Advanced Manufacturing of 3-Aminobenzo[d]isothiazole: A Safe and Scalable Route for Global Pharma Supply Chains
Advanced Manufacturing of 3-Aminobenzo[d]isothiazole: A Safe and Scalable Route for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways that balance chemical efficiency with operational safety, particularly for complex heterocyclic scaffolds like 3-aminobenzo[d]isothiazole. Patent CN102040564B introduces a transformative preparation method that addresses critical bottlenecks in the production of this valuable intermediate, which serves as a core structure for compounds exhibiting stabilizing and antifungal activities. This technical disclosure outlines a nucleophilic substitution strategy that replaces hazardous organometallic processes with a mild, phosphorus salt-mediated protocol. By leveraging common reagents such as PyBroP or PyBOP alongside readily available amines, the invention establishes a new standard for manufacturing reliability. For R&D directors and procurement specialists, this shift represents not just a chemical optimization, but a strategic advantage in securing a stable supply of high-purity pharmaceutical intermediates without the logistical burdens associated with dangerous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted benzo[d]isothiazoles has relied heavily on organometallic chemistry, a approach fraught with significant industrial challenges. As illustrated in prior art references such as Synthesis 1997, these traditional routes necessitate the use of highly reactive organometallic reagents, which are inherently unstable and pyrophoric. Handling such materials requires specialized infrastructure, inert atmosphere capabilities, and rigorous safety protocols that dramatically inflate operational expenditures. Furthermore, the quenching of these reactive species generates substantial volumes of hazardous waste, complicating environmental compliance and disposal logistics. The sensitivity of these reactions to moisture and oxygen often leads to inconsistent batch-to-batch reproducibility, posing a severe risk to supply chain continuity for downstream API manufacturers who demand strict quality control.
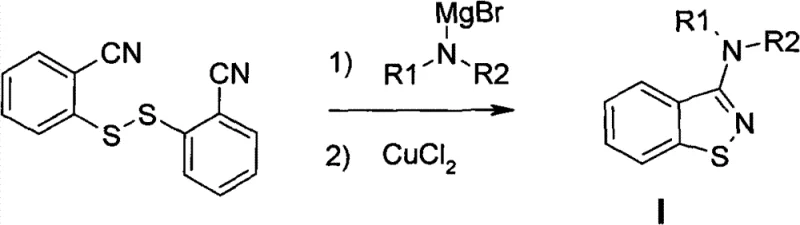
The Novel Approach
In stark contrast, the methodology disclosed in CN102040564B utilizes a benign nucleophilic substitution mechanism that operates under significantly milder conditions. The core innovation involves the activation of benzisothiazolone (Compound II) using phosphorus coupling reagents, which facilitates the subsequent attack by various amines (Compound III) to form the target 3-aminobenzo[d]isothiazole (Compound I). This route eliminates the need for cryogenic temperatures or strictly anhydrous environments typically required for organometallics, allowing reactions to proceed comfortably between 50°C and 100°C. The versatility of this approach is evident in its tolerance for a wide range of amine substrates, including cyclic amines like pyrrolidine and piperidine, as well as acyclic variants. By shifting to this chemistry, manufacturers can achieve yields generally reaching 70% to 80% while utilizing standard reactor setups, thereby democratizing access to this critical chemical building block.
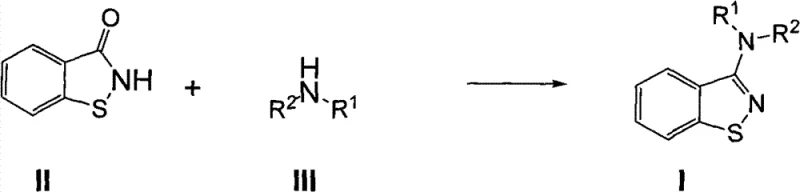
Mechanistic Insights into Phosphorus Salt-Mediated Activation
The success of this synthetic route hinges on the efficient activation of the carbonyl group within the benzisothiazolone ring by phosphorus salts such as PyBroP (tri(pyrrolidin-1-yl)phosphonium hexafluorophosphate) or PyBOP. In the initial step, the phosphorus reagent reacts with the lactam carbonyl in the presence of a base like triethylamine or sodium tert-butoxide to generate a highly electrophilic intermediate, likely an imidoyl phosphate or similar activated species. This activation lowers the energy barrier for nucleophilic attack, enabling even sterically hindered or less nucleophilic amines to react effectively. The choice of base and the molar ratios are critical; the patent specifies a molar ratio of Compound II to phosphorus salt to base ranging from 1:1:1 to 1:2:6, ensuring complete conversion of the starting material while minimizing side reactions. This precise stoichiometric control is key to maintaining high purity profiles essential for pharmaceutical applications.
