Advanced Visible Light Photocatalytic Synthesis of Alpha-Bromo-Aromatic Ketones for Commercial Scale-Up
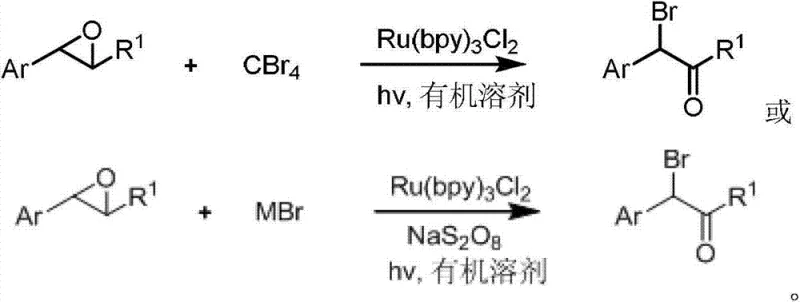
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the production of high-value intermediates. Patent CN103214328A introduces a groundbreaking visible light photocatalytic method for the synthesis of alpha-bromo-aromatic ketone compounds, addressing critical limitations associated with conventional bromination techniques. This innovative approach utilizes aromatic oxirane compounds as starting materials, reacting them with brominating agents such as carbon tetrabromide or metal bromides in the presence of a ruthenium-based photoreaction catalyst. By leveraging clean visible light energy sources, including household LED lamps or even sunlight, the process achieves remarkable reaction efficiency at room temperature without the need for stringent inert gas protection or anhydrous conditions. This technology represents a significant leap forward for manufacturers seeking reliable pharmaceutical intermediate supplier partnerships that prioritize both environmental compliance and operational excellence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-bromo-aromatic ketones has relied heavily on the use of liquid bromine or hydrogen bromide as the primary brominating reagents. These traditional pathways are fraught with substantial operational and safety challenges that hinder large-scale manufacturing efficiency. Liquid bromine is highly toxic, volatile, and corrosive, posing severe risks to equipment integrity and requiring specialized, expensive containment systems to prevent leakage and exposure. Furthermore, the reaction selectivity using these aggressive reagents is notoriously poor, often yielding a mixture of monobrominated and dibrominated products with yields hovering around only 50 percent. The generation of large quantities of acidic waste gas not only necessitates complex scrubbing systems but also creates significant environmental liabilities, making these processes increasingly untenable in modern regulatory landscapes focused on green chemistry and sustainability.
The Novel Approach
In stark contrast, the novel photocatalytic method described in the patent offers a transformative solution by replacing hazardous reagents with low-toxicity alternatives like carbon tetrabromide or metal bromides. This approach operates under mild conditions, specifically at room temperature and atmospheric pressure, eliminating the need for energy-intensive heating or cooling cycles. The use of visible light as the energy driver allows for precise control over the reaction kinetics, resulting in exceptional selectivity where only the desired monobrominated product is formed, effectively suppressing the formation of dibromo byproducts. Yields in this system can exceed 90 percent, representing a dramatic improvement over legacy methods. The simplicity of the operation, which allows for arbitrary feeding sequences and does not require strict moisture exclusion, significantly lowers the barrier to entry for commercial adoption.
Mechanistic Insights into Ru(bpy)3Cl2-Catalyzed Photoredox Bromination
The core of this technological advancement lies in the sophisticated photoredox catalytic cycle mediated by tris(bipyridine)ruthenium(II) chloride, commonly known as Ru(bpy)3Cl2. Upon irradiation with visible light, the ruthenium complex absorbs photons to reach an excited state, possessing sufficient redox potential to initiate single electron transfer (SET) processes with the brominating agent. This activation generates reactive bromine radicals in situ under extremely mild conditions, which then attack the aromatic oxirane substrate. The mechanism involves a regioselective ring-opening of the epoxide followed by radical recombination or oxidation steps that ultimately furnish the alpha-bromo ketone structure. This controlled radical generation is key to the method's success, as it avoids the uncontrolled electrophilic attacks characteristic of molecular bromine, thereby ensuring high chemoselectivity and preventing over-bromination side reactions that plague conventional acid-catalyzed pathways.
From an impurity control perspective, this mechanistic pathway offers distinct advantages for producing high-purity pharmaceutical intermediates. The specificity of the radical interaction with the oxirane ring ensures that functional groups elsewhere on the molecule, such as esters or other aromatic substituents, remain intact. For instance, complex substrates containing sensitive moieties like benzyl ethers or nitro groups can be successfully transformed without degradation, as evidenced by the successful synthesis of diverse derivatives shown in the patent data. The absence of strong acids or harsh oxidants means that acid-sensitive protecting groups do not need to be employed, simplifying the overall synthetic route. This level of precision is critical for R&D directors who require consistent quality and minimal downstream purification burdens when scaling up complex synthetic sequences for active pharmaceutical ingredients.
