Advanced Synthesis of Multi-substituted Pyrimidinones for HIV-1 Integrase Inhibition
The pharmaceutical landscape for antiretroviral therapy continues to evolve with the discovery of novel chemical entities targeting critical viral enzymes. Patent CN102617487A introduces a significant advancement in this field by disclosing a series of N-((4-(1-methyl-2-substituted benzyl-5-methoxy-6-oxo-1,6-dihydropyrimidine-4-carbonyl)piperazin-1-yl)alkyl)multi-substituted benzamide compounds. These sophisticated molecules, represented generally by Formula (I), exhibit potent inhibitory activity against HIV-1 integrase, a key enzyme responsible for the integration of viral DNA into the host genome. The structural diversity allowed by variables R1 through R5 and the alkyl linker length n provides a robust platform for optimizing pharmacokinetic properties and binding affinity. As a reliable HIV-1 integrase inhibitor supplier, understanding the nuanced chemistry behind these scaffolds is essential for developing next-generation therapeutics that address drug resistance and improve patient outcomes.
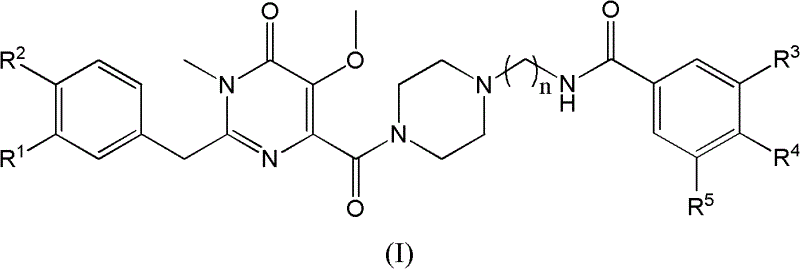
The development of effective HIV-1 integrase inhibitors has historically faced challenges related to metabolic stability and synthetic complexity. Conventional methods for constructing the pyrimidinone core often involve multi-step sequences with low atom economy or require expensive transition metal catalysts that introduce purification burdens. For instance, traditional approaches might rely on condensation reactions that demand strict anhydrous conditions or high-pressure equipment, limiting their feasibility for large-scale production. Furthermore, the introduction of specific substituents on the benzyl and benzamide moieties can be cumbersome, often leading to significant impurity profiles that complicate downstream processing. These limitations underscore the need for innovative synthetic strategies that balance structural complexity with manufacturing practicality, ensuring that promising drug candidates can transition smoothly from the laboratory to commercial supply chains without prohibitive cost increases.
In contrast, the novel approach detailed in the patent offers a streamlined and highly efficient pathway to these complex architectures. The synthesis begins with the straightforward formation of an amidine intermediate from substituted phenylacetonitriles and hydroxylamine hydrochloride, a reaction that proceeds under mild conditions with high conversion. This is followed by a key cyclization step with dimethyl butynedioic acid in xylene, which elegantly constructs the pyrimidine ring system in a single operation. The subsequent methylation and hydrolysis steps are performed using standard industrial reagents like methyl iodide and potassium hydroxide, avoiding exotic chemicals. Finally, the coupling of the pyrimidine acid fragment with the piperazinyl alkyl benzamide moiety utilizes N,N'-carbonyldiimidazole (CDI) as a coupling agent, ensuring high purity and yield. This modular strategy not only simplifies the overall process but also enhances the flexibility to generate diverse analogues for structure-activity relationship studies.
Mechanistically, the construction of the pyrimidinone scaffold relies on the nucleophilic attack of the amidine nitrogen on the electron-deficient alkyne of dimethyl butynedioic acid. This initial addition triggers a cascade of intramolecular cyclizations that establish the six-membered heterocyclic ring with precise regiocontrol. The presence of the methoxy group at the 5-position and the carbonyl at the 6-position is critical for the chelation of magnesium ions within the integrase active site, mimicking the natural substrate interaction. The subsequent N-methylation at the 1-position is achieved through base-mediated alkylation, which locks the conformation of the molecule and enhances its metabolic stability by blocking potential oxidative sites. Impurity control is meticulously managed during the hydrolysis step, where pH adjustment to 2-4 ensures the selective precipitation of the carboxylic acid while leaving sensitive functional groups intact. This level of mechanistic understanding allows for the fine-tuning of reaction parameters to minimize byproduct formation, resulting in a high-purity pharmaceutical intermediate suitable for stringent regulatory standards.
