Revolutionizing Chiral Pyrroloimidazole Production: Advanced Asymmetric Catalysis for Commercial Scale
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the demand for highly efficient and stereoselective synthetic routes. Patent CN114105993B introduces a groundbreaking methodology for the preparation of chiral pyrroloimidazole compounds, a critical structural motif found in numerous bioactive molecules and advanced pharmaceutical intermediates. This innovation leverages an asymmetric catalytic reaction system that utilizes alpha-amino ketones and alpha,beta-unsaturated aldehydes as key building blocks, orchestrated by a chiral secondary amine catalyst. Unlike traditional approaches that often rely on stoichiometric chiral auxiliaries or expensive transition metals, this organocatalytic strategy offers a streamlined pathway that enhances atom economy while minimizing environmental impact. The technical breakthrough lies in the seamless integration of cyclization, selective reduction, and aromatization steps, which collectively ensure high yields and exceptional stereochemical control. For global procurement teams and R&D directors, this patent represents a pivotal shift towards more sustainable and cost-effective production capabilities, addressing the growing need for reliable supply chains in the competitive pharmaceutical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrroloimidazole derivatives has been fraught with significant technical challenges that hinder large-scale commercial viability. Conventional methodologies often involve complex multi-step sequences requiring harsh reaction conditions, such as strong bases, high temperatures, or toxic heavy metal catalysts like silver or palladium complexes. As illustrated in the prior art comparison, earlier strategies frequently suffer from poor regioselectivity and low overall yields due to the formation of unwanted by-products during cyclization events. Furthermore, the reliance on precious metal catalysts introduces severe complications regarding residual metal removal, which is a critical quality attribute for any pharmaceutical intermediate intended for human consumption. These legacy processes also tend to generate substantial amounts of hazardous waste, increasing the burden on environmental compliance and waste treatment infrastructure. Consequently, manufacturers face inflated production costs and extended lead times, creating bottlenecks that disrupt the stability of the global supply chain for these essential chemical building blocks.
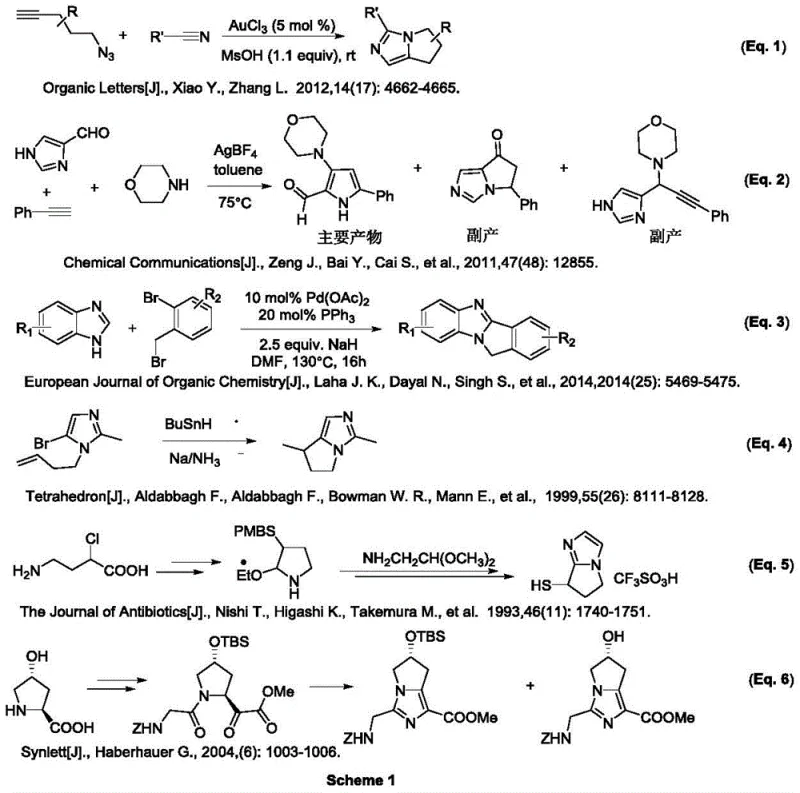
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in the patent utilizes a sophisticated organocatalytic cascade that dramatically simplifies the synthetic architecture. By employing a chiral secondary amine catalyst in conjunction with specific acidic or basic additives, the reaction proceeds through a highly controlled asymmetric cyclization mechanism that inherently favors the desired stereoisomer. This method operates under remarkably mild conditions, typically at ambient or slightly elevated temperatures, which significantly reduces energy consumption and operational risks associated with high-pressure or high-temperature reactors. The elimination of transition metals not only lowers the raw material costs but also streamlines the purification workflow, as there is no need for expensive scavenging resins or complex filtration steps to meet strict heavy metal specifications. Moreover, the versatility of this system allows for a broad substrate scope, accommodating various functional groups on the aromatic rings without compromising the efficiency of the transformation. This robustness makes it an ideal candidate for industrial adoption, offering a clear pathway to cost reduction in fine chemical manufacturing while maintaining the highest standards of product quality.
