Advanced Anthracene-Based OLED Materials: Scalable Synthesis and Commercial Viability
Advanced Anthracene-Based OLED Materials: Scalable Synthesis and Commercial Viability
The rapid evolution of the organic electronics sector demands materials that combine high performance with manufacturability, a challenge addressed comprehensively in patent CN108586353B. This intellectual property discloses a novel class of organic light-emitting materials based on anthracene and its derivatives, specifically engineered to overcome the limitations of traditional emitters regarding thermal stability and quantum yield. The core innovation lies in the modular structural design, represented by Formula I, which allows for precise tuning of optoelectronic properties through the strategic substitution of aryl and heteroaryl groups at the 9,10-positions of the anthracene core. For R&D directors seeking high-purity OLED material candidates, this patent provides a robust framework for developing next-generation display and lighting technologies that require exceptional efficiency and longevity.
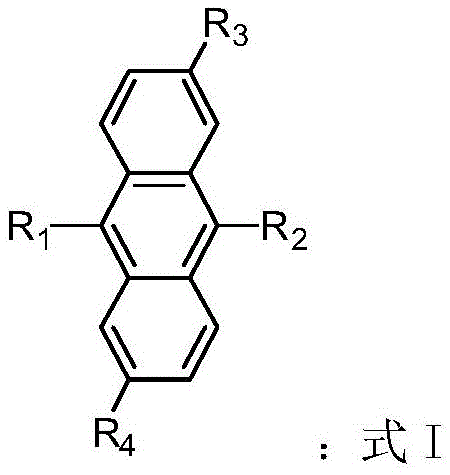
Furthermore, the versatility of this chemical platform extends beyond simple emission; it encompasses potential applications in organic field-effect transistors and fluorescent sensors, making it a valuable asset for diversified electronic chemical portfolios. The patent emphasizes the adjustability of the emission spectrum, a critical parameter for achieving full-color displays without relying on complex filtering systems. By leveraging the inherent stability of the anthracene backbone and enhancing it with bulky substituents like tert-butyl groups, the material achieves a glass transition temperature (Tg) of 163°C and a decomposition temperature (Td) of 240°C, ensuring operational stability under the thermal stress of device fabrication. This technical foundation sets the stage for a detailed analysis of the synthetic methodology and its commercial implications for supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance anthracene derivatives has been plagued by inefficient multi-step sequences that suffer from poor regioselectivity and harsh reaction conditions. Traditional methods often involve direct halogenation at elevated temperatures, which can lead to over-halogenation and the formation of difficult-to-remove isomeric impurities that degrade device performance. Additionally, conventional cross-coupling strategies frequently require expensive ligands or unstable catalysts that complicate the purification process and increase the overall cost of goods sold. For procurement managers, these inefficiencies translate into volatile pricing and extended lead times, as the yield losses in early synthetic stages necessitate larger batch sizes to meet final output targets. The reliance on cryogenic conditions for certain lithiation steps further exacerbates energy costs and safety risks, creating significant bottlenecks in the scale-up phase.
The Novel Approach
In stark contrast, the methodology outlined in CN108586353B introduces a streamlined, three-stage synthetic route that maximizes atom economy and operational simplicity. The process begins with a Friedel-Crafts alkylation to install solubilizing tert-butyl groups, followed by a highly selective room-temperature bromination that avoids the pitfalls of thermal degradation. The cornerstone of this approach is the utilization of sequential Suzuki-Miyaura cross-coupling reactions, which allow for the modular attachment of diverse functional units like tetraphenylethylene and benzimidazole moieties. As illustrated in the synthesis route for TPE-TAPBI, this strategy enables the construction of complex asymmetric molecules with high precision and excellent yields, such as the 90% yield observed in the bromination step and 81% in the final coupling.
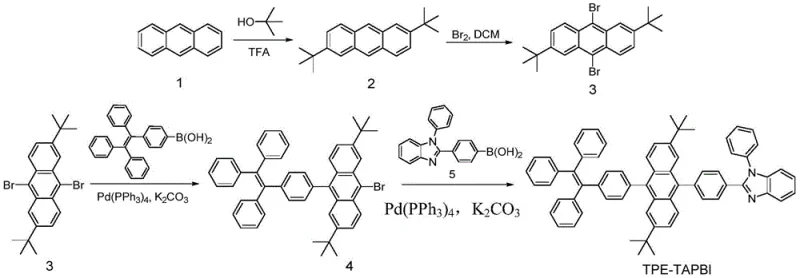
This novel approach not only simplifies the purification workflow but also significantly enhances the reproducibility of the final product, a key metric for quality assurance teams. By employing standard palladium catalysts like tetrakis(triphenylphosphine)palladium and common bases like potassium carbonate, the process utilizes readily available reagents that are compatible with existing industrial infrastructure. The ability to perform these reactions in a toluene-ethanol-water solvent system further reduces the environmental footprint and solvent recovery costs. For supply chain heads, this translates to a more resilient sourcing strategy where raw material availability is less of a constraint, ensuring consistent delivery schedules for high-purity electronic chemical manufacturing.
