Advanced Synthesis of 1,6,7,12-Tetraphenyl Perylene Bisimide Derivatives for Next-Gen Optoelectronics
Advanced Synthesis of 1,6,7,12-Tetraphenyl Perylene Bisimide Derivatives for Next-Gen Optoelectronics
The landscape of organic optoelectronic materials is undergoing a transformative shift, driven by the demand for higher efficiency and tunable spectral properties in devices such as organic light-emitting diodes (OLEDs) and organic photovoltaics (OPVs). Central to this evolution is the strategic modification of perylene bisimide (PBI) cores, a class of compounds renowned for their exceptional thermal stability and electron-transport capabilities. Patent CN101665492A introduces a sophisticated methodology for synthesizing 1,6,7,12-tetraphenyl perylene bisimide derivatives, offering a versatile platform for engineering advanced functional materials. This technology moves beyond traditional dye applications, targeting high-value sectors where precise control over HOMO-LUMO energy levels and morphological stability is paramount. By enabling the attachment of diverse charge-transport functional groups at the sterically hindered bay positions, this innovation provides a reliable OLED material supplier with the tools to create next-generation semiconductors that exhibit significantly red-shifted absorption and luminescence compared to conventional analogues.
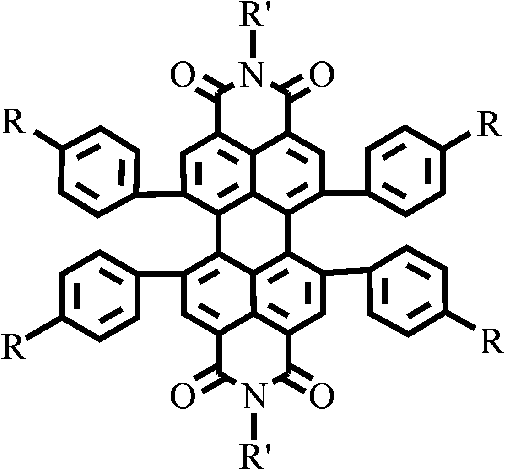
The structural versatility offered by this patent is exemplified by the general formula, which allows for the systematic variation of peripheral substituents to fine-tune material properties. Unlike earlier generations of PBIs where modifications were largely confined to the imide nitrogen atoms—changes that often yielded negligible improvements in photoelectric performance—this approach targets the four bay regions of the perylene nucleus. This strategic positioning allows for the direct introduction of conjugated systems such as carbazoles, naphthylamines, and thiophenes, which act as efficient hole-transport moieties. For research directors focused on purity and杂质 profile control, the ability to modularly assemble these complex architectures from a common halogenated or alkynylated precursor represents a significant advancement in process chemistry, ensuring consistent batch-to-batch reproducibility essential for commercial device fabrication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of perylene bisimides has been constrained by the chemical inertness of the bay positions and the limited impact of imide substitution on electronic properties. Traditional methods often relied on nucleophilic aromatic substitution using phenols to introduce phenoxy groups, a process that frequently suffered from harsh reaction conditions and limited substrate scope. Furthermore, direct arylation at the bay positions was notoriously difficult due to steric congestion, often resulting in low yields and complex mixtures of regioisomers that were challenging to purify. These limitations hindered the development of PBIs with tailored bandgaps, forcing manufacturers to rely on a narrow range of commercially available dyes that lacked the specific spectral characteristics required for emerging applications like near-infrared fluorescent probes or high-efficiency solar cells. The inability to easily install bulky, solubilizing groups also led to materials with poor processability, limiting their utility in solution-based manufacturing techniques such as spin-coating or inkjet printing.
The Novel Approach
The methodology disclosed in CN101665492A circumvents these historical bottlenecks by employing a multi-step strategy that begins with the activation of the 1,6,7,12-tetraphenyl PBI core. By first converting the peripheral phenyl rings into reactive handles such as iodides or terminal acetylenes, the patent establishes a robust foundation for subsequent diversification. This "core-first" activation strategy enables the use of powerful cross-coupling reactions, including Suzuki-Miyaura and Sonogashira couplings, which are highly tolerant of functional groups and offer excellent regioselectivity. Moreover, the utilization of Diels-Alder cycloaddition with substituted cyclopentadienones provides a unique pathway to construct extended conjugated systems that are otherwise inaccessible. This novel approach not only expands the library of accessible derivatives but also ensures cost reduction in electronic chemical manufacturing by streamlining the synthesis of complex architectures from a single, well-defined intermediate, thereby reducing the need for multiple distinct synthetic routes.
