Revolutionizing Benzimidazole Alkynylamines Production with Metal-Free One-Pot Technology
Revolutionizing Benzimidazole Alkynylamines Production with Metal-Free One-Pot Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective synthetic routes for complex heterocyclic compounds. Patent CN109320503B introduces a groundbreaking metal-free one-pot synthesis method for benzimidazole alkynylamine compounds, addressing critical pain points in traditional manufacturing. This innovative approach utilizes 1-R1-formylmethyl-2-R2-benzimidazole compounds reacting with pyridinium salts and organic bases under a protective gas atmosphere to yield high-purity target structures. The significance of this technology lies in its ability to bypass the need for transition metal catalysts, which are often expensive, toxic, and difficult to remove from the final active pharmaceutical ingredients (APIs). By leveraging this proprietary methodology, manufacturers can achieve superior impurity profiles and streamlined processing, positioning this route as a preferred choice for reliable pharmaceutical intermediates supplier networks globally.
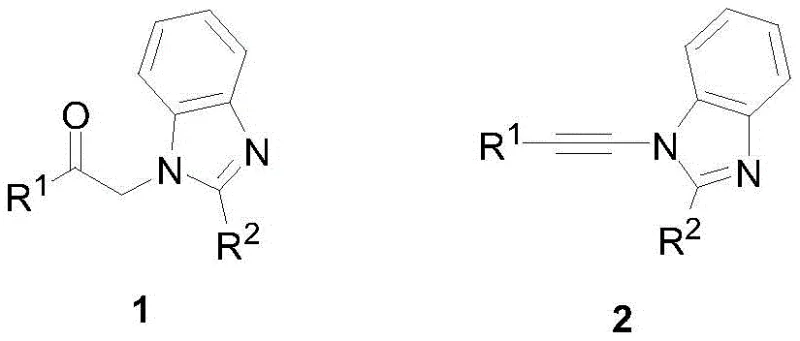
Furthermore, the structural versatility allowed by this synthesis is remarkable, accommodating a wide range of substituents at the R1 and R2 positions, including alkyl, aryl, and heteroaryl groups. This flexibility is crucial for medicinal chemists who require diverse libraries of benzimidazole derivatives for structure-activity relationship (SAR) studies during drug discovery phases. The patent data indicates that the reaction proceeds with exceptional efficiency, often achieving yields not less than 90 percent, which is a substantial improvement over many conventional multi-step protocols. For procurement managers and supply chain heads, this high efficiency translates directly into better raw material utilization and reduced waste generation. The ability to produce high-purity benzimidazole derivatives with such consistency ensures a stable supply chain for downstream drug manufacturing, mitigating risks associated with batch-to-batch variability and regulatory compliance regarding heavy metal limits.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole alkynylamine derivatives has relied heavily on methods involving elimination reactions, alkynyl isomerization, or metal-catalyzed coupling processes. These traditional pathways often suffer from significant drawbacks that hinder their scalability and economic viability in a commercial setting. For instance, metal-catalyzed reactions typically require expensive palladium or copper catalysts, which not only increase the raw material costs but also introduce the risk of heavy metal contamination in the final product. Removing these trace metals to meet stringent pharmaceutical standards often necessitates additional purification steps, such as specialized scavenging resins or repeated recrystallization, which drastically reduces the overall yield and extends the production cycle time. Moreover, many conventional methods require harsh reaction conditions, including high temperatures or strong acidic environments, which can lead to the decomposition of sensitive functional groups and the formation of complex impurity profiles that are difficult to characterize and control.
Another critical limitation of existing technologies is the complexity of the operational procedures. Multi-step syntheses often involve the isolation and purification of unstable intermediates, which increases the handling risks and operational costs. The use of hazardous reagents, such as alkynyl iodonium salts, further complicates the safety profile of the manufacturing process, requiring specialized equipment and strict safety protocols to prevent accidents. These factors collectively contribute to higher production costs and longer lead times, making it challenging for manufacturers to respond quickly to market demands. Additionally, the environmental impact of these traditional methods is considerable, as they often generate significant amounts of chemical waste and consume large volumes of organic solvents. In an era where green chemistry and sustainability are becoming mandatory requirements for regulatory approval and corporate responsibility, these legacy processes are increasingly becoming obsolete and unsustainable for modern cost reduction in fine chemical manufacturing strategies.
