Scalable Synthesis of 2-Fluoro-3-Aminobenzoic Acid for High-Efficiency Broflanilide Production
Scalable Synthesis of 2-Fluoro-3-Aminobenzoic Acid for High-Efficiency Broflanilide Production
The global demand for next-generation insecticides, particularly diamide-class compounds like broflanilide, has placed immense pressure on the supply chains of critical intermediates such as 2-fluoro-3-aminobenzoic acid. Patent CN114920661A introduces a transformative synthetic methodology that addresses the longstanding bottlenecks of cost, safety, and purity associated with traditional manufacturing routes. This innovation leverages a strategic three-step sequence starting from readily available dihalobenzoic acids, effectively bypassing the need for hazardous reagents like sodium cyanide or explosive phosphine azides. By optimizing reaction conditions for nitration, nucleophilic fluorination, and catalytic dehalogenation, the disclosed process offers a robust pathway for producing high-purity intermediates essential for modern agrochemical formulations.
For R&D directors and process chemists, the significance of this patent lies in its ability to streamline the synthesis while maintaining rigorous quality control standards. The method employs a logical progression of functional group transformations that maximize atom economy and minimize waste generation. Specifically, the use of mixed acid systems for nitration followed by selective halogen exchange in polar aprotic solvents ensures high conversion rates. Furthermore, the final reductive step cleverly combines nitro group reduction with the removal of residual halogen substituents, simplifying the downstream purification process. This holistic approach not only enhances the technical feasibility of the synthesis but also aligns perfectly with the green chemistry principles increasingly demanded by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-fluoro-3-aminobenzoic acid has been plagued by significant technical and safety challenges that hinder efficient commercial production. One prominent prior art route, illustrated in 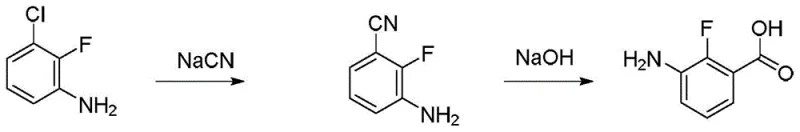 , relies on the nucleophilic substitution of a chloro-aniline derivative with sodium cyanide followed by hydrolysis. This approach is fundamentally flawed for large-scale operations due to the extreme toxicity of cyanide salts, which necessitates specialized containment facilities and complex waste treatment protocols, thereby driving up operational costs exponentially. Additionally, the starting materials for this route are often expensive and less readily available compared to bulk commodity chemicals.
, relies on the nucleophilic substitution of a chloro-aniline derivative with sodium cyanide followed by hydrolysis. This approach is fundamentally flawed for large-scale operations due to the extreme toxicity of cyanide salts, which necessitates specialized containment facilities and complex waste treatment protocols, thereby driving up operational costs exponentially. Additionally, the starting materials for this route are often expensive and less readily available compared to bulk commodity chemicals.
Another conventional strategy involves the oxidation of fluorotoluene derivatives, as depicted in 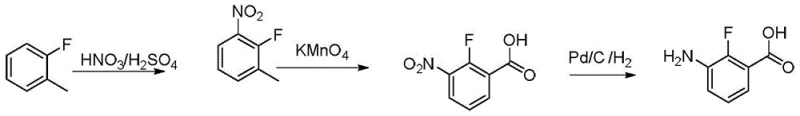 , which utilizes potassium permanganate as a strong oxidizing agent. While chemically feasible, this method suffers from poor selectivity during the initial nitration step, leading to a mixture of ortho and para isomers that are notoriously difficult to separate. The use of stoichiometric amounts of heavy metal oxidants generates massive quantities of manganese dioxide sludge, creating severe environmental disposal issues and violating modern sustainability mandates. Furthermore, alternative routes utilizing diphenylphosphine azide, shown in
, which utilizes potassium permanganate as a strong oxidizing agent. While chemically feasible, this method suffers from poor selectivity during the initial nitration step, leading to a mixture of ortho and para isomers that are notoriously difficult to separate. The use of stoichiometric amounts of heavy metal oxidants generates massive quantities of manganese dioxide sludge, creating severe environmental disposal issues and violating modern sustainability mandates. Furthermore, alternative routes utilizing diphenylphosphine azide, shown in 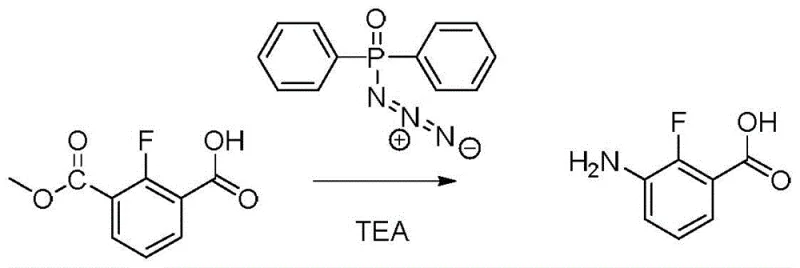 , introduce unacceptable safety risks due to the explosive nature of the azide reagent, making them unsuitable for multi-ton manufacturing environments.
