Scalable Production of (S)-2-Piperidinecarboxylic Acid via Chiral Auxiliary Strategy
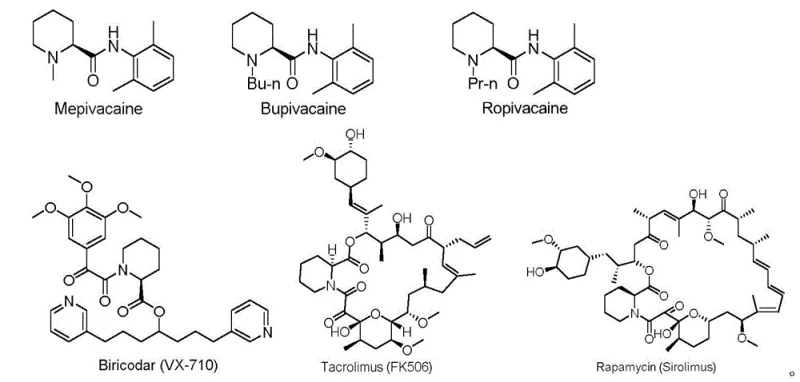
The pharmaceutical industry continuously seeks robust and economically viable pathways for synthesizing chiral building blocks that serve as the backbone for complex active pharmaceutical ingredients (APIs). A significant advancement in this domain is detailed in patent CN111995565A, which discloses a novel preparation method for (S)-2-piperidinecarboxylic acid, also known as (S)-pipecolic acid. This non-proteinogenic amino acid is a critical structural motif found in numerous bioactive compounds, ranging from immunosuppressants like Rapamycin and FK506 to local anesthetics such as Mepivacaine and Bupivacaine. The patented technology addresses the longstanding challenges associated with traditional synthetic routes by introducing a streamlined three-step process that leverages L-camphorsulfonamide as a recoverable chiral auxiliary. This approach not only ensures high stereoselectivity but also circumvents the reliance on precious metal catalysts, offering a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
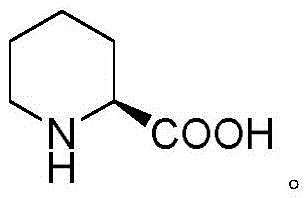
The versatility of (S)-2-piperidinecarboxylic acid extends across multiple therapeutic classes, underpinning its status as a high-demand commodity in fine chemical manufacturing. As illustrated in the structural diversity of downstream drugs, this moiety is integral to the pharmacological activity of agents targeting the immune system and nervous system. The ability to produce this intermediate with consistent optical purity and at a competitive cost is paramount for drug developers. By adopting the methodology outlined in the patent, producers can secure a reliable source of this key building block, mitigating risks associated with supply chain disruptions often caused by the scarcity of specialized catalysts or complex multi-step syntheses required by older technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric synthesis of (S)-2-piperidinecarboxylic acid has relied heavily on transition metal-catalyzed hydrogenation or carbonylation reactions, which present significant economic and environmental hurdles. Literature precedents often utilize expensive chiral ligands coordinated with ruthenium, rhodium, or palladium, such as the (S)-BINAP-RuCl2 system or Rh-DuPHOS complexes. While these methods can achieve high enantiomeric excess, they suffer from substantial drawbacks including the high cost of the catalysts themselves and the stringent requirement for removing trace heavy metals from the final product to meet regulatory safety limits. Furthermore, alternative routes involving Sharpless epoxidation followed by ring-closing metathesis (RCM) involve multiple isolation steps and the use of sensitive reagents that complicate process control. These conventional pathways often result in lower overall yields due to cumulative losses across numerous unit operations, making them less attractive for large-scale industrial production where cost efficiency and throughput are critical metrics.
The Novel Approach
In stark contrast, the method described in patent CN111995565A introduces a paradigm shift by utilizing an organocatalytic strategy centered on a chiral sulfonamide auxiliary. This novel approach condenses L-camphorsulfonamide with a diphenylimine ester under Lewis acid catalysis to establish the initial chiral environment. The subsequent key transformation involves a "one-pot" sequence where asymmetric alkylation, imine hydrolysis, and intramolecular cyclization occur consecutively without isolating intermediates. This telescoping of reactions drastically reduces solvent usage and processing time. By replacing noble metals with abundant aluminum-based Lewis acids and lithium bases, the process achieves a dramatic reduction in raw material costs. Moreover, the chiral auxiliary can be theoretically recovered and recycled, further enhancing the atom economy and sustainability profile of the synthesis, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into L-Camphorsulfonamide Mediated Asymmetric Alkylation
The core of this synthetic strategy lies in the precise stereochemical control exerted by the bulky L-camphorsulfonamide scaffold during the alkylation step. Initially, the condensation of the sulfonamide with the glycine ester derivative forms a chiral N-diphenylmethylene imine. Upon treatment with a strong base such as lithium hexamethyldisilazide (LiHMDS) at low temperatures, a rigid enolate species is generated. The steric bulk of the camphor framework effectively shields one face of the enolate, directing the incoming electrophile, typically a 1,4-dihaloalkane, to attack from the less hindered face. This diastereoselective alkylation sets the absolute configuration at the alpha-carbon with high fidelity. Following the alkylation, acidic workup hydrolyzes the imine bond, releasing the free amine which immediately undergoes an intramolecular nucleophilic substitution with the remaining halide tail to close the six-membered piperidine ring. This cascade mechanism ensures that the chirality established in the first step is faithfully transferred to the final cyclic product.