Mechanistic Insights into Chiral Secondary Amine Catalyzed Cyclization
The core of this technological advancement resides in the precise mechanistic orchestration of the asymmetric cyclization step, which sets the stereochemical foundation for the entire synthesis. The chiral secondary amine catalyst activates the alpha,beta-unsaturated aldehyde through the formation of a reactive iminium ion intermediate, thereby lowering the LUMO energy level and facilitating nucleophilic attack by the alpha-amino ketone. This activation mode ensures that the carbon-carbon bond formation occurs with high facial selectivity, dictated by the steric environment of the chiral catalyst scaffold. Following the initial cyclization, the resulting intermediate undergoes a selective hydroxyl reduction and dehydration sequence, which is carefully tuned to prevent epimerization at the newly formed chiral centers. The use of specific silane reducing agents in the presence of Lewis acids allows for the gentle removal of the hydroxyl group while preserving the integrity of the sensitive pyrrolidine ring system. This level of control is paramount for achieving the high diastereomeric ratios observed in the experimental data, ensuring that the downstream aromatization step proceeds cleanly to the final target structure.
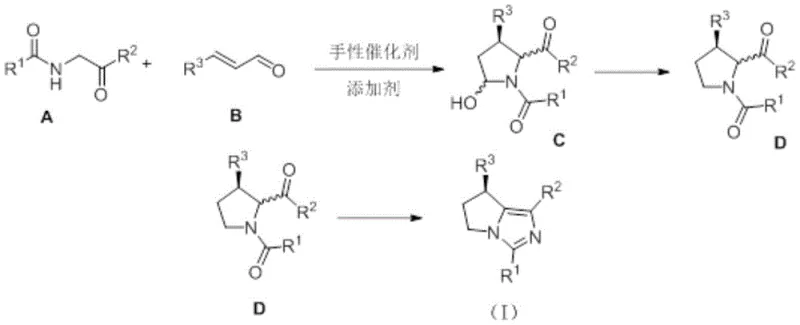
Furthermore, the final aromatization step is engineered to be completely free of racemization, a common pitfall in many heterocyclic synthesis protocols. By utilizing ammonium salts as the nitrogen source in a polar aprotic solvent system, the reaction promotes the closure of the imidazole ring without disturbing the existing chiral configuration established in the earlier steps. The mechanistic pathway avoids the formation of planar intermediates that could lead to loss of optical purity, thereby guaranteeing that the high enantiomeric excess achieved in the first step is carried through to the final product. Impurity control is inherently built into this design, as the specific reactivity of the catalyst suppresses side reactions such as polymerization of the aldehyde or self-condensation of the ketone. For R&D directors evaluating process feasibility, this mechanistic robustness translates to a more predictable and reproducible manufacturing process, reducing the risk of batch failures and ensuring consistent quality across different production scales. The ability to fine-tune the electronic properties of the substrates through various substituents further enhances the utility of this method for generating diverse libraries of chiral intermediates.