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling
The efficacy of this synthetic pathway hinges on the mechanistic precision of the palladium-catalyzed Suzuki-Miyaura cross-coupling, which facilitates the formation of carbon-carbon bonds between the dibromo-anthracene intermediate and various aryl boronic acids. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the anthracene core, a step that is facilitated by the electron-rich nature of the substituted anthracene ring. Subsequent transmetallation with the organoboron compound, activated by the carbonate base, transfers the aryl group to the palladium center, setting the stage for the final reductive elimination that releases the coupled product and regenerates the active catalyst. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters, such as the 110°C reflux temperature and the specific molar ratios of 1:2 to 1:3 between the dibromo compound and boronic acid.
Impurity control is another critical aspect managed through the mechanistic understanding of the bromination and coupling steps. The use of liquid bromine in dichloromethane at room temperature ensures mono- or di-bromination without attacking the sensitive alkyl substituents, thereby maintaining the structural integrity required for high thermal stability. Furthermore, the stepwise coupling strategy allows for the isolation of the mono-coupled intermediate, enabling rigorous quality control before the final functionalization. This staged approach minimizes the formation of homocoupling byproducts and unreacted starting materials, which are common contaminants in one-pot symmetric syntheses. For technical directors, this level of control over the impurity profile is essential for meeting the stringent purity specifications demanded by top-tier OLED panel manufacturers.
How to Synthesize TPE-TAPBI Efficiently
The synthesis of TPE-TAPBI serves as a prime example of how to execute this patented methodology to achieve high-quality organic light-emitting materials suitable for commercial application. The process integrates robust chemical transformations with practical workup procedures, ensuring that the final product meets the necessary standards for device integration. Detailed below is the strategic overview of the synthesis, highlighting the critical control points that ensure high yield and purity throughout the production campaign. For a comprehensive breakdown of the standardized operating procedures, please refer to the technical guide provided in the subsequent section.
- Perform Friedel-Crafts alkylation of anthracene with tert-butanol in trifluoroacetic acid to obtain alkyl-substituted anthracene intermediates.
- Conduct selective bromination using liquid bromine in dichloromethane at room temperature to generate dibromo-substituted precursors.
- Execute sequential Suzuki-Miyaura cross-coupling reactions using palladium catalysts to attach functional aryl groups for final OLED material assembly.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from laboratory-scale discovery to industrial production is often hindered by cost and scalability issues, yet the process described in this patent offers distinct commercial advantages that address these pain points directly. By utilizing a convergent synthesis strategy, the manufacturing timeline is significantly compressed, allowing for faster time-to-market for new OLED formulations. The reliance on commodity chemicals such as anthracene, tert-butanol, and standard boronic acids mitigates the risk of supply chain disruptions associated with exotic or proprietary reagents. This accessibility ensures that procurement managers can secure raw materials from multiple qualified vendors, fostering a competitive pricing environment that drives down the overall cost of production without sacrificing quality.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions and the use of ambient temperature for the bromination step drastically reduce energy consumption and equipment requirements. Furthermore, the high yields achieved in the final coupling steps minimize waste generation and maximize the throughput of the reactor vessels, leading to substantial cost savings in terms of raw material utilization. The avoidance of expensive ligand systems in favor of standard triphenylphosphine palladium complexes further lowers the catalyst cost burden, making the process economically viable for large-scale electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the decoupling of intermediate production from final assembly, enabling a more flexible inventory management strategy. Intermediates like the dibromo-anthracene derivative can be stockpiled and then rapidly converted into various final products based on market demand, reducing lead time for high-purity OLED materials. This flexibility is crucial for responding to the dynamic needs of the display industry, where product lifecycles are short and demand fluctuations are common.
- Scalability and Environmental Compliance: The solvent system employed, consisting of toluene, ethanol, and water, is amenable to standard distillation and recycling protocols, aligning with modern environmental regulations and sustainability goals. The process generates minimal hazardous waste compared to traditional methods involving heavy metal oxidants or chlorinated solvents in excess. This environmental compatibility simplifies the permitting process for new production facilities and reduces the long-term liability associated with waste disposal, ensuring a sustainable supply chain for complex polymer additives and electronic materials.
Frequently Asked Questions (FAQ)
To assist stakeholders in evaluating the technical and commercial feasibility of these anthracene derivatives, we have compiled a set of frequently asked questions based on the patent data. These inquiries address common concerns regarding synthesis scalability, material performance, and regulatory compliance, providing clarity for decision-makers. The answers are derived directly from the experimental examples and technical specifications outlined in the intellectual property documentation.
Q: What are the key advantages of the anthracene-based synthesis route described in CN108586353B?
A: The synthesis route offers high yields in critical steps, such as 90% for bromination and 81% for the final coupling, utilizing mild conditions like room temperature bromination which simplifies process control and reduces energy consumption compared to harsh halogenation methods.
Q: How does the structural modification affect the optoelectronic properties?
A: By varying the aryl substituents at the R1 and R2 positions, such as introducing tetraphenylethylene or benzimidazole groups, the absorption and emission spectra can be precisely tuned, allowing for customizable quantum yields and thermal stability suitable for diverse OLED applications.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method relies on robust reactions like Suzuki coupling and standard purification techniques like recrystallization and column chromatography, which are well-established in industrial settings, ensuring scalability from kilogram to ton-level production without compromising purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TPE-TAPBI Supplier
As the global demand for high-efficiency OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative for securing a stable supply of advanced electronic chemicals. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot batches to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of TPE-TAPBI or related anthracene derivatives meets the exacting standards of the optoelectronics industry.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific material requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can reduce your overall manufacturing expenses. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next-generation display projects are built on a foundation of reliable, high-performance organic light-emitting materials.