, introduce unacceptable safety risks due to the explosive nature of the azide reagent, making them unsuitable for multi-ton manufacturing environments.
The Novel Approach
In stark contrast to these problematic legacy methods, the novel approach disclosed in the patent utilizes a safer and more economical starting material strategy centered on dihalobenzoic acids. As illustrated in the core reaction scheme 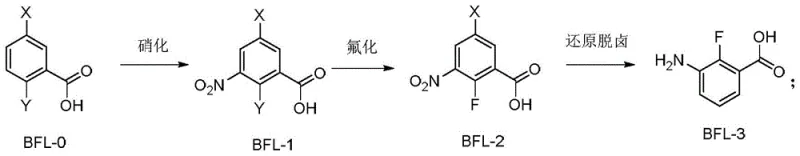 , the process initiates with the nitration of a dihalobenzoic acid (BFL-0) to form a nitro-dihalobenzoic acid intermediate (BFL-1). This is followed by a highly selective nucleophilic aromatic substitution where one halogen atom is replaced by fluorine using inexpensive alkali metal fluorides. The final step involves a catalytic hydrogenation that simultaneously reduces the nitro group to an amine and removes the remaining halogen substituent. This convergent strategy eliminates the need for toxic cyanides and harsh oxidants, replacing them with standard industrial reagents like sulfuric acid, potassium fluoride, and hydrogen gas over palladium catalysts.
, the process initiates with the nitration of a dihalobenzoic acid (BFL-0) to form a nitro-dihalobenzoic acid intermediate (BFL-1). This is followed by a highly selective nucleophilic aromatic substitution where one halogen atom is replaced by fluorine using inexpensive alkali metal fluorides. The final step involves a catalytic hydrogenation that simultaneously reduces the nitro group to an amine and removes the remaining halogen substituent. This convergent strategy eliminates the need for toxic cyanides and harsh oxidants, replacing them with standard industrial reagents like sulfuric acid, potassium fluoride, and hydrogen gas over palladium catalysts.
The superiority of this new route is further highlighted when compared to another recent attempt shown in 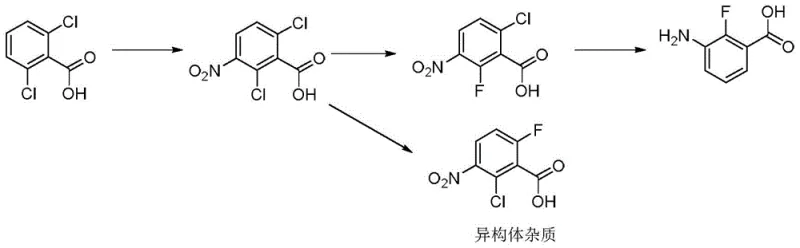 , which suffered from poor regioselectivity during fluorination, resulting in a 30:1 ratio of product to isomer impurities. The new method optimizes the electronic environment of the substrate through careful selection of the leaving group (X and Y can be Cl, Br, or I) and reaction conditions, ensuring that the fluorine atom installs exclusively at the desired position. This dramatic improvement in selectivity translates directly to higher yields and significantly reduced purification burdens, making the process economically viable for the production of high-value agrochemical intermediates on a global scale.
, which suffered from poor regioselectivity during fluorination, resulting in a 30:1 ratio of product to isomer impurities. The new method optimizes the electronic environment of the substrate through careful selection of the leaving group (X and Y can be Cl, Br, or I) and reaction conditions, ensuring that the fluorine atom installs exclusively at the desired position. This dramatic improvement in selectivity translates directly to higher yields and significantly reduced purification burdens, making the process economically viable for the production of high-value agrochemical intermediates on a global scale.
Mechanistic Insights into Halogen Exchange and Reductive Dehalogenation
The success of this synthesis hinges on the precise control of nucleophilic aromatic substitution (SnAr) kinetics during the fluorination step. In the transformation of BFL-1 to BFL-2, the presence of the electron-withdrawing nitro group ortho to the leaving halogen activates the aromatic ring towards nucleophilic attack by fluoride ions. The patent specifies the use of polar aprotic solvents such as dimethyl sulfoxide (DMSO), which are critical for solvating the cation of the fluoride salt (e.g., K+ or Na+) while leaving the fluoride anion "naked" and highly reactive. This solvation effect lowers the activation energy for the displacement of the chlorine or bromine atom, allowing the reaction to proceed efficiently at moderate temperatures around 60°C. The choice of the leaving group is also pivotal; while chlorine is cost-effective, bromine or iodine may offer faster reaction rates due to better leaving group ability, providing process chemists with flexibility to tune the reaction speed based on specific production needs.
