Advanced Rhodium Catalysis for Scalable Indole-3-Carboxamide Manufacturing and Commercial Supply
Advanced Rhodium Catalysis for Scalable Indole-3-Carboxamide Manufacturing and Commercial Supply
The pharmaceutical and agrochemical industries continuously demand more efficient, sustainable, and cost-effective pathways for constructing critical molecular scaffolds, particularly amide bonds which serve as the backbone of countless bioactive molecules. Patent CN109761875B introduces a groundbreaking methodology for the synthesis of heteroaromatic amide derivatives, specifically targeting the challenging construction of indole-3-carboxamides through a novel transition metal-catalyzed cascade. This technology leverages the unique reactivity of rhodium complexes to facilitate the coupling of organic azides and carbon monoxide with diverse heteroarenes, bypassing the limitations of classical acylation strategies. By utilizing readily available starting materials such as substituted indoles and sulfonyl azides under mild conditions, this invention provides a robust platform for generating high-value intermediates essential for drug discovery and process development. The significance of this patent lies not only in its chemical innovation but also in its potential to streamline supply chains for complex nitrogen-containing compounds, offering a viable alternative to legacy synthetic routes that often suffer from poor atom economy and harsh operational requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of amide bonds in fine chemical manufacturing has relied heavily on the condensation of amines with activated carboxylic acid derivatives, such as acid chlorides or mixed anhydrides, which inherently suffer from significant drawbacks regarding sustainability and operational safety. These classical approaches typically necessitate the use of stoichiometric amounts of strong Lewis acid activating reagents or coupling agents, leading to the generation of substantial quantities of salt waste and complicating downstream purification processes. Furthermore, alternative transition metal-catalyzed carbonylation methods, such as palladium-catalyzed aminocarbonylation of aryl halides, require the prior installation of halogen handles on the aromatic ring, adding extra synthetic steps and increasing the overall cost of goods. The reliance on pre-functionalized substrates not only reduces the overall atom economy of the process but also limits the structural diversity accessible to medicinal chemists who wish to explore late-stage functionalization of sensitive heterocyclic cores. Additionally, many conventional protocols demand rigorous exclusion of moisture and oxygen or utilize toxic solvents, creating significant hurdles for safe implementation in large-scale commercial facilities where environmental compliance and operator safety are paramount concerns.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in patent CN109761875B employs a direct C-H functionalization strategy that merges organic azides and carbon monoxide in the presence of a rhodium catalyst to forge the amide linkage with exceptional efficiency. This innovative route eliminates the need for pre-halogenated starting materials or activated acid chlorides, thereby simplifying the synthetic sequence and drastically reducing the consumption of auxiliary reagents. The process operates under relatively mild thermal conditions, typically around 80°C in acetonitrile, and crucially, proceeds without the requirement for additional bases, oxidants, or specialized ligands that often complicate reaction workups. By directly utilizing the intrinsic reactivity of the heteroarene C-H bond, this approach achieves high regioselectivity at the C3 position of the indole ring, a feat that is difficult to accomplish with traditional electrophilic substitution methods without extensive protecting group manipulation. The simplicity of the reaction setup, combined with the use of gaseous carbon monoxide as a C1 building block, represents a paradigm shift towards more streamlined and economically viable manufacturing processes for complex amide-containing pharmaceutical intermediates.
Mechanistic Insights into Rhodium-Catalyzed Nitrene Formation and CO Insertion
The core of this transformative chemistry lies in the generation of a highly reactive rhodium-nitrene intermediate formed through the decomposition of the organic azide by the rhodium catalyst, specifically the dimeric complex [Rh(cod)Cl]2. Upon exposure to carbon monoxide, this transient metal-nitrene species undergoes a migratory insertion to form a rhodium-isocyanate intermediate, which serves as the key electrophilic partner in the subsequent bond-forming event. This mechanistic pathway is distinct from standard carbonylation reactions as it avoids the formation of discrete acyl-metal species derived from oxidative addition, instead leveraging the unique electronic properties of the nitrene moiety to activate the CO molecule. The resulting rhodium-isocyanate complex is sufficiently electrophilic to engage in a nucleophilic attack by the electron-rich C3 position of the indole substrate, facilitating the formation of the new carbon-carbon bond and the eventual amide functionality after proton transfer. Understanding this catalytic cycle is crucial for process optimization, as it highlights the importance of maintaining a steady concentration of CO to drive the insertion step forward while preventing the dimerization or decomposition of the reactive nitrene species.
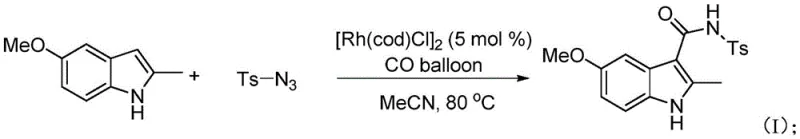
Furthermore, the regioselectivity observed in this transformation is dictated by the electronic nature of the heteroaromatic substrate, where the C3 position of the indole ring possesses the highest nucleophilicity and is therefore the preferred site for coupling with the rhodium-isocyanate intermediate. This inherent selectivity minimizes the formation of regioisomeric impurities, which is a critical quality attribute for pharmaceutical intermediates where isomeric purity can significantly impact biological activity and regulatory approval. The tolerance of the catalytic system towards various substituents on both the indole ring and the sulfonyl azide component suggests a broad substrate scope, allowing for the synthesis of a diverse library of derivatives without the need for extensive re-optimization of reaction parameters. From a process chemistry perspective, the absence of competing side reactions, such as azide reduction or homocoupling, further underscores the robustness of this rhodium-mediated pathway, ensuring that the final product profile remains clean and amenable to standard purification techniques like column chromatography or crystallization.