Mechanistic Insights into Diels-Alder and Cross-Coupling Functionalization
The heart of this synthetic innovation lies in the precise orchestration of transition metal catalysis and pericyclic reactions to build molecular complexity. A critical step involves the Sonogashira coupling of the tetra-iodinated PBI intermediate with trimethylsilylacetylene, facilitated by a palladium-copper catalytic system. This reaction installs protected alkyne groups which, upon subsequent deprotection with fluoride sources, yield the highly reactive tetra-ethynyl PBI core. This alkyne-functionalized nucleus serves as a potent dienophile in the subsequent Diels-Alder cycloaddition with tetraphenylcyclopentadienone derivatives. The thermal cycloaddition proceeds through a concerted mechanism to form new six-membered rings, effectively fusing additional aromatic systems onto the perylene scaffold. This process not only extends the pi-conjugation length, resulting in the desired red-shift in optical properties, but also introduces significant steric bulk that prevents detrimental aggregation in the solid state.
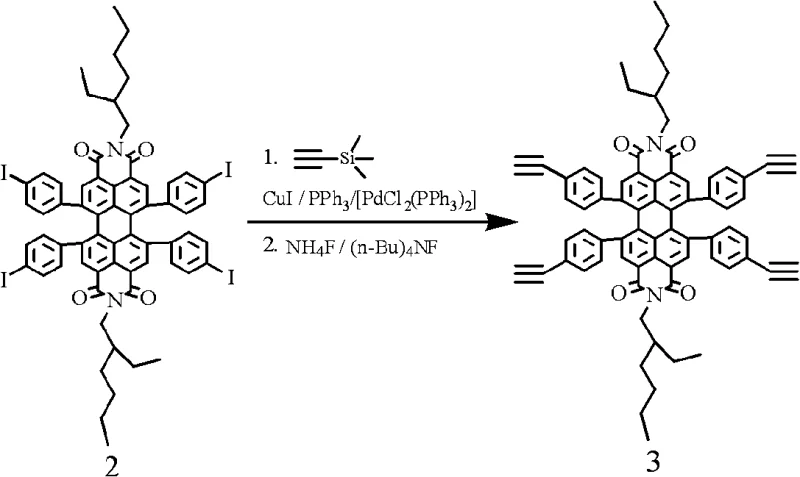
Complementing the Diels-Alder route, the patent also details C-N coupling protocols for attaching electron-rich amines such as 3,6-di-tert-butylcarbazole directly to the halogenated core. This copper-mediated Ullmann-type coupling is particularly valuable for introducing hole-transport capabilities directly into the PBI framework. The mechanistic elegance of these reactions lies in their orthogonality; the conditions used for C-N coupling do not interfere with the imide functionalities, preserving the electron-deficient nature of the PBI core while modifying its periphery. For R&D teams, understanding these mechanistic nuances is crucial for impurity control, as side reactions such as homocoupling of alkynes or over-arylation can be minimized by strictly controlling stoichiometry and temperature profiles. The result is a high-purity OLED material with a defined molecular weight distribution and minimal trace metal contamination, meeting the stringent specifications required for display applications.
How to Synthesize 1,6,7,12-Tetraphenyl Perylene Bisimide Efficiently
The synthesis of these advanced derivatives follows a logical progression from core activation to peripheral functionalization, designed to maximize yield and minimize purification burdens. The process begins with the electrophilic iodination of the tetraphenyl PBI precursor, followed by the installation of alkyne handles or direct coupling with amines. Each step is optimized for scalability, utilizing common solvents and reagents that are readily available in industrial supply chains. The detailed standardized synthesis steps for producing these high-performance materials are outlined below, providing a clear roadmap for process chemists to replicate the patented results.