Following activation, the introduction of the amine component drives the formation of the C-N bond through a classic nucleophilic acyl substitution pathway. The reaction kinetics are favorable at elevated temperatures (70-90°C), promoting the displacement of the phosphorus leaving group and the restoration of aromaticity or conjugation within the heterocyclic system. Importantly, the use of polar aprotic solvents like 1,4-dioxane or acetonitrile stabilizes the transition states and ensures homogeneous reaction conditions. From an impurity control perspective, this mechanism is clean; the byproducts are primarily phosphine oxides and amine salts, which are easily removed during the aqueous workup phase involving ethyl acetate extraction and brine washing. This simplicity in purification significantly reduces the burden on downstream processing teams.
How to Synthesize 3-Aminobenzo[d]isothiazole Efficiently
Implementing this synthesis requires careful attention to the sequence of reagent addition and temperature control to maximize the 70-80% yield potential described in the patent. The process begins with the activation of benzisothiazolone in a dry polar aprotic solvent under nitrogen protection, followed by the controlled addition of the amine substrate. While the general procedure is robust, scaling this reaction requires monitoring the exotherm during the activation phase and ensuring sufficient mixing during the heating stage to prevent localized hot spots. The detailed standardized synthesis steps, including specific reagent equivalents and workup procedures validated across multiple examples, are outlined below to guide process chemists in replication.
- Activate benzisothiazolone (Compound II) in a polar aprotic solvent like 1,4-dioxane using a phosphorus salt (PyBroP) and an organic base (Triethylamine) at 0-50°C.
- Introduce the amine component (Compound III) to the activated mixture and heat the reaction to 50-100°C to drive the nucleophilic substitution to completion.
- Perform standard aqueous workup involving ethyl acetate extraction, washing with water and brine, drying over sodium sulfate, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this phosphorus-mediated synthesis offers tangible benefits that extend beyond mere chemical yield. The elimination of hazardous organometallic reagents fundamentally alters the risk profile of the manufacturing process, leading to lower insurance costs and reduced regulatory overhead. Since the raw materials—benzisothiazolone, PyBroP/PyBOP, and common amines—are commercially available commodities, the supply chain is far less susceptible to the geopolitical or logistical disruptions that often plague specialty metal reagents. This reliability ensures consistent delivery schedules for downstream clients, a critical factor in Just-In-Time manufacturing environments.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the simplification of the operational workflow. By removing the need for specialized cryogenic equipment and expensive metal scavengers required to remove residual organometallics from the final product, the overall cost of goods sold is significantly reduced. Furthermore, the high atom economy and the ability to use stoichiometric amounts of relatively inexpensive coupling reagents mean that material costs are optimized. The simplified workup procedure, which avoids complex distillation or chromatography on a large scale due to the cleanliness of the reaction, further drives down processing time and utility consumption.
- Enhanced Supply Chain Reliability: Sourcing stability is markedly improved because the key reagents are stable solids or liquids with long shelf lives, unlike pyrophoric organometallics that degrade rapidly upon exposure to air. This allows manufacturers to maintain strategic stockpiles of raw materials without the fear of degradation, ensuring production continuity even during market fluctuations. The mild reaction conditions also mean that the process can be executed in a wider range of multipurpose chemical facilities, expanding the pool of qualified contract manufacturing organizations (CMOs) capable of producing this intermediate.
- Scalability and Environmental Compliance: From an environmental standpoint, this route is superior as it avoids the generation of heavy metal waste streams. The byproducts are primarily organic phosphates and salts that are easier to treat in standard wastewater facilities. This alignment with green chemistry principles facilitates smoother environmental permitting and reduces the carbon footprint associated with waste incineration. The scalability is proven by the broad temperature and solvent tolerance, allowing the reaction to be safely scaled from kilogram to multi-ton batches without encountering the heat transfer limitations often seen with highly exothermic organometallic additions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is essential for process engineers aiming to adapt this laboratory-scale method to full commercial production.
Q: Why is the new nucleophilic substitution method preferred over traditional organometallic routes?
A: Traditional methods often rely on hazardous organometallic reagents which pose significant safety risks and require stringent handling protocols. The new method utilizes stable phosphorus salts and common amines, drastically reducing operational hazards and simplifying waste management.
Q: What yields can be expected from this synthesis protocol?
A: According to patent data, this optimized protocol consistently achieves product yields in the range of 70% to 80%, which is considered highly efficient for this class of heterocyclic intermediates.
Q: Which solvents are compatible with this reaction system?
A: The process demonstrates flexibility with various polar aprotic solvents, with 1,4-dioxane being the preferred choice for optimal results, though tetrahydrofuran (THF) and acetonitrile are also viable alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminobenzo[d]isothiazole Supplier
The technological advancements detailed in CN102040564B underscore the potential for efficient, safe, and high-quality production of 3-aminobenzo[d]isothiazole derivatives. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of phosphorus residues and other impurities, guaranteeing that every batch is ready for immediate use in sensitive pharmaceutical syntheses. We understand that consistency is the cornerstone of drug development, and our manufacturing protocols are designed to deliver that reliability every time.
We invite global partners to leverage our expertise to optimize their supply chains for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term commercial goals.
