Revolutionizing Rhodomyrtone Production: A Scalable Route for Global Pharmaceutical Supply Chains
Revolutionizing Rhodomyrtone Production: A Scalable Route for Global Pharmaceutical Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable synthetic routes for bioactive natural products that possess significant therapeutic potential. Patent CN114524795A represents a pivotal breakthrough in the chemical synthesis of Rhodomyrtone, a potent acylphloroglucinol compound isolated from Rhodomyrtus tomentosa. Historically, the supply of this promising antibiotic and antiviral agent has been severely constrained by its low natural abundance and the inefficiency of prior synthetic methodologies. This patent discloses an improved preparation method that effectively optimizes reaction conditions across critical transformations, including Fries rearrangement, Friedel-Crafts acylation, and transition metal-catalyzed cyclization. By addressing the fundamental bottlenecks of previous generations of synthesis, this technology enables the production of high-purity Rhodomyrtone without the need for silica gel column chromatography, achieving a remarkable total yield of 34.6 percent and purity levels exceeding 99.8 percent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
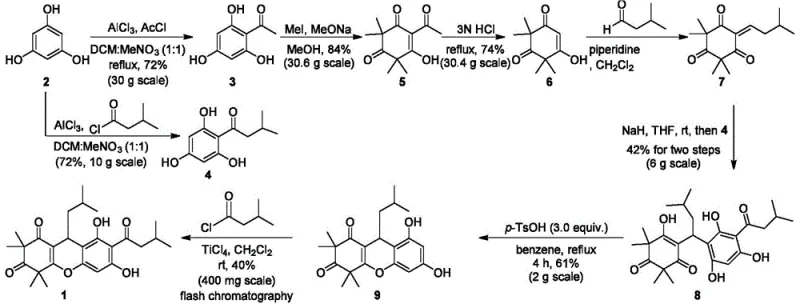
Prior to this innovation, the state-of-the-art synthesis of Rhodomyrtone was plagued by severe inefficiencies that rendered industrial application virtually impossible. The seminal work by Maier et al. in 2013 established the first total synthesis, but it relied on an eight-step linear sequence that delivered a dismal overall yield of merely 0.033 percent. This route necessitated the use of hazardous solvents such as benzene and large quantities of explosive nitromethane, creating significant safety and environmental liabilities for any manufacturing facility. Furthermore, nearly every step required purification via silica gel column chromatography, a technique that is notoriously difficult to scale beyond gram quantities due to excessive solvent consumption and low throughput. Subsequent optimizations by Maier in 2015 and Tan in 2020 failed to resolve these core issues, with yields remaining below 1 percent and reliance on preparative TLC or column chromatography persisting throughout the synthesis.
The Novel Approach
In stark contrast to these legacy methods, the methodology outlined in CN114524795A introduces a streamlined five-step protocol that fundamentally reimagines the process architecture for cost reduction in pharmaceutical intermediate manufacturing. The inventors have strategically replaced chromatographic purification with crystallization and extraction techniques at every stage, ensuring that the process is amenable to multi-kilogram and ton-scale production. By optimizing the solvent systems, such as using a specific 10:1 ratio of dichloroethane to nitromethane in the initial acylation, the reaction efficiency is maximized while minimizing waste. The result is a dramatic escalation in total yield from less than 0.1 percent to 34.6 percent, transforming Rhodomyrtone from a laboratory curiosity into a commercially viable active pharmaceutical ingredient candidate. This shift not only reduces the cost of goods sold but also drastically shortens the production cycle time.
Mechanistic Insights into Nickel-Catalyzed Cyclization and Fries Rearrangement
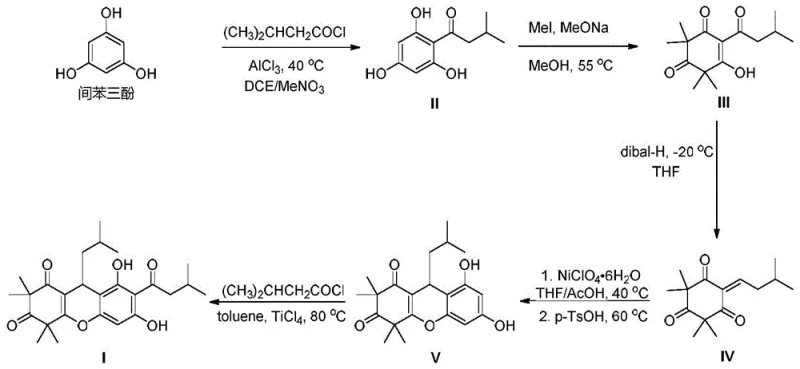
The chemical elegance of this new route lies in its sophisticated handling of the xanthene core formation and the final functionalization. A critical mechanistic advancement is found in Step 4, where a transition metal-catalyzed Michael addition is coupled with an acid-mediated cyclization. The use of a nickel catalyst, specifically nickel perchlorate hexahydrate or Ni(PPh3)4, facilitates the conjugate addition under mild conditions (35-45°C) in a THF/acetic acid mixture. This catalytic system promotes the formation of the carbon-carbon bond necessary for ring closure with high regioselectivity, avoiding the formation of complex byproduct mixtures that typically plague non-catalyzed thermal cyclizations. Following this, the acid-mediated cyclization using p-toluenesulfonic acid at 60-70°C ensures the rapid construction of the tricyclic xanthene scaffold (Intermediate V) with excellent conversion rates.