The Novel Approach
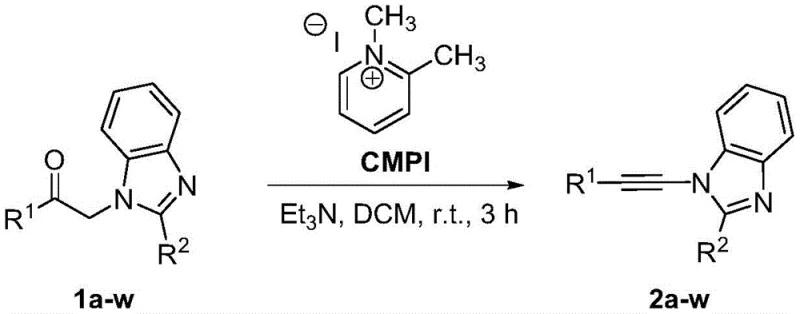
In stark contrast to the cumbersome traditional methods, the novel metal-free one-pot synthesis described in patent CN109320503B offers a streamlined and robust alternative that fundamentally changes the production landscape. This approach utilizes a Mukaiyama-type reagent, specifically pyridinium salts like 2-chloro-1-methylpyridinium iodide or 1,2-dimethylpyridinium iodide, in conjunction with a simple organic base such as triethylamine. The reaction proceeds smoothly in common organic solvents like dichloromethane or dichloroethane at mild temperatures ranging from 10 to 40 degrees Celsius. This mildness is a game-changer for thermal-sensitive substrates, ensuring that the structural integrity of complex molecules is preserved throughout the transformation. The one-pot nature of the reaction means that the conversion from the formylmethyl precursor to the alkynylamine product occurs without the need to isolate intermediates, significantly reducing the operational complexity and the potential for material loss during transfer steps.
The elimination of metal catalysts is perhaps the most significant advantage of this new methodology, as it inherently solves the heavy metal residue problem that plagues many pharmaceutical intermediates. This feature simplifies the downstream purification process, often allowing for straightforward work-up procedures involving aqueous quenching and standard extraction, followed by simple column chromatography or crystallization. The high reaction yields, consistently reported above 90 percent across a broad scope of substrates, demonstrate the robustness and generality of this chemical transformation. For supply chain professionals, this reliability ensures predictable output volumes and minimizes the risk of production failures. Furthermore, the use of readily available and inexpensive reagents contributes to a substantial reduction in the overall cost of goods sold (COGS). By adopting this novel approach, manufacturers can achieve commercial scale-up of complex heterocyclic compounds with greater efficiency, lower environmental impact, and improved safety profiles, making it an ideal candidate for next-generation industrial applications.
Mechanistic Insights into Metal-Free Alkynylation
The mechanistic pathway of this metal-free transformation is a fascinating example of organic activation without the reliance on transition metals. The reaction initiates with the activation of the formylmethyl group on the benzimidazole ring by the pyridinium salt. This activation likely involves the formation of a reactive intermediate, possibly an enol ether or a similar activated species, which is then susceptible to nucleophilic attack or elimination. The organic base, typically triethylamine, plays a dual role in this process: it neutralizes the acid generated during the reaction and facilitates the deprotonation steps necessary for the formation of the alkyne triple bond. The absence of metal coordination complexes means that the reaction kinetics are driven purely by electronic and steric factors of the organic molecules involved, leading to a cleaner reaction profile with fewer side products derived from metal-ligand interactions. This mechanistic simplicity is key to the high selectivity observed, as it minimizes the formation of by-products that often arise from competing metal-catalyzed pathways.