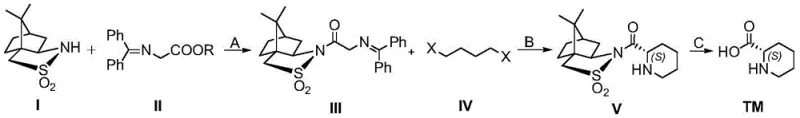
Impurity control in this process is inherently managed through the crystallinity and solubility differences of the intermediates. The use of L-camphorsulfonamide not only directs stereochemistry but also imparts distinct physical properties to the intermediates, facilitating purification via standard column chromatography or crystallization techniques before the final hydrolysis step. The final cleavage of the auxiliary under basic conditions, using reagents like lithium hydroxide, is a clean reaction that generates water-soluble byproducts, allowing for easy separation of the target amino acid. This mechanistic robustness minimizes the formation of racemic byproducts or regioisomers that often plague metal-catalyzed routes. Consequently, the resulting (S)-2-piperidinecarboxylic acid exhibits excellent optical purity, meeting the stringent specifications required for use in the synthesis of potent APIs where even minor impurities can impact biological efficacy or safety profiles.
How to Synthesize (S)-2-Piperidinecarboxylic Acid Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly temperature control during the enolate formation and alkylation phases to maximize diastereoselectivity. The process begins with the activation of the sulfonamide using trimethylaluminum, followed by the addition of the imine ester. The critical "one-pot" cyclization step demands the sequential addition of base and dihaloalkane at cryogenic temperatures before warming to room temperature to facilitate ring closure. Detailed standard operating procedures regarding stoichiometry, solvent selection, and workup protocols are essential for reproducing the high yields reported in the patent literature. For process chemists looking to implement this route, understanding the nuances of the quenching and extraction steps is vital to ensure maximum recovery of the valuable chiral intermediate.
- Condensation of L-camphorsulfonamide with diphenylimine ester using a Lewis acid catalyst like trimethylaluminum to form the chiral imine intermediate.
- One-pot asymmetric alkylation with a dihaloalkane followed by acid hydrolysis and intramolecular cyclization to close the piperidine ring.
- Basic hydrolysis to cleave the chiral auxiliary group and isolate the final (S)-2-piperidinecarboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this L-camphorsulfonamide-based route offers transformative benefits that directly impact the bottom line and operational resilience. Traditional methods relying on precious metals expose manufacturers to volatile market prices for elements like Ruthenium and Rhodium, as well as the logistical complexities of sourcing high-purity chiral ligands. By shifting to a chemistry based on abundant main-group elements like Aluminum and Lithium, companies can stabilize their raw material costs and reduce dependency on single-source suppliers of exotic catalysts. Furthermore, the simplified workflow reduces the number of discrete manufacturing steps, which translates to shorter production cycles and faster time-to-market for downstream drug products. This efficiency is crucial for maintaining competitive advantage in the fast-paced pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and specialized chiral ligands results in substantial cost savings per kilogram of product. Additionally, the "one-pot" nature of the cyclization step significantly lowers solvent consumption and energy requirements associated with multiple heating and cooling cycles. The potential for recovering and reusing the L-camphorsulfonamide auxiliary further enhances the economic viability of the process, driving down the overall cost of goods sold (COGS) for this high-value intermediate.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as L-camphorsulfonamide, alkyl halides, and common bases ensures a stable and diversified supply base. Unlike proprietary catalyst systems that may face supply bottlenecks, the reagents for this synthesis are widely available from multiple global vendors. This redundancy mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for clients relying on a reliable pharmaceutical intermediate supplier for their API synthesis needs.
- Scalability and Environmental Compliance: The avoidance of heavy metals simplifies waste stream management and reduces the environmental footprint of the manufacturing process. Regulatory compliance regarding residual metal limits in APIs is easier to achieve without the need for complex scavenging resins or additional purification stages. The robust nature of the reaction conditions allows for seamless scale-up from laboratory to pilot and commercial production scales, ensuring that quality and yield remain consistent regardless of batch size, thus supporting the commercial scale-up of complex amino acids.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (S)-2-piperidinecarboxylic acid using this patented methodology. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this intermediate into their existing manufacturing portfolios. The answers are derived from the technical specifications and comparative data provided in the patent documentation, highlighting the practical benefits of this synthetic route over legacy technologies.
Q: Why is this chiral auxiliary method preferred over transition metal catalysis?
A: This method eliminates the need for expensive and potentially toxic noble metal catalysts such as Ruthenium, Rhodium, or Palladium, significantly reducing raw material costs and simplifying the removal of heavy metal residues to meet strict pharmaceutical purity standards.
Q: What are the key advantages of the 'one-pot' cyclization step?
A: Combining asymmetric alkylation, imine hydrolysis, and cyclization into a single operational sequence minimizes solvent consumption, reduces labor hours, and prevents the isolation of unstable intermediates, thereby improving overall process efficiency and yield.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the route utilizes readily available starting materials like L-camphorsulfonamide and avoids cryogenic conditions or high-pressure hydrogenation equipment, making it highly amenable to commercial scale-up in standard multipurpose reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-Piperidinecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the route described in CN111995565A and possesses the technical capability to optimize it for industrial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets rigorous quality standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of (S)-2-piperidinecarboxylic acid delivered adheres to the highest levels of chemical and optical purity required for GMP manufacturing.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this cost-effective synthesis technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you streamline your supply chain and accelerate your drug development timeline with our reliable supply of advanced pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
