Advanced Catalytic Synthesis of Chiral Phosphine Oxides for High-Value Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access high-value chiral building blocks, particularly those serving as critical ligands in asymmetric catalysis. Patent CN110078763B introduces a groundbreaking methodology for the synthesis of all-carbon-linked chiral phosphine oxide compounds, addressing long-standing challenges in stereocontrol and atom economy. This innovation leverages a copper-catalyzed azide-alkyne cycloaddition (CuAAC) strategy, specifically employing desymmetrization and kinetic resolution tactics to construct tetrahedral phosphorus centers with exceptional precision. For R&D directors and procurement specialists, this technology represents a significant leap forward, offering a robust alternative to traditional resolution methods that are often plagued by low yields and high waste generation. By utilizing base metal catalysis under mild conditions, this process not only enhances the sustainability profile of the synthesis but also opens new avenues for the cost-effective production of complex organophosphorus intermediates essential for modern drug discovery and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of all-carbon-linked chiral phosphine oxides has been a formidable challenge due to the tetrahedral geometry of the phosphorus atom, which complicates stereochemical control similar to quaternary carbon centers. Traditional approaches predominantly relied on chiral resolution techniques, which inherently suffer from a maximum theoretical yield of only 50%, effectively wasting half of the starting material and driving up production costs significantly. Furthermore, methods utilizing chiral auxiliaries often involve multi-step sequences for the attachment and subsequent removal of the auxiliary group, leading to poor atom utilization and increased environmental burden through excessive solvent and reagent consumption. Even recent attempts at asymmetric catalysis have frequently depended on expensive noble metal catalysts, which pose supply chain risks and require rigorous removal protocols to meet stringent pharmaceutical purity standards, thereby limiting their practical utility in large-scale manufacturing environments.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a highly efficient copper-catalyzed system that operates under remarkably mild conditions, typically ranging from -30°C to 50°C, to achieve high enantioselectivity and yield. This approach employs readily available diacetylene or monoalkyne substrates reacting with organic azides in the presence of specialized chiral ligands and copper salts, facilitating a direct and convergent route to the target molecules. The process eliminates the need for stoichiometric chiral reagents or precious metals, significantly reducing the raw material costs and simplifying the downstream purification processes. As illustrated in the general reaction scheme below, this strategy allows for the flexible modification of substituents on the phosphorus center, providing a versatile platform for generating diverse libraries of chiral ligands tailored for specific catalytic applications.

Mechanistic Insights into Copper-Catalyzed Azide-Alkyne Cycloaddition
The success of this synthetic route lies in the intricate dual-copper catalytic cycle that governs the regioselective formation of the 1,4-substituted triazole ring while simultaneously establishing the chiral phosphorus center. The mechanism initiates with the coordination of a terminal alkyne to a copper species to form a pi-alkyne complex, which is subsequently deprotonated by a second copper molecule to generate a sigma-alkynyl copper intermediate. This activation step is crucial as it enhances the nucleophilicity of the alkyne, allowing it to attack the electrophilic terminal nitrogen of the coordinated azide molecule. The formation of a cyclic metal intermediate stabilizes the transition state through electronic interactions between the two copper atoms, ensuring that the reaction proceeds with high fidelity and minimal side product formation. Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters such as temperature and catalyst loading to maximize throughput and optical purity in a production setting.
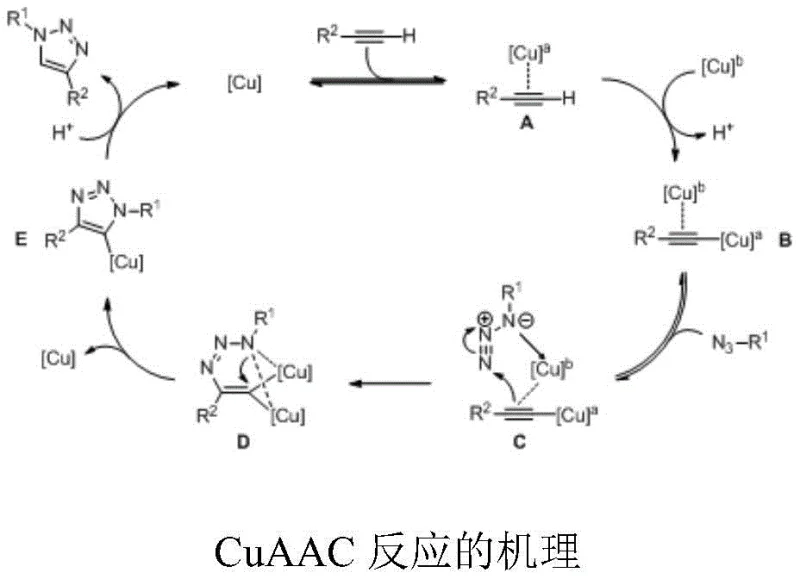
Furthermore, the chiral environment provided by the specialized ligands, such as the pyridine-bis(oxazoline) derivatives described in the patent, plays a pivotal role in discriminating between the enantiotopic groups of the prochiral substrate. The steric bulk and electronic properties of these ligands create a confined space around the active copper center, effectively guiding the approach of the azide reactant to favor the formation of one enantiomer over the other. This level of control is essential for producing high-purity intermediates required for sensitive biological applications, where even trace amounts of the wrong enantiomer can compromise the efficacy or safety of the final drug product. The ability to tune the ligand structure allows for the adaptation of this chemistry to a wide range of substrates, ensuring broad applicability across different chemical series and enhancing the overall robustness of the manufacturing process.