How to Synthesize Chiral Pyrroloimidazole Efficiently
The practical implementation of this synthesis route involves a well-defined three-step protocol that balances reaction efficiency with operational simplicity. The process begins with the asymmetric cyclization in a suitable organic solvent, followed by a direct reduction workup, and concludes with a thermal aromatization step using an ammonia source. Detailed standardized operating procedures for each stage, including specific molar ratios, temperature profiles, and purification techniques, are essential for replicating the high yields and selectivity reported in the patent literature. Understanding the nuances of reagent addition and reaction monitoring is critical for maximizing the throughput of this valuable transformation in a commercial setting.
- Perform asymmetric cyclization of alpha-amino ketones and alpha,beta-unsaturated aldehydes using a chiral secondary amine catalyst.
- Execute selective hydroxyl reduction and dehydration to form the diastereomeric pyrrolidine intermediate.
- Complete the synthesis via racemization-free cyclization and aromatization using an ammonia source to yield the final chiral pyrroloimidazole structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this organocatalytic technology offers profound advantages that extend far beyond simple chemical curiosity. The shift away from precious metal catalysts to organic amines represents a fundamental change in the cost structure of production, removing the volatility associated with the pricing of rhodium, palladium, or silver. This stability allows for more accurate long-term budgeting and contract negotiations, providing supply chain managers with greater predictability in their raw material expenditures. Additionally, the simplified purification requirements mean that processing times can be significantly shortened, leading to faster turnaround times from order placement to delivery. The reduced complexity of the waste stream also lowers the environmental compliance costs, making the overall manufacturing process more sustainable and aligned with modern green chemistry initiatives. These factors combine to create a compelling value proposition for companies seeking to optimize their supply chain resilience and reduce total cost of ownership for critical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated removal steps leads to substantial cost savings in the overall production budget. By utilizing readily available organic catalysts and common reducing agents, the raw material costs are drastically minimized compared to traditional metal-mediated pathways. Furthermore, the high yields achieved in each step reduce the amount of starting material required per unit of product, enhancing the overall material efficiency of the process. The simplified workup procedures also decrease the consumption of solvents and consumables, contributing to a leaner and more economical manufacturing operation that directly improves profit margins.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures a robust supply chain that is less susceptible to geopolitical disruptions or shortages of specialized reagents. Unlike processes dependent on scarce noble metals, this organocatalytic route utilizes commodity chemicals that can be sourced from multiple vendors globally. This diversification of the supply base mitigates the risk of single-source dependency and ensures continuous production capability even during market fluctuations. The mild reaction conditions also allow for manufacturing in a wider range of facilities, increasing the flexibility of the production network and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The inherent safety of the mild reaction conditions facilitates straightforward scale-up from pilot plant to full commercial production without the need for specialized high-pressure equipment. The absence of toxic heavy metals simplifies the environmental permitting process and reduces the liability associated with hazardous waste disposal. This alignment with green chemistry principles not only meets regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity. The process generates less hazardous waste and consumes less energy, making it a sustainable choice for long-term industrial operations focused on minimizing environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits and technical feasibility for potential partners. Understanding these details is crucial for making informed decisions about integrating this technology into existing production workflows.
Q: What are the primary advantages of this organocatalytic route over traditional metal-catalyzed methods?
A: This method eliminates the need for expensive transition metal catalysts and harsh reaction conditions, significantly reducing heavy metal contamination risks and simplifying downstream purification processes for pharmaceutical applications.
Q: How does this synthesis method impact the enantiomeric excess (ee) of the final product?
A: By utilizing specific chiral secondary amine catalysts and optimized additives, the process achieves high stereoselectivity, consistently delivering products with excellent enantiomeric excess values suitable for strict regulatory compliance.
Q: Is this synthetic route scalable for industrial manufacturing of fine chemical intermediates?
A: Yes, the protocol employs readily available starting materials and mild reaction conditions, facilitating straightforward scale-up from laboratory grams to multi-ton commercial production without compromising yield or purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Pyrroloimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial realities for our global partners. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this asymmetric catalytic route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral pyrroloimidazole intermediate meets the exacting standards required by the pharmaceutical industry. Our commitment to quality and consistency makes us a trusted partner for companies looking to secure a stable supply of high-value fine chemical intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume needs. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this technology for your supply chain. Let us collaborate to drive efficiency and innovation in your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