Furthermore, the final step employs a Lewis acid-mediated Fries rearrangement to install the crucial acyl group at the C-7 position. Utilizing titanium tetrachloride (TiCl4) in a high-boiling solvent like toluene at 80-100°C allows for the migration of the acyl group with precise control over the substitution pattern. This step is particularly sensitive to impurity profiles; however, the patent describes a rigorous workup procedure involving quenching with saturated potassium sodium tartrate and ice water. This specific quenching protocol effectively complexes residual metal species and hydrolyzes reactive intermediates, preventing the carryover of colored impurities or metal residues into the final product. The subsequent crystallization from dichloromethane and petroleum ether ensures that the final Rhodomyrtone meets the stringent purity specifications of greater than 99.8 percent required for clinical applications.
How to Synthesize Rhodomyrtone Efficiently
The synthesis of Rhodomyrtone via this patented route involves a logical sequence of functional group manipulations starting from the inexpensive and readily available feedstock, phloroglucinol. The process begins with a Friedel-Crafts acylation to introduce the isovaleryl side chain, followed by methylation to protect the phenolic hydroxyls and establish the dimedone-like motif. Subsequent reduction of the ketone to an alkene sets the stage for the critical nickel-catalyzed cyclization. The detailed operational parameters, including specific temperature ramps, stoichiometric ratios of Lewis acids, and precise quenching protocols, are essential for reproducing the high yields reported in the patent. For a comprehensive guide on executing these reactions safely and effectively, please refer to the standardized synthesis steps provided below.
- Perform Friedel-Crafts acylation of phloroglucinol with isovaleryl chloride using AlCl3 in DCE/nitromethane to form Intermediate II.
- Execute methylation of Intermediate II with methyl iodide and sodium methoxide, followed by Dibal-H reduction to generate the enone Intermediate IV.
- Conduct nickel-catalyzed Michael addition and acid-mediated cyclization to form the xanthene core (Intermediate V), finalized by TiCl4-mediated Fries rearrangement.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative advantages that directly impact the bottom line and operational resilience. The most significant value driver is the elimination of silica gel column chromatography, which is a major bottleneck in fine chemical manufacturing. By replacing this batch-intensive process with crystallization, the facility can achieve continuous or semi-continuous processing, significantly reducing solvent usage and waste disposal costs. This change alone simplifies the equipment requirements, allowing standard stainless steel reactors to be used without the need for specialized chromatography columns or extensive solvent recovery systems dedicated to flash purification. Consequently, the capital expenditure required to set up production lines is substantially lowered.
- Cost Reduction in Manufacturing: The increase in total yield from 0.033 percent to 34.6 percent represents a thousand-fold improvement in material efficiency. This drastic reduction in raw material consumption per kilogram of final product translates directly into a lower cost of goods sold (COGS). Additionally, the use of common industrial solvents like toluene, dichloroethane, and ethyl acetate, rather than exotic or highly regulated solvents, further stabilizes the supply chain against price volatility. The ability to recover and recycle these solvents through standard distillation adds another layer of economic efficiency, ensuring that the manufacturing process remains profitable even at competitive market prices.
- Enhanced Supply Chain Reliability: The robustness of this synthesis ensures a consistent supply of high-purity Rhodomyrtone, mitigating the risks associated with natural extraction which is subject to seasonal and geographical variations. The synthetic route relies on commodity chemicals such as phloroglucinol and isovaleryl chloride, which are widely available from multiple global suppliers, reducing the risk of single-source dependency. The simplified purification workflow also means that production lead times are significantly shortened, allowing manufacturers to respond more agilely to fluctuations in demand from downstream pharmaceutical partners without maintaining excessive inventory buffers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is far superior to previous iterations. While nitromethane is still utilized in the first step, its volume is minimized through optimized solvent ratios, and the elimination of benzene removes a known carcinogen from the process entirely. The shift to crystallization reduces the volume of hazardous waste generated, simplifying compliance with increasingly strict environmental regulations. The process is inherently safer due to milder reaction conditions in the cyclization steps and the avoidance of pyrophoric reagents in favor of more manageable Lewis acid solutions, facilitating easier regulatory approval for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Rhodomyrtone synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN114524795A, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: How does the new synthesis route improve upon previous methods regarding yield?
A: The patented method achieves a total yield of approximately 34.6%, a massive improvement over the 0.033% yield reported in earlier literature (Maier et al., 2013), primarily by eliminating yield-lossive purification steps.
Q: Is silica gel column chromatography required for purification in this process?
A: No, the process is specifically designed to avoid silica gel column chromatography. All intermediates and the final product are purified via crystallization or simple extraction, making it suitable for industrial scale-up.
Q: What is the purity level of Rhodomyrtone produced by this method?
A: The method consistently produces Rhodomyrtone with a purity exceeding 99.8%, meeting the stringent quality standards required for clinical research and potential API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rhodomyrtone Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of Rhodomyrtone as a next-generation therapeutic agent for treating bacterial and viral infections. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate the innovations described in CN114524795A from the laboratory bench to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Rhodomyrtone meets the highest quality standards required for clinical trials and eventual market launch.
We invite you to collaborate with us to secure a stable and cost-effective supply of this critical intermediate. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized synthesis can reduce your overall project costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us partner with you to accelerate the development of Rhodomyrtone-based therapies and bring this promising medicine to patients worldwide.
