen in US 2007/157456, is another major bottleneck; this technique is notoriously difficult to scale and imposes severe limitations on throughput, making it unsuitable for meeting the demands of a commercial scale-up of complex anticoagulants. Additionally, the use of hazardous reagents like phosgene or butyllithium in alternative pathways introduces significant safety risks and regulatory hurdles, further inflating the cost of goods sold (COGS).
The Novel Approach
In stark contrast, the methodology outlined in CN103864773A offers a robust solution by introducing a stable, isolable intermediate, Formula (5). This route bypasses the need for expensive cyclization promoters in the early stages by utilizing an acylation-modified precursor, Formula (3). The reaction of this precursor with 4-(3-oxomorpholino)phenyl carbamate (Formula 4) proceeds under mild base catalysis to form the oxazolidinone ring with exceptional efficiency. As illustrated in the comprehensive reaction scheme below, this pathway avoids the pitfalls of low-yielding extraction steps and high-temperature refluxes associated with prior art.
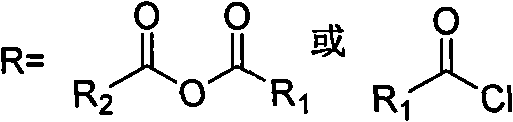
The strategic advantage lies in the stability and purifiability of Formula (5). Unlike transient intermediates in other routes, Formula (5) can be isolated and purified via simple recrystallization from ethanol, effectively removing impurities before the final deprotection and acylation steps. This "purify-early" philosophy drastically reduces the burden on the final API purification, leading to higher overall yields and superior product quality. By eliminating the need for chiral columns and replacing hazardous reagents with safer, commercially available alternatives like benzyl chloroformate and acetic anhydride, this novel approach delivers substantial cost reduction in API manufacturing while enhancing process safety and environmental compliance.
Mechanistic Insights into Acylation-Driven Cyclization
The core innovation of this synthesis lies in the precise manipulation of the chiral center and the oxazolidinone ring formation. The process begins with the acylation of S-1-amino-3-chloro-2-propanol (Formula 2) to generate Formula (3). This acylation step is critical as it protects the amine and activates the alcohol for subsequent nucleophilic attack, setting the stage for stereoselective ring closure. When Formula (3) reacts with the aniline derivative Formula (4) in the presence of a base such as lithium tert-butoxide or sodium methoxide, an intramolecular substitution occurs. The phenolic oxygen or the carbamate nitrogen attacks the activated carbon, displacing the chloride and closing the five-membered oxazolidinone ring. This mechanism is highly favorable due to the entropic advantage of intramolecular reactions and the electronic activation provided by the acyl group.
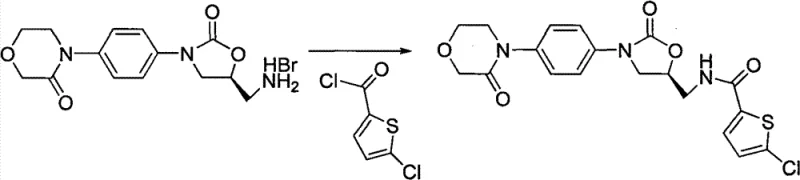
Following the formation of the key intermediate Formula (5), the synthesis proceeds through a controlled acid hydrolysis step. Using hydrobromic acid or hydrochloric acid, the acyl protecting groups are cleaved to reveal the free amine in Formula (6). This step is remarkably efficient, with yields exceeding 90%, demonstrating the robustness of the intermediate design. The final transformation involves the acylation of the primary amine in Formula (6) with 5-chlorothiophene-2-formyl chloride (Formula c). This reaction, typically conducted in dichloromethane with a tertiary amine base like tri-n-butylamine, forms the final amide bond of Rivaroxaban. The mild conditions (0-20°C) prevent racemization and side reactions, ensuring the optical purity of the final product is maintained throughout the sequence.
Impurity control is inherently built into this mechanistic pathway. The use of benzyl chloroformate to generate Formula (4) creates a carbamate that is stable enough to withstand the cyclization conditions but labile enough to be removed or transformed in later steps without generating persistent byproducts. Furthermore, the crystallization of Formula (5) acts as a powerful purification gate, rejecting structurally similar impurities that might arise from incomplete acylation or side reactions. This rigorous control over the impurity profile is essential for meeting the stringent regulatory requirements for anticoagulant APIs, where even trace impurities can have significant biological consequences.
