Advanced Synthesis of High-Mobility Amine Derivatives for Next-Gen OLED Displays
The landscape of organic semiconductor materials is undergoing a significant transformation driven by the demand for higher efficiency in display technologies. Patent CN110734453A introduces a groundbreaking class of amine derivatives based on a hybrid [1]benzothiophene[3,2-b][1]benzofuran core, specifically engineered to overcome the historical limitations of organic light-emitting diodes (OLEDs). This innovation addresses the critical challenge where traditional materials often sacrifice fluorescence efficiency for charge carrier mobility. By strategically integrating the high hole mobility characteristics of thiophene derivatives with the superior fluorescent properties of furan derivatives, this technology offers a dual-functionality that is rare in the current market. The molecular architecture is designed to minimize the heavy-atom effect typically associated with sulfur-containing compounds, thereby preventing fluorescence quenching while maintaining excellent charge transport pathways. For R&D directors and procurement specialists in the electronic chemical sector, this represents a pivotal shift towards more efficient, stable, and commercially viable OLED materials.
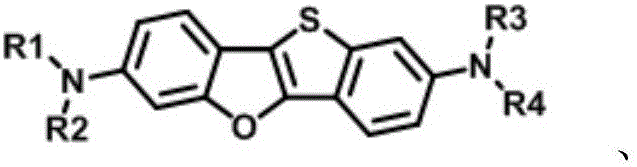
The limitations of conventional methods in OLED material synthesis often stem from the inherent physical properties of the core heterocycles used. Traditional benzothiophene-based semiconductors, such as the classic BTBT core, exhibit exceptional carrier mobility but suffer from significant fluorescence quenching due to the spin-orbit coupling induced by the heavy sulfur atoms. This internal conversion process drastically reduces the luminous efficiency of the device, forcing manufacturers to choose between high mobility or high brightness. Conversely, pure furan-based systems offer better fluorescence but often lack the robust charge transport capabilities required for high-performance devices. The novel approach detailed in this patent circumvents these issues by creating a fused system where the oxygen atom in the furan ring lowers the aromaticity and raises the HOMO energy level, facilitating easier hole injection. Simultaneously, the thiophene component ensures the structural rigidity and pi-stacking necessary for high mobility. This synergistic design allows for the creation of materials that do not force a compromise between electrical performance and optical output.
Mechanistic Insights into Pd-Catalyzed Cyclization and Amination
The synthesis of these advanced amine derivatives relies on a sophisticated sequence of organometallic transformations that ensure high regioselectivity and purity. The process begins with the selective bromination of benzothiophene using N-bromosuccinimide (NBS), followed by oxidation to form a sulfoxide intermediate. This oxidation step is crucial as it activates the ring for subsequent nucleophilic attack by phenol, leading to the formation of a phenoxy-substituted intermediate. The mechanism then proceeds through a reduction step using diisobutylaluminum hydride (DIBAL-H), which removes the oxygen from the sulfoxide to restore the thiophene aromaticity while retaining the phenoxy group. The pivotal moment in the synthesis is the intramolecular cyclization catalyzed by PdCl2(PPh3)2, which closes the furan ring to form the rigid [1]benzothiophene[3,2-b][1]benzofuran core. This step requires precise temperature control and inert atmosphere conditions to prevent catalyst deactivation. Finally, the core is functionalized via dibromination and subsequent Buchwald-Hartwig amination, where bulky aryl amine groups are attached to further enhance hole mobility and thermal stability.
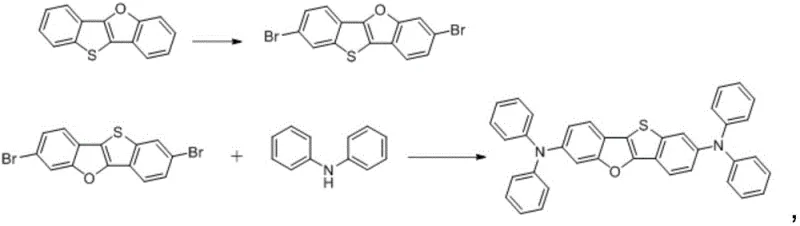
Impurity control in this synthetic pathway is managed through the distinct chemical reactivity of the intermediates. The use of column chromatography at multiple stages, particularly after the bromination and cyclization steps, ensures that regioisomers and over-brominated byproducts are removed before they can propagate through the synthesis. The choice of ligands in the palladium-catalyzed amination step, such as HF4P(t-Bu)3, is critical for suppressing homocoupling side reactions which are common in diarylamine synthesis. Furthermore, the final purification via high vacuum sublimation, as demonstrated in the patent examples, guarantees that the final electronic grade material meets the stringent purity specifications required for OLED deposition processes. This rigorous control over the impurity profile is essential for preventing dark spots and extending the operational lifetime of the final display device.
