Scalable Synthesis of Vilazodone Hydrochloride: A Technical Breakthrough for Commercial API Production
Scalable Synthesis of Vilazodone Hydrochloride: A Technical Breakthrough for Commercial API Production
The pharmaceutical landscape for antidepressant treatments has been significantly advanced by the introduction of Vilazodone Hydrochloride, a dual-function agent acting as a 5-HT1A receptor partial agonist and serotonin reuptake inhibitor. However, the commercial viability of such complex molecules often hinges on the robustness of their synthetic pathways. Patent CN102267985A discloses a transformative preparation method that addresses critical bottlenecks in the existing supply chain for this high-value active pharmaceutical ingredient (API). By shifting away from hazardous Lewis acid catalysis and precious metal coupling, this technology offers a streamlined, cost-effective route suitable for multi-ton production. The core innovation lies in the strategic use of a tosylate intermediate, which facilitates a cleaner convergence of the indole and benzofuran fragments. 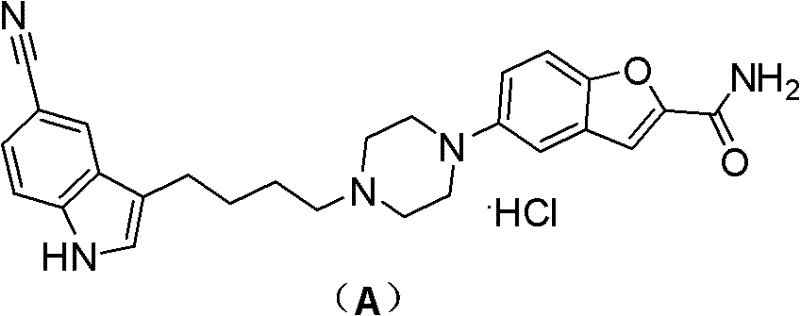
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Vilazodone and its intermediates are plagued by significant technical and economic inefficiencies that hinder large-scale adoption. Traditional routes often rely on Friedel-Crafts acylation using isobutylaluminum dichloride, a reagent that is not only prohibitively expensive but also pyrophoric, requiring specialized inert atmosphere handling and posing severe safety risks in a plant environment. Furthermore, subsequent reduction steps frequently employ diisobutylaluminum hydride (DIBAL-H) or sodium cyanoborohydride, reagents known for their high cost, toxicity, and difficult quenching procedures. Alternative strategies involving palladium-catalyzed cross-coupling introduce another layer of complexity, necessitating rigorous removal of residual heavy metals to meet stringent ICH Q3D guidelines for elemental impurities in drug substances. These legacy processes often suffer from low overall yields, sometimes as low as 27% in critical reduction steps, and invariably require purification via silica gel column chromatography, a technique that is fundamentally incompatible with industrial-scale manufacturing due to solvent intensity and throughput limitations.
The Novel Approach
The methodology outlined in CN102267985A represents a paradigm shift towards greener and more economical chemical manufacturing. Instead of struggling with unreactive alkyl chlorides or hazardous acylations, this novel approach utilizes a highly reactive tosylate intermediate derived from a safe hydride reduction. The process converges the key fragments through a nucleophilic substitution reaction between 4-(5-cyanoindole-3-yl)butyl 4-toluenesulfonate and 5-(1-piperazinyl)benzofuran-2-carboxamide. This coupling proceeds efficiently in common solvents like acetonitrile or butanone under mild basic conditions, eliminating the need for exotic catalysts. 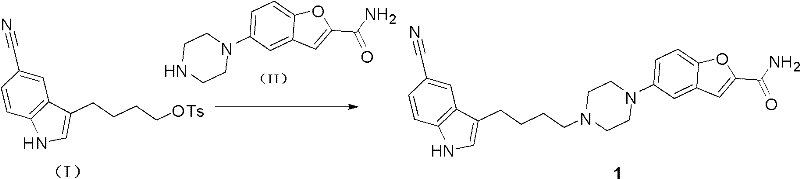
Mechanistic Insights into Safe Hydride Reduction and Tosylate Activation
A critical mechanistic advantage of this patent is the unexpected and efficient transformation of the ketone precursor into the requisite alcohol intermediate. The process utilizes sodium borohydride in aliphatic alcohols to reduce 3-(4-chlorobutyryl)indole-5-carbonitrile. Unlike traditional reductions that might stop at the chlorohydrin or require distinct steps for dehalogenation, this protocol achieves a cascade transformation yielding 3-(4-hydroxybutyl)indole-5-carbonitrile directly. This step is pivotal as it replaces dangerous aluminum hydrides with benign sodium borohydride, drastically simplifying the workup procedure to a standard aqueous quench and extraction. The resulting hydroxyl group is then activated via tosylation using p-toluenesulfonyl chloride and an organic base such as triethylamine or DIPEA. This activation converts a poor leaving group (hydroxyl) into an excellent leaving group (tosylate), thereby enhancing the electrophilicity of the butyl chain for the subsequent piperazine attack. 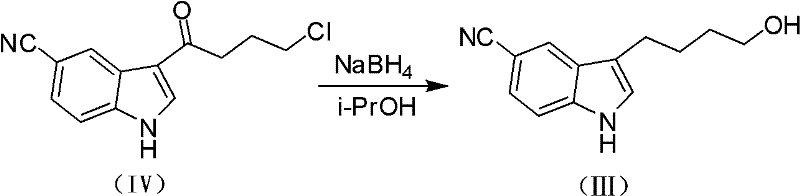
From an impurity control perspective, this route is superior because it avoids the generation of aluminum salts and cyanide byproducts that are notoriously difficult to purge from the final API. The use of tosylate chemistry ensures that the final coupling step proceeds with high specificity, minimizing the formation of bis-alkylated piperazine byproducts that often plague direct alkylation routes. The final salt formation with hydrochloric acid in tetrahydrofuran yields the target hydrochloride salt with exceptional purity, reported in examples to exceed 99.35% by HPLC, demonstrating the robustness of the purification protocol which relies solely on recrystallization rather than chromatography.