How to Synthesize Multi-substituted Pyrimidinones Efficiently
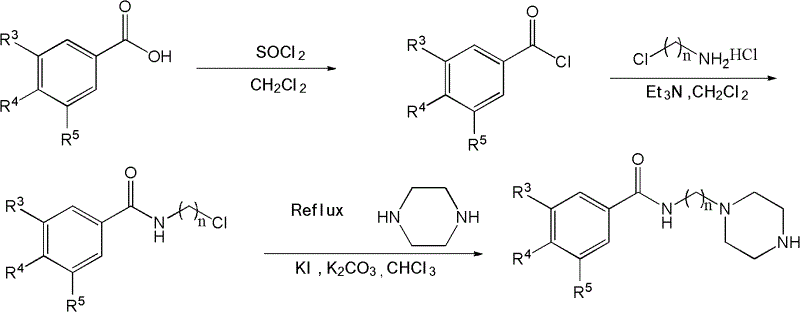
The synthesis of these high-value HIV-1 integrase inhibitor intermediates requires precise control over reaction conditions to ensure optimal yield and purity. The process involves a sequence of well-defined transformations, starting from simple nitrile precursors and culminating in the final amide coupling. Each step, from the initial amidine formation to the final deprotection, has been optimized to utilize common solvents and reagents, facilitating easy scale-up. The use of CDI for the amide bond formation is particularly advantageous as it avoids the formation of difficult-to-remove urea byproducts often associated with other coupling agents. For research and development teams looking to replicate or adapt this chemistry, adhering to the specified molar ratios and temperature ranges is crucial for success. The detailed standardized synthesis steps are outlined in the guide below to assist in your process development efforts.
- React hydroxylamine hydrochloride with substituted phenylacetonitrile to form N'-hydroxyl-2-substituted phenylacetamidine.
- Cyclize the amidine with dimethyl butynedioic acid in xylene to generate the dihydroxy pyrimidine core.
- Methylate the pyrimidine core, hydrolyze the ester, and couple with a piperazinyl alkyl benzamide using CDI.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route described in patent CN102617487A offers substantial advantages in terms of cost reduction and operational reliability. The reliance on commodity chemicals such as phenylacetonitriles, piperazine, and dimethyl acetylenedicarboxylate means that raw material sourcing is not constrained by niche suppliers or geopolitical volatility. This abundance of starting materials translates directly into cost reduction in pharmaceutical intermediate manufacturing, as bulk purchasing power can be leveraged to secure favorable pricing. Furthermore, the elimination of precious metal catalysts removes the need for expensive scavenging steps and rigorous heavy metal testing, thereby simplifying the quality control workflow and reducing the overall cost of goods sold. The process operates at atmospheric pressure and moderate temperatures, which reduces energy consumption and lowers the barrier for implementation in standard multipurpose chemical plants.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts and specialized high-pressure equipment, relying instead on widely available organic reagents and standard glass-lined reactors. This shift significantly lowers the capital expenditure required for production setup and reduces the operational costs associated with catalyst recovery and waste disposal. By utilizing efficient coupling agents like CDI and straightforward workup procedures involving filtration and crystallization, the process minimizes solvent usage and processing time. These factors collectively contribute to a leaner manufacturing model that maximizes resource utilization and drives down the unit cost of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of this synthesis is underpinned by the use of stable, shelf-stable reagents that are readily accessible from multiple global vendors. This diversification of the supply base mitigates the risk of production delays caused by single-source dependencies or logistics disruptions. The mild reaction conditions also enhance safety profiles, reducing the likelihood of unplanned shutdowns due to thermal runaways or hazardous incidents. Consequently, manufacturers can maintain consistent production schedules and meet delivery commitments with greater confidence, ensuring a steady flow of critical intermediates to downstream drug formulation facilities without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to kilogram scales in the patent examples, with clear pathways to tonnage production. The solvents employed, such as dichloromethane, methanol, and xylene, are well-understood in terms of recovery and recycling, allowing for the implementation of closed-loop systems that minimize environmental impact. The absence of toxic heavy metals simplifies wastewater treatment and aligns with increasingly stringent environmental regulations. This commitment to green chemistry principles not only safeguards the environment but also future-proofs the manufacturing process against evolving regulatory landscapes, ensuring long-term viability and sustainability.
Frequently Asked Questions (FAQ)
Understanding the technical and commercial implications of this patented technology is vital for stakeholders involved in the development and procurement of antiretroviral drugs. The following questions address common inquiries regarding the synthesis, application, and supply of these multi-substituted pyrimidinone compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Whether you are evaluating the feasibility of in-house production or seeking a reliable external partner, these insights provide a solid foundation for decision-making and strategic planning.
Q: What is the primary therapeutic application of these multi-substituted pyrimidinone compounds?
A: These compounds are designed as potent inhibitors of HIV-1 integrase, a critical enzyme for viral replication, offering a strategic approach to antiretroviral therapy.
Q: How does this synthesis method improve upon conventional manufacturing processes?
A: The patented route utilizes mild reaction conditions and readily available industrial reagents, significantly simplifying the process compared to methods requiring harsh catalysts or extreme temperatures.
Q: Are the starting materials for this synthesis commercially scalable?
A: Yes, the synthesis relies on common building blocks such as substituted phenylacetonitriles and piperazine derivatives, ensuring robust supply chain continuity and cost efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multi-substituted Pyrimidinones Supplier
The potential of these multi-substituted pyrimidinone compounds to serve as effective HIV-1 integrase inhibitors represents a significant opportunity for advancing antiretroviral therapy. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the management of reactive intermediates and the execution of precise coupling reactions. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the highest standards of quality and consistency required for pharmaceutical applications. Our commitment to excellence ensures that you receive materials that are ready for immediate use in your drug development pipelines.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how we can tailor our services to your needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall project costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments for these valuable intermediates. Together, we can accelerate the delivery of life-saving medications to patients worldwide while maintaining the highest levels of efficiency and quality in the supply chain.