- Perform bay-region iodination of 1,6,7,12-tetraphenyl perylene bisimide using bis(trifluoroacetoxy)iodobenzene and iodine in carbon tetrachloride to activate the core.
- Execute Sonogashira coupling with trimethylsilylacetylene followed by deprotection to install terminal acetylene groups on the periphery.
- Conduct final functionalization via Diels-Alder cycloaddition with substituted cyclopentadienones or C-N coupling with arylamines to complete the derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic platform offers tangible benefits beyond mere technical performance. The modular nature of the synthesis means that a single batch of the activated PBI core can be diverted into multiple product lines depending on market demand, significantly enhancing supply chain reliability and reducing inventory risks. By consolidating the synthesis of various derivatives onto a common intermediate, manufacturers can achieve substantial cost savings through economies of scale in the production of the precursor. Furthermore, the elimination of exotic reagents in favor of standard palladium and copper catalysts simplifies the sourcing of raw materials, ensuring continuity of supply even during periods of global chemical shortage. This flexibility allows for a responsive manufacturing model that can adapt quickly to the evolving needs of the organic electronics industry.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates the need for multiple, disjointed synthesis pathways for different derivatives, thereby consolidating production overhead. By utilizing robust catalytic cycles that operate under relatively mild conditions, the process reduces energy consumption and minimizes the degradation of expensive starting materials. The high atom economy of the Diels-Alder and coupling reactions ensures that a greater proportion of raw materials ends up in the final product, drastically lowering the cost of goods sold (COGS) per kilogram of active material. Additionally, the improved solubility of the final derivatives simplifies downstream processing and purification, further contributing to overall manufacturing efficiency.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as perylene anhydride, iodine, and standard boronic acids mitigates the risk of supply disruptions associated with specialized reagents. The synthetic protocol is designed to be robust against minor variations in reaction parameters, which translates to higher first-pass yields and more predictable production schedules. This reliability is critical for maintaining the just-in-time delivery models required by major display and solar panel manufacturers. By establishing a stable source of high-quality intermediates, companies can secure long-term contracts with confidence, knowing that the underlying chemistry supports consistent, large-scale output without compromising on quality standards.
- Scalability and Environmental Compliance: The reactions described, including the use of solvents like o-xylene and nitrobenzene, are well-suited for translation from laboratory glassware to industrial reactors. The process avoids the use of highly toxic or unstable reagents that would necessitate expensive containment and waste treatment infrastructure. While solvent recovery is standard practice, the inherent stability of the PBI core reduces the formation of hazardous byproducts, simplifying waste stream management. This alignment with green chemistry principles facilitates regulatory compliance and supports corporate sustainability goals, making the technology attractive for environmentally conscious investors and partners in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized perylene derivatives. The answers are derived directly from the experimental data and theoretical framework presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these aspects is vital for evaluating the feasibility of integrating these materials into existing product lines or R&D pipelines.
Q: What are the primary advantages of bay-region functionalization over imide substitution?
A: Bay-region functionalization significantly alters the electronic conjugation of the perylene core, leading to red-shifted absorption and emission spectra, whereas imide substitution has minimal impact on photoelectric properties.
Q: How does the Diels-Alder approach improve material solubility?
A: The introduction of bulky substituents such as tert-butyl carbazole or extended aromatic systems via Diels-Alder cycloaddition disrupts strong pi-pi stacking interactions, thereby enhancing solubility in common organic solvents for solution processing.
Q: Is this synthesis scalable for commercial production of OLED materials?
A: Yes, the protocol utilizes standard transition metal catalysts and commercially available solvents like nitrobenzene and o-xylene, making it amenable to scale-up from gram to kilogram quantities without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,6,7,12-Tetraphenyl Perylene Bisimide Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage in bringing these complex molecules to market. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 1,6,7,12-tetraphenyl perylene bisimide meets the exacting standards of the optoelectronics industry. Our commitment to quality assurance means that you receive materials with consistent optical and electronic properties, critical for the performance of your final devices.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthetic technology to your specific application needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of a new derivative, our experts are ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can accelerate your product development timeline and enhance your competitive position in the global market.