How to Synthesize Rivaroxaban Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters, particularly temperature control and stoichiometry during the cyclization and hydrolysis steps. The patent provides detailed embodiments demonstrating that the reaction between Formula (3) and Formula (4) achieves optimal yields (up to 93%) when conducted in polar aprotic solvents like DMF or NMP at temperatures between 20°C and 65°C. The subsequent hydrolysis to Formula (6) is best performed using concentrated hydrobromic acid at elevated temperatures (40-100°C) to ensure complete deprotection. For the final acylation, maintaining low temperatures (0-5°C) is crucial to minimize the formation of N-acyl urea byproducts and ensure high purity. The detailed standardized synthesis steps for implementing this high-yield pathway are provided in the guide below.
- Acylation of S-1-amino-3-chloro-2-propanol (Formula 2) using acetic anhydride or acid chlorides to yield Formula (3).
- Base-catalyzed reaction of Formula (3) with 4-(3-oxomorpholino)phenyl carbamate (Formula 4) to generate the key intermediate Formula (5).
- Acid hydrolysis of Formula (5) to obtain Formula (6), followed by final acylation with 5-chlorothiophene-2-formyl chloride to yield Rivaroxaban.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary driver is the significant optimization of the cost structure. By eliminating the dependency on expensive reagents like CDI and avoiding the capital-intensive infrastructure required for chiral chromatography, manufacturers can achieve a leaner cost base. The raw materials utilized, such as 4-(3-oxomorpholino)aniline and benzyl chloroformate, are commodity chemicals with stable supply chains, mitigating the risk of raw material shortages. This stability translates directly into reducing lead time for high-purity pharmaceutical intermediates, allowing for more responsive inventory management and just-in-time delivery models.
- Cost Reduction in Manufacturing: The economic argument for this route is compelling. The replacement of high-cost cyclization agents with inexpensive acylating reagents like acetic anhydride drastically lowers the direct material costs. Furthermore, the simplification of the purification process—replacing complex chromatographic separations with standard crystallizations—reduces solvent consumption and processing time. This operational efficiency leads to substantial savings in utility costs and waste treatment, contributing to a lower overall cost of goods sold without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Unlike specialized chiral building blocks that may have single-source suppliers, the precursors for Formula (3) and Formula (4) are produced by multiple vendors globally. This diversification reduces supply risk and provides procurement teams with greater negotiating leverage. Additionally, the robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures or strict environmental controls associated with hazardous reagents, ensuring a steady flow of intermediates to the API production line.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is superior. The avoidance of toxic solvents like toluene and hazardous reagents like phosgene aligns with modern green chemistry principles and increasingly stringent environmental regulations. The process generates less hazardous waste, simplifying disposal and reducing compliance costs. The mild reaction temperatures and atmospheric pressure operations facilitate easy scale-up from pilot plant to multi-ton commercial production, ensuring that supply can be rapidly ramped up to meet market demand without the need for specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Rivaroxaban synthesis pathway. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of the Formula (5) intermediate route over conventional CDI methods?
A: The novel route utilizing intermediate Formula (5) eliminates the need for expensive carbonyldiimidazole (CDI) reagents and toxic solvents like toluene found in older patents. It offers significantly higher yields (over 90% in key steps), milder reaction conditions, and simplified purification processes suitable for industrial scale-up.
Q: How does this synthesis method impact the purity profile of the final Rivaroxaban API?
A: By introducing a stable intermediate (Formula 5) that can be easily purified via recrystallization from ethanol, the process effectively removes impurities before the final cyclization and acylation steps. This results in a final product with superior purity specifications and a cleaner impurity profile compared to routes requiring chiral column separation.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrialization. It utilizes readily available raw materials such as benzyl chloroformate and avoids hazardous reagents like phosgene or butyllithium. The reaction conditions are mild (20-65°C), and the work-up procedures involve standard extraction and crystallization, ensuring robust scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivaroxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex anticoagulants hinges on the reliability and quality of the supply chain. Our technical team has extensively evaluated the route described in CN103864773A and confirmed its potential for high-efficiency manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of intermediates regardless of market fluctuations. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Rivaroxaban intermediate meets the highest international pharmacopoeia standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthetic technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your API supply chain with a solution that balances technical excellence with commercial pragmatism.
