Advanced Synthetic Route for Eribulin Intermediate VII: Technical & Commercial Analysis
Advanced Synthetic Route for Eribulin Intermediate VII: Technical & Commercial Analysis
The pharmaceutical landscape for oncology treatments continues to evolve, with Eribulin mesylate standing as a cornerstone therapy for metastatic breast cancer. The synthesis of this complex macrocyclic ketone presents significant challenges due to its intricate architecture containing 19 chiral centers. Patent CN113372305B, published in late 2022, introduces a robust and highly efficient preparation method for a critical precursor, designated as Intermediate (VII). This technical disclosure represents a substantial leap forward in process chemistry, addressing the historical bottlenecks associated with constructing the polyether backbone of halichondrin analogs. By streamlining the synthetic sequence and optimizing reaction conditions, this methodology offers a viable pathway for industrial-scale production.
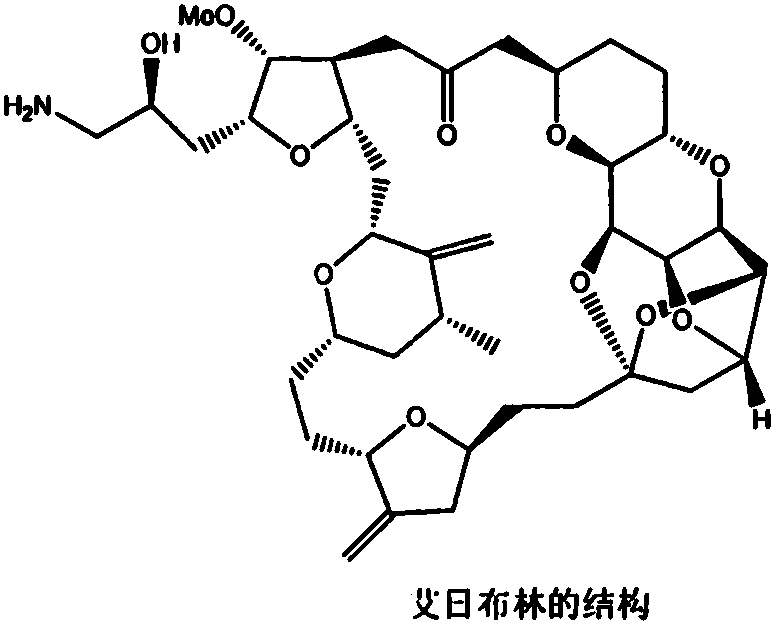
The structural complexity of the final active pharmaceutical ingredient necessitates a modular approach to synthesis, where the reliability of each intermediate is paramount. The patent specifically targets the construction of the right-hand wing fragment, a crucial building block that dictates the overall stereochemical outcome of the final coupling reactions. For R&D directors and process chemists, understanding the nuances of this six-step sequence is essential for evaluating its potential integration into existing manufacturing pipelines. The reported yields are exceptionally high, suggesting a mature and well-optimized protocol that minimizes material loss and waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of eribulin and its analogs has been plagued by lengthy linear sequences that suffer from cumulative yield losses. Traditional routes often rely on harsh reaction conditions, such as strong acids or cryogenic temperatures that are difficult to maintain on a multi-kilogram scale. Furthermore, many legacy methods utilize expensive and sensitive reagents that require stringent handling protocols, increasing the operational risk and cost profile. Purification of intermediates in older pathways frequently involves multiple chromatographic separations, which are not only time-consuming but also generate significant volumes of organic solvent waste. These factors collectively contribute to high production costs and extended lead times, creating supply chain vulnerabilities for pharmaceutical manufacturers relying on these complex intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN113372305B employs a convergent strategy that prioritizes operational simplicity and chemical efficiency. The route utilizes readily available starting materials, such as substituted phosphonium salts and common oxidants, which are commercially accessible in bulk quantities. A key innovation lies in the use of titanium tetrachloride mediated ring-opening reactions, which proceed under relatively mild conditions compared to traditional Lewis acid protocols. This approach not only enhances the safety profile of the manufacturing process but also simplifies the work-up procedures, allowing for easier isolation of products. The consistent high yields reported across all transformation steps indicate a robust process capable of withstanding the rigors of commercial production without compromising quality.
Mechanistic Insights into Titanium-Mediated Ring Opening and Hydroboration
The core of this synthetic strategy revolves around two pivotal transformations: the titanium-mediated ring opening and the stereoselective hydroboration. The ring-opening step utilizes titanium tetrachloride in conjunction with allyltrimethylsilane to cleave the cyclic ether precursor. This reaction is conducted at low temperatures, typically below 5°C, to ensure precise control over the regioselectivity and stereochemistry of the newly formed carbon-carbon bond. The mechanism likely involves the coordination of the titanium species to the ether oxygen, activating the ring towards nucleophilic attack by the allylsilane. This step is critical for establishing the correct carbon skeleton required for the subsequent functionalization.