How to Synthesize [1]Benzothiophene[3,2-b][1]benzofuran Amine Derivatives Efficiently
The preparation of these high-performance materials involves a multi-step protocol that balances yield with purity. The process starts with readily available commodity chemicals like benzothiophene and phenol, making the raw material supply chain robust and cost-effective. The key to efficiency lies in the optimization of the cyclization and amination steps, where catalyst loading and reaction time must be carefully tuned. Detailed standardized synthesis steps for producing these derivatives, including specific molar ratios, solvent choices, and workup procedures, are outlined in the technical guide below.
- Bromination of benzothiophene using N-bromosuccinimide (NBS) followed by oxidation with hydrogen peroxide to form benzothiophene oxide.
- Coupling the oxide with phenol and subsequent reduction using diisobutylaluminum hydride (DIBAL-H) to generate the phenoxy-intermediate.
- Cyclization via Pd-catalyzed reaction to form the [1]benzothiophene[3,2-b][1]benzofuran core, followed by dibromination and final amination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical performance. The reliance on established reaction types, such as electrophilic aromatic substitution and palladium-catalyzed cross-coupling, means that the manufacturing process can be scaled using existing infrastructure without requiring exotic or hazardous reagents that complicate logistics. The starting materials, including benzothiophene and various substituted anilines, are commercially available in bulk quantities, reducing the risk of supply bottlenecks. Moreover, the elimination of the fluorescence quenching problem means that device manufacturers can achieve higher brightness at lower currents, which translates directly into energy savings and reduced thermal management costs in the final consumer electronics. This efficiency gain is a significant value driver for downstream clients looking to optimize their product bills of materials.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of extremely expensive transition metal catalysts in stoichiometric amounts, relying instead on catalytic quantities of palladium which can potentially be recovered. Additionally, the high fluorescence efficiency of the final material reduces the amount of emitter material needed per device to achieve standard brightness levels. By streamlining the purification process through well-defined intermediate isolation, the overall cost of goods sold (COGS) for high-purity electronic chemicals is significantly optimized compared to more complex heteroacene syntheses.
- Enhanced Supply Chain Reliability: The robustness of the intermediates, such as the dibromo-core, allows for stockpiling at various stages of production. This flexibility enables a responsive supply chain capable of meeting fluctuating demand from the display industry. Since the synthesis does not depend on single-source proprietary reagents, the risk of supply disruption is minimized. The ability to produce the material via vacuum evaporation or solution processing (spin coating) also provides manufacturing partners with flexibility in their device fabrication lines, further securing the supply chain against technological shifts.
- Scalability and Environmental Compliance: The reaction conditions described, primarily operating at moderate temperatures and using common organic solvents like toluene and chloroform, are amenable to large-scale batch processing. The waste streams generated are typical of fine chemical synthesis and can be managed through standard treatment protocols, ensuring compliance with increasingly strict environmental regulations. The high atom economy of the cyclization step, where the furan ring is formed intramolecularly, minimizes waste generation. This environmental compatibility is crucial for maintaining long-term operational licenses and meeting the sustainability goals of major electronics brands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this new class of amine derivatives in OLED manufacturing. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and processing capabilities.
Q: How does this new derivative solve the trade-off between hole mobility and fluorescence efficiency?
A: By fusing a thiophene unit (known for high carrier mobility) with a furan unit (known for high fluorescence and lack of heavy-atom quenching), the material achieves both properties simultaneously, unlike traditional BTBT cores which suffer from fluorescence quenching due to sulfur's spin-orbit coupling.
Q: What specific layers in an OLED device can utilize these amine derivatives?
A: These derivatives are specifically designed for the hole functional layer, serving effectively as either a hole injection layer (HIL) or a hole transport layer (HTL) due to their elevated HOMO energy levels and excellent film-forming capabilities.
Q: Are the synthetic intermediates stable enough for large-scale production?
A: Yes, the patent describes robust intermediates such as 3-phenoxybenzothiophene and 2,7-dibromo-[1]benzothiophene[3,2-b][1]benzofuran, which can be isolated and purified via standard column chromatography, ensuring process reliability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiophene-Benzofuran Amine Derivative Supplier
As the global demand for high-resolution and flexible displays continues to surge, the need for advanced hole transport materials has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in heterocyclic chemistry to deliver next-generation OLED materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major panel manufacturers. We maintain stringent purity specifications through our rigorous QC labs, utilizing HPLC and GC-MS to guarantee that every batch of benzothiophene-benzofuran derivatives meets the exacting standards required for vacuum deposition and solution processing.
We invite you to collaborate with us to integrate these high-mobility, high-fluorescence materials into your product roadmap. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes. Contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate the development of superior organic light-emitting diodes.