How to Synthesize Chiral Phosphine Oxides Efficiently
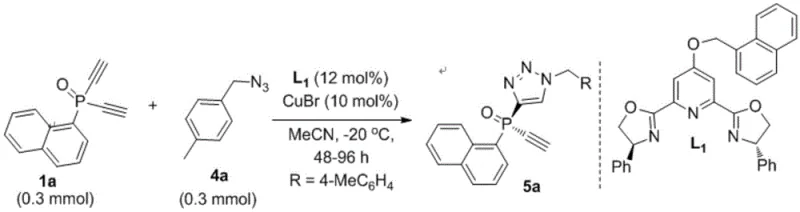
Implementing this synthesis in a laboratory or pilot plant setting involves a straightforward protocol that balances reaction efficiency with operational simplicity. The process typically begins with the preparation of the catalytic solution by mixing the chiral ligand and copper salt in an anhydrous solvent like acetonitrile, followed by the addition of the alkyne substrate at low temperatures to prevent premature decomposition or racemization. Once the system is equilibrated, the azide component is introduced, and the mixture is stirred for an extended period, often between 48 to 96 hours, to ensure complete conversion while maintaining high enantiomeric excess. The following example demonstrates the practical execution of this method, highlighting the specific conditions and reagents used to achieve optimal results for a representative compound.
- Prepare the catalytic system by mixing a chiral ligand (such as L1, L2, or L3) with a copper salt catalyst like cuprous bromide in an anhydrous solvent such as acetonitrile.
- Add the prochiral diacetylene or monoalkyne substrate to the reaction mixture and stir at low temperatures, typically around -20°C, to ensure high enantioselectivity.
- Introduce the organic azide reactant slowly, maintain the reaction temperature between -30°C and 50°C for 48 to 96 hours, and isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The shift from precious metal catalysts to abundant copper salts drastically reduces the raw material expenditure and mitigates the volatility associated with the pricing of rare earth metals, leading to more predictable budgeting and cost structures. Additionally, the mild reaction conditions reduce the energy consumption required for heating or cooling, contributing to lower operational expenditures and a smaller carbon footprint, which aligns with increasingly strict environmental regulations and corporate sustainability goals. The simplified workup procedures, which often avoid complex extraction or heavy metal scavenging steps, further streamline the production timeline, allowing for faster turnaround times and improved responsiveness to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and chiral auxiliaries significantly lowers the direct material costs associated with producing these high-value intermediates. By utilizing base metal catalysis and avoiding the 50% yield loss inherent in resolution processes, the overall cost per kilogram of the final product is substantially decreased, enhancing profit margins for downstream applications. This economic efficiency is further amplified by the high atom economy of the cycloaddition reaction, which minimizes waste disposal costs and maximizes the utility of every gram of starting material purchased.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as simple alkynes and azides, ensures a robust and resilient supply chain that is less susceptible to disruptions compared to processes requiring specialized or proprietary reagents. The use of common solvents like acetonitrile and standard laboratory equipment facilitates easy technology transfer between different manufacturing sites, reducing the risk of production bottlenecks. This accessibility allows for the rapid scaling of production capacity to meet sudden increases in demand without the need for lengthy lead times associated with sourcing exotic catalysts or custom-synthesized auxiliaries.
- Scalability and Environmental Compliance: The mild operating temperatures and ambient pressure conditions make this process inherently safer and easier to scale from gram to ton quantities without requiring specialized high-pressure reactors or extreme thermal management systems. The reduction in hazardous waste generation and the avoidance of toxic heavy metals simplify the regulatory compliance landscape, reducing the administrative burden and potential liabilities associated with environmental permits. This scalability ensures that the technology can grow alongside the commercial success of the final pharmaceutical product, providing a seamless path from early-stage development to full-scale commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These insights are derived directly from the patent specifications and are intended to provide clarity for stakeholders evaluating the feasibility of integrating this method into their existing production workflows. Understanding these details is crucial for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this copper-catalyzed method over traditional resolution techniques?
A: Unlike traditional chiral resolution which is limited to a maximum theoretical yield of 50%, this asymmetric catalytic approach utilizes desymmetrization and kinetic resolution strategies to achieve significantly higher atom economy and yields exceeding 90% with excellent enantiomeric excess.
Q: Can the chirality of the phosphine oxide be retained during further derivatization?
A: Yes, the patent demonstrates that the synthesized chiral phosphine oxides can be successfully converted into chiral tertiary phosphines and other derivatives while maintaining high optical purity, making them versatile intermediates for complex ligand synthesis.
Q: Is this synthesis method suitable for large-scale industrial production?
A: The process utilizes mild reaction conditions, inexpensive copper catalysts instead of precious metals, and readily available starting materials, which collectively simplify the operational complexity and facilitate commercial scale-up for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is smooth and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of chiral phosphine oxide or phosphine ligand supplied adheres to the highest international standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient copper-catalyzed route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your development timelines and secure your supply chain for the future.