Following the oxidation and methylenation steps, the final transformation involves a hydroboration-oxidation sequence to install the terminal hydroxyl group with high fidelity. The use of borane complexed with 2-methyl-2-butene serves as a bulky hydroborating agent, which directs the addition of boron to the less hindered position of the terminal alkene. This steric control is vital for achieving the desired regioisomer, preventing the formation of internal alcohols that would act as difficult-to-remove impurities. The subsequent oxidation with sodium perborate ensures a clean conversion to the alcohol without affecting other sensitive functional groups present in the molecule.
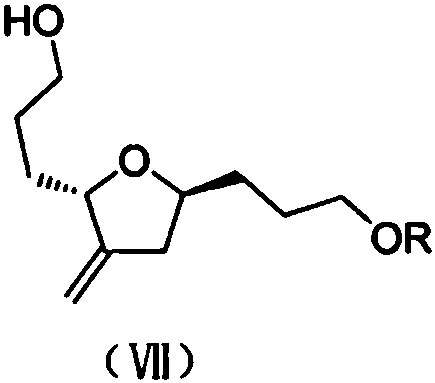
The structural integrity of Intermediate (VII), as depicted above, is maintained throughout the sequence through careful selection of protecting groups. The patent notes that the R substituent can vary widely, including pivaloyl, benzyl, or silyl groups, offering flexibility for downstream coupling strategies. This modularity allows process chemists to tailor the intermediate to specific synthetic needs without altering the core reaction conditions. The ability to tolerate diverse protecting groups underscores the versatility of this route, making it adaptable to various analog programs within the halichondrin family.
How to Synthesize Eribulin Intermediate Efficiently
The execution of this synthesis requires strict adherence to the specified reaction parameters to maximize yield and purity. The process begins with the preparation of the phosphonium ylide, followed by sequential functional group manipulations that build complexity while maintaining stereochemical control. Each step has been optimized to minimize side reactions, such as over-oxidation or elimination, which are common pitfalls in polyether synthesis. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Perform Wittig reaction on Compound I with (2-(R-substituted oxy)ethyl)triphenylphosphonium bromide in THF to yield Compound II.
- Execute catalytic hydrogenation of Compound II using Pd(OH)2/C in isopropanol to obtain Compound III.
- Conduct titanium tetrachloride mediated ring opening of Compound III with allyltrimethylsilane to generate Compound IV.
- Oxidize Compound IV using Dess-Martin periodinane to form ketone Compound V.
- Perform methylenation on Compound V using MePPh3Br and potassium tert-butoxide to yield Compound VI.
- Finalize synthesis via hydroboration-oxidation of Compound VI using borane and 2-methyl-2-butene to produce Intermediate VII.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant reduction of manufacturing complexity, which directly translates to lower operational expenditures. By eliminating the need for exotic catalysts and reducing the number of purification stages, the overall cost of goods sold (COGS) can be drastically reduced. Furthermore, the use of common solvents like tetrahydrofuran and dichloromethane simplifies solvent recovery and recycling processes, contributing to a more sustainable and cost-effective operation.
- Cost Reduction in Manufacturing: The streamlined nature of this six-step sequence eliminates several unit operations found in traditional routes, leading to substantial savings in labor and equipment usage. The high yields achieved at each stage mean that less raw material is required to produce a given amount of intermediate, effectively lowering the material cost per kilogram. Additionally, the avoidance of transition metal catalysts that require expensive removal steps further reduces downstream processing costs. These efficiencies combine to create a highly competitive cost structure for the production of this critical pharmaceutical building block.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and readily available reagents mitigates the risk of supply disruptions caused by raw material shortages. Unlike routes dependent on specialized chiral pool materials that may have limited suppliers, this method utilizes synthetic precursors that can be sourced from multiple vendors globally. This diversification of the supply base ensures continuity of supply, a critical factor for maintaining uninterrupted API production schedules. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, further stabilizing the supply chain.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified work-up procedures make this route highly amenable to scale-up from pilot plant to commercial tonnage. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, reducing the burden on waste treatment facilities. The process avoids the generation of heavy metal waste, simplifying disposal and compliance reporting. These factors facilitate faster regulatory approval for the manufacturing site and reduce the long-term environmental liability associated with chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these details is crucial for assessing the feasibility of integrating this technology into your current manufacturing portfolio.
Q: What are the key advantages of the synthesis route in CN113372305B?
A: The patented route offers mild reaction conditions, easily available raw materials, and simplified purification processes, resulting in consistently high yields across all six steps.
Q: How does this method improve supply chain reliability for eribulin production?
A: By utilizing common reagents like triphenylphosphonium salts and avoiding exotic catalysts, the method reduces dependency on scarce materials, ensuring stable lead times and continuous manufacturing capability.
Q: Is the stereochemistry controlled effectively in this process?
A: Yes, the process employs specific conditions such as low-temperature titanium-mediated reactions and selective hydroboration to maintain the critical chiral integrity required for the final eribulin API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eribulin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the laboratory to the market. We are committed to delivering compounds with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our facility is equipped to handle the specific requirements of complex polyether synthesis, including low-temperature reactors and specialized containment systems for sensitive reagents.
We invite you to collaborate with us to leverage this advanced synthetic technology for your eribulin programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and product quality.