Regarding impurity control, the metal-free nature of this synthesis provides a distinct advantage in maintaining high purity standards. In traditional metal-catalyzed reactions, impurities often include metal complexes, ligand residues, and homocoupling products of the alkyne. In this novel protocol, the primary impurities are likely to be unreacted starting materials or simple hydrolysis products, which are generally easier to separate from the target molecule using standard purification techniques. The mild reaction conditions also prevent thermal degradation, which is a common source of polymeric impurities in high-temperature processes. The patent data supports this by showing consistent high purity across various examples, indicating that the reaction is highly chemoselective. For R&D directors, this means that the process is not only efficient but also robust enough to handle diverse functional groups without compromising the quality of the final API intermediate. The ability to control the impurity profile so effectively reduces the burden on analytical quality control labs and accelerates the regulatory filing process for new drug applications.
How to Synthesize Benzimidazole Alkynylamines Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction environment to maximize the benefits of this metal-free protocol. The general procedure involves dissolving the 1-R1-formylmethyl-2-R2-benzimidazole substrate in a dry organic solvent under an inert atmosphere, such as nitrogen, to prevent moisture interference. The pyridinium salt and organic base are then added in molar ratios that ensure complete conversion, typically around 1:1:1, although slight excesses can be used to drive the reaction to completion. The mixture is stirred at room temperature for a period of 2 to 4 hours, during which the transformation occurs smoothly. Detailed standardized synthesis steps see the guide below.
- React 1-R1-formylmethyl-2-R2-benzimidazole with pyridinium salt in organic solvent under protective gas.
- Add organic base such as triethylamine and maintain temperature between 10-40°C for 2-4 hours.
- Quench reaction, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free synthesis route offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring supply continuity. The elimination of precious metal catalysts removes a significant variable cost from the manufacturing equation, as the prices of metals like palladium and platinum are subject to high market volatility. By replacing these with inexpensive organic salts and amines, the raw material costs are drastically simplified and stabilized. Furthermore, the reduction in purification steps required to remove metal residues leads to significant savings in processing time and solvent consumption. This efficiency gain allows for higher throughput in existing manufacturing facilities without the need for major capital investment in new equipment. The overall effect is a substantial cost savings in the production of these valuable intermediates, making the final drug products more competitive in the global market.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated scavenging processes leads to a direct decrease in material and processing expenses. The one-pot nature reduces labor and utility costs associated with multiple isolation steps. This streamlined approach ensures that the manufacturing budget is utilized more effectively, allowing for better margin management in high-volume production scenarios.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents ensures that the supply chain is not vulnerable to the shortages often seen with specialized metal catalysts. The robustness of the reaction conditions means that production can be maintained consistently across different batches and facilities. This reliability is critical for meeting the strict delivery schedules required by pharmaceutical clients, reducing the risk of supply disruptions that could halt downstream drug manufacturing.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process highly scalable and environmentally friendly. It simplifies waste treatment procedures, as the effluent does not require complex heavy metal removal systems. This alignment with green chemistry principles facilitates regulatory compliance and enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, based on the detailed data provided in the patent documentation. These answers are designed to clarify the operational feasibility and strategic benefits of adopting this technology for your specific production needs. Understanding these details is essential for making informed decisions about process integration and supply chain optimization.
Q: What are the advantages of the metal-free synthesis method for benzimidazole alkynylamines?
A: The method eliminates the need for expensive transition metal catalysts, significantly reducing heavy metal residue concerns and purification costs while maintaining high yields under mild conditions.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction proceeds efficiently at room temperature (10-40°C) in solvents like dichloromethane, using pyridinium salts and organic bases, completing within 2 to 4 hours.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the one-pot nature, mild conditions, and absence of sensitive metal catalysts make it highly scalable and robust for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Alkynylamines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the production of high-value pharmaceutical intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the manufacturing floor. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including strict moisture control and efficient solvent recovery systems. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our team of expert chemists is ready to assist you in optimizing this process for your specific target molecules, ensuring maximum yield and quality.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Let us help you reduce lead time for high-purity active ingredients and secure a stable, cost-effective supply of these critical building blocks for your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