How to Synthesize Alpha-Bromo-Aromatic Ketones Efficiently
The implementation of this photocatalytic protocol is designed for ease of use and scalability, making it an attractive option for process chemists looking to optimize their manufacturing workflows. The procedure involves a straightforward mixing of the aromatic oxirane substrate with the chosen brominating agent and the ruthenium catalyst in a common organic solvent such as acetonitrile or methanol. Once the mixture is prepared, it is simply exposed to a visible light source, where the reaction proceeds smoothly over a period of 6 to 12 hours. The workup procedure is equally uncomplicated, involving filtration to remove any solid residues followed by concentration and standard silica gel chromatography. For detailed operational parameters and specific stoichiometric ratios required to replicate these results in your facility, please refer to the standardized synthesis guide below.
- Mix aromatic oxirane compounds with a brominating reagent (carbon tetrabromide or metal bromide) and a photoreaction catalyst (Ru(bpy)3Cl2) in an organic solvent.
- Irradiate the reaction mixture with visible light (LED, incandescent, or sunlight) at room temperature for 6 to 12 hours to facilitate the ring-opening and bromination.
- Filter and concentrate the reaction solution, then purify the crude product using silica gel column chromatography with a petroleum ether and ethyl acetate mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this visible light photocatalytic technology presents a compelling value proposition centered on risk mitigation and cost optimization. By eliminating the need for hazardous liquid bromine, companies can drastically reduce the costs associated with specialized storage infrastructure, safety training, and regulatory compliance reporting. The use of solid or less volatile brominating agents simplifies logistics and transportation, reducing the likelihood of supply chain disruptions caused by hazardous material shipping restrictions. Furthermore, the mild reaction conditions translate to lower energy consumption, as there is no need for extensive heating or cryogenic cooling, directly impacting the operational expenditure profile of the manufacturing site. These factors combined create a more resilient and cost-effective supply chain for critical chemical intermediates.
- Cost Reduction in Manufacturing: The transition away from corrosive liquid bromine eliminates the necessity for expensive corrosion-resistant reactors and piping systems, leading to significant capital expenditure savings. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the volume of solvents and adsorbents required for purification, thereby lowering waste disposal costs. The ability to use inexpensive and readily available light sources, including natural sunlight or standard LED arrays, further drives down utility costs compared to traditional thermal processes that require sustained high-energy input.
- Enhanced Supply Chain Reliability: Sourcing safe, solid brominating reagents like carbon tetrabromide or metal bromides is generally more stable and less prone to regulatory bottlenecks than sourcing bulk liquid bromine. The robustness of the reaction conditions, which tolerate ambient moisture and do not require inert atmospheres, reduces the risk of batch failures due to minor environmental fluctuations. This reliability ensures consistent production schedules and shorter lead times for high-purity pharmaceutical intermediates, allowing downstream customers to maintain leaner inventory levels without fear of stockouts.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly stringent global environmental regulations regarding volatile organic compounds and hazardous waste. The absence of acidic off-gases simplifies废气 treatment requirements, making it easier to obtain permits for capacity expansion. The scalability is further supported by the homogeneous nature of the photocatalytic reaction, which can be adapted to continuous flow reactors for even greater throughput, ensuring that the technology can grow alongside market demand without hitting technical ceilings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this process into existing production lines or new product development pipelines.
Q: What are the primary advantages of this photocatalytic method over traditional bromination?
A: Unlike traditional methods using toxic liquid bromine or hydrogen bromide which suffer from corrosion, safety hazards, and poor selectivity (approx. 50% yield), this visible light-driven method operates at room temperature with high selectivity (>90% yield) and generates single brominated products without dibromo byproducts.
Q: Is the ruthenium photocatalyst reusable in this process?
A: Yes, the patent indicates that the amount of photocatalyst required is small and it can be recycled and reused, contributing to the overall cost-effectiveness and environmental sustainability of the process.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, successfully converting various aromatic oxiranes containing alkyl, ketone, ester, and aromatic groups into the corresponding alpha-bromo ketones with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Bromo-Aromatic Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in modern organic synthesis and are fully equipped to leverage this technology for our clients' benefit. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our facilities are outfitted with state-of-the-art photoreactors and rigorous QC labs capable of meeting stringent purity specifications required by the global pharmaceutical industry. We are committed to delivering high-quality intermediates that adhere to the highest standards of safety and efficacy, supporting your drug development timelines with unwavering reliability.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this greener protocol can impact your bottom line. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects. Together, we can build a more sustainable and efficient supply chain for the next generation of life-saving medicines.