How to Synthesize Vilazodone Efficiently
The synthesis of Vilazodone via this patented route involves a logical sequence of reduction, activation, and coupling that is amenable to standard reactor setups found in most fine chemical facilities. The process begins with the reduction of the indole ketone, followed by tosylation to create the electrophilic linker, and concludes with the assembly of the final pharmacophore. Detailed standardized operating procedures for temperature control, stoichiometry, and isolation are critical for maximizing yield and ensuring batch-to-batch consistency.
- Reduce 3-(4-chlorobutyryl)indole-5-carbonitrile using sodium borohydride in alcohol to form the hydroxybutyl intermediate.
- Activate the hydroxyl group by reacting with tosyl chloride and organic base to generate the reactive tosylate ester.
- Couple the tosylate intermediate with 5-(1-piperazinyl)benzofuran-2-carboxamide in acetonitrile or butanone to yield Vilazodone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible improvements in cost structure and supply security. By removing dependency on scarce and volatile reagents like palladium catalysts and pyrophoric aluminum compounds, manufacturers can stabilize their raw material costs and reduce exposure to supply chain disruptions associated with specialty chemicals. The elimination of column chromatography is perhaps the most significant operational benefit, as it allows for continuous processing or large-batch crystallization, significantly increasing throughput capacity without proportional increases in capital expenditure or floor space.
- Cost Reduction in Manufacturing: The replacement of expensive reductants like DIBAL-H and sodium cyanoborohydride with commodity-grade sodium borohydride results in substantial raw material savings. Furthermore, the avoidance of precious metal catalysts eliminates the need for costly metal scavenging resins and extensive analytical testing for residual metals, streamlining the quality control budget. The simplified workup procedures reduce solvent consumption and waste disposal fees, contributing to a lower overall cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as tosyl chloride, sodium borohydride, and common organic bases, are globally available commodities with stable pricing and reliable lead times. This contrasts sharply with legacy routes that depend on custom-synthesized or hazard-classified reagents which often face shipping restrictions and long procurement cycles. By utilizing a chemistry based on stable, shelf-stable inputs, production schedules become more predictable, reducing the risk of delays in delivering high-purity pharmaceutical intermediates to downstream partners.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily managed in standard stainless steel reactors without the need for specialized lining or cryogenic cooling. The absence of heavy metals and cyanide sources simplifies environmental compliance and wastewater treatment, aligning with modern green chemistry principles. This facilitates easier regulatory approval for manufacturing sites and reduces the environmental footprint of the production facility, a key metric for increasingly sustainability-conscious pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of this novel approach for industrial application.
Q: Why is the tosylate route superior to the traditional chlorobutyl coupling method?
A: The tosylate intermediate exhibits higher reactivity in nucleophilic substitution compared to the chlorobutyl analog, allowing for milder reaction conditions and improved yields without requiring expensive phase transfer catalysts or harsh temperatures.
Q: How does this patent address the safety concerns of previous Vilazodone synthesis routes?
A: This method eliminates the use of pyrophoric isobutylaluminum dichloride and toxic sodium cyanoborohydride, replacing them with stable sodium borohydride and standard organic bases, significantly reducing operational risk and waste treatment costs.
Q: Is column chromatography required for purification in this new process?
A: No, the process is designed for industrial scalability, utilizing simple extraction, crystallization, and filtration steps to achieve high purity (over 99%), avoiding the bottleneck of silica gel column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vilazodone Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in your supply chain. Our state-of-the-art facilities are equipped to handle the specific solvent systems and crystallization protocols required for this route, guaranteeing stringent purity specifications and rigorous QC labs that validate every batch against global pharmacopeia standards.
We invite you to leverage our technical expertise to optimize your sourcing strategy for this critical antidepressant intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can enhance your project's profitability and timeline.