How to Synthesize Indole-3-Carboxamides Efficiently
To implement this advanced synthetic methodology in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that ensure optimal catalyst performance and safety when handling carbon monoxide and organic azides. The general protocol involves charging a reaction vessel with the heteroaromatic substrate, the chosen organic azide, and the rhodium catalyst in a polar aprotic solvent, followed by the careful introduction of CO gas to establish the necessary atmosphere for the carbonylation step. While the detailed stoichiometry and specific temperature profiles may vary slightly depending on the electronic properties of the substrates, the fundamental workflow remains consistent across the examples provided in the patent documentation. For a comprehensive understanding of the precise operational parameters, including catalyst loading, reaction times, and workup procedures, please refer to the standardized synthesis guide provided below which encapsulates the critical steps for successful execution.
- Combine the heteroaromatic substrate (e.g., N-methylindole), organic azide (e.g., sulfonyl azide), and [Rh(cod)Cl]2 catalyst in acetonitrile solvent.
- Introduce carbon monoxide (CO) gas into the reaction vessel and maintain the temperature at 80°C for approximately 12 hours.
- Upon completion, remove the solvent under reduced pressure and purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed technology offers compelling strategic advantages by addressing several persistent pain points associated with the sourcing and manufacturing of complex amide intermediates. The primary benefit stems from the drastic simplification of the raw material portfolio, as the process relies on commodity chemicals like indoles and sulfonyl azides rather than expensive, custom-synthesized acid chlorides or halogenated precursors that often have long lead times and volatile pricing. By removing the dependency on these specialized starting materials, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing through improved supply chain resilience and reduced exposure to single-source supplier risks. Moreover, the elimination of stoichiometric activating reagents and the associated waste streams translates to lower disposal costs and a reduced environmental footprint, aligning with increasingly stringent global regulations on chemical manufacturing sustainability.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the high atom economy and the use of catalytic amounts of rhodium, which, despite being a precious metal, is used at low loadings and can potentially be recovered and recycled in continuous flow setups. Unlike traditional methods that generate equimolar amounts of salt waste requiring costly neutralization and disposal, this catalytic cycle produces minimal byproducts, thereby lowering the overall cost of goods sold. The avoidance of cryogenic conditions or high-pressure reactors further reduces capital expenditure and energy consumption, making the process financially attractive for both small-batch clinical supply and large-scale commercial production. Additionally, the high yields reported in the patent, often exceeding 90%, minimize the loss of valuable starting materials, ensuring that the theoretical cost savings are realized in practical operations.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of stable and commercially available reagents such as acetonitrile and common sulfonyl azides ensures a consistent and reliable supply of inputs, mitigating the risk of production delays caused by raw material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-dry solvents, simplifies the quality control requirements for incoming materials, allowing for greater flexibility in vendor selection. This flexibility is crucial for maintaining business continuity in a global market where geopolitical factors can disrupt the flow of specialized chemicals. Furthermore, the scalability of the reaction from milligram to kilogram scales without significant loss in efficiency means that supply partners can rapidly ramp up production to meet fluctuating demand without the need for extensive process re-validation.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to conventional acylation methods, as it avoids the use of chlorinating agents like thionyl chloride or oxalyl chloride which pose significant safety and corrosion hazards in large reactors. The benign nature of the byproducts and the use of standard organic solvents facilitate easier waste treatment and solvent recovery, supporting corporate sustainability goals and reducing the regulatory burden associated with hazardous waste management. The mild reaction temperature of 80°C allows for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure or low-temperature equipment, lowering the barrier to entry for contract manufacturing organizations. This ease of scale-up ensures that the technology can be seamlessly transferred from R&D laboratories to multi-ton production facilities, securing a stable long-term supply of high-purity intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed amide bond construction method, drawing directly from the experimental data and beneficial effects described in the patent literature. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for the production of indole-based scaffolds. By understanding the specific advantages related to catalyst selection, substrate scope, and reaction conditions, stakeholders can make informed decisions about integrating this methodology into their existing manufacturing portfolios. The answers provided reflect the optimized parameters identified during the patent development process, ensuring accuracy and relevance for technical evaluation.
Q: What represents the primary advantage of this Rh-catalyzed method over traditional acylation?
A: Unlike traditional methods requiring pre-functionalized acid chlorides or aryl halides which generate significant waste, this method utilizes direct C-H activation with simple organic azides and CO, offering superior atom economy and eliminating the need for strong Lewis acid activators.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the reaction conditions are mild (80°C), use common solvents like acetonitrile, and do not require sensitive additives, making the protocol highly robust and adaptable for commercial scale-up from kilogram to tonnage levels.
Q: What is the regioselectivity of the amide bond formation on the indole ring?
A: The reaction exhibits excellent regioselectivity, specifically constructing the amide bond at the C3 position of the indole ring due to the high nucleophilicity and activity of the C-H bond at this specific location during the rhodium-isocyanate intermediate coupling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-3-Carboxamide Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies such as the rhodium-catalyzed carbonylation described in patent CN109761875B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your project transitions smoothly from benchtop discovery to full-scale manufacturing without compromising on quality or timeline. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch, ensuring that our indole-3-carboxamide derivatives meet the exacting standards required by the pharmaceutical industry. Our commitment to technical excellence allows us to navigate the complexities of transition metal catalysis, managing catalyst removal and residual metal specifications to levels that satisfy even the most demanding regulatory requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can be tailored to your specific project needs, potentially offering a Customized Cost-Saving Analysis that quantifies the economic benefits of switching from your current legacy process. By collaborating with us, you gain access to our deep reservoir of process knowledge and the ability to rapidly prototype new analogues using this versatile chemistry. We encourage you to request specific COA data and route feasibility assessments for your target molecules, allowing us to demonstrate concretely how our capabilities align with your supply chain objectives. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