Following fluorination, the final transformation involves a sophisticated dual-function catalytic hydrogenation step that converts BFL-2 into the target 2-fluoro-3-aminobenzoic acid (BFL-3). This step utilizes a palladium-on-carbon (Pd/C) or Raney nickel catalyst under hydrogen pressure, typically around 0.2 MPa. Mechanistically, the catalyst facilitates the reduction of the nitro group to an amine via a series of electron and proton transfers. Simultaneously, the catalyst promotes hydrodehalogenation, cleaving the carbon-halogen bond of the remaining substituent (the one not replaced by fluorine) and replacing it with hydrogen. This tandem reaction is crucial because it removes the need for a separate dehalogenation step, streamlining the workflow. The mild conditions (25°C to 45°C) ensure that the sensitive fluoro-substituent remains intact, preventing defluorination which would lead to unwanted byproducts, thus securing the structural integrity of the final molecule.
How to Synthesize 2-Fluoro-3-Aminobenzoic Acid Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process begins with the nitration of the dihalobenzoic acid precursor using a mixed acid system, where temperature control is vital to prevent over-nitration or decomposition. Following isolation of the nitro-intermediate, the fluorination is conducted in DMSO with alkali fluorides, requiring efficient mixing to handle the heterogeneous nature of the salt in the solvent. Finally, the hydrogenation step must be performed in a pressurized reactor with rigorous safety protocols for handling hydrogen gas. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Nitration of dihalobenzoic acid (BFL-0) using a nitric/sulfuric acid system to form the nitro-intermediate (BFL-1).
- Nucleophilic fluorination of BFL-1 using alkali metal fluorides (e.g., KF) in polar aprotic solvents like DMSO to yield BFL-2.
- Catalytic hydrogenation of BFL-2 using Pd/C or Raney Nickel under pressure to effect simultaneous nitro reduction and dehalogenation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic advantages by decoupling production from volatile and restricted chemical markets. Traditional routes often depend on reagents like sodium cyanide, which are subject to stringent regulatory controls and require expensive security measures for transport and storage. By shifting to a pathway based on bulk commodity acids and simple inorganic salts, manufacturers can significantly reduce the total cost of ownership associated with raw material sourcing. The elimination of hazardous waste streams, such as manganese sludge or cyanide effluent, further lowers the environmental compliance costs, allowing for more competitive pricing structures in the final supply of the active ingredient.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of low-cost starting materials such as dichlorobenzoic acid or dibromobenzoic acid, which are produced on a massive industrial scale for other applications. Unlike methods requiring precious metal oxidants or custom-synthesized azides, the reagents here—sulfuric acid, potassium fluoride, and methanol—are ubiquitous and inexpensive. Furthermore, the high selectivity of the fluorination step minimizes the loss of valuable intermediates to isomeric byproducts, effectively increasing the overall mass balance and reducing the cost per kilogram of the final API intermediate. This efficiency gain is compounded by the simplified downstream processing, which requires fewer chromatographic separations or recrystallizations.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by avoiding reliance on single-source or geographically constrained reagents. The flexibility to use chlorine, bromine, or iodine as the leaving groups (X and Y) allows manufacturers to adapt to market fluctuations in halogen prices without altering the core process flow. Additionally, the avoidance of toxic cyanides removes a major logistical bottleneck, as shipping and handling become simpler and less prone to regulatory delays. This robustness ensures a consistent and uninterrupted flow of 2-fluoro-3-aminobenzoic acid to downstream formulators, safeguarding the production schedules of finished insecticide products against raw material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations that are standard in fine chemical plants, such as stirred tank reactors for nitration and fluorination, and fixed-bed or slurry reactors for hydrogenation. The absence of exotic catalysts or extreme high-pressure conditions reduces the capital expenditure required for plant retrofitting. From an environmental standpoint, the switch away from stoichiometric oxidants and toxic nucleophiles drastically reduces the E-factor (mass of waste per mass of product). This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile, a key metric for modern agrochemical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of adopting this technology for industrial production.
Q: How does this new method improve upon previous synthesis routes for 2-fluoro-3-aminobenzoic acid?
A: Unlike prior art that relies on toxic sodium cyanide, explosive azides, or harsh potassium permanganate oxidation, this method utilizes safe, inexpensive dihalobenzoic acids and standard fluorination agents, significantly reducing environmental hazards and operational risks.
Q: What is the key advantage regarding impurity control in this synthesis?
A: The process demonstrates superior regioselectivity during the fluorination step compared to older dichloro-based routes, minimizing the formation of difficult-to-remove isomeric impurities and ensuring high final product purity suitable for pharmaceutical and agrochemical standards.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method avoids exotic reagents and extreme conditions, utilizing common industrial solvents like DMSO and methanol, along with standard catalytic hydrogenation equipment, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-3-Aminobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation crop protection solutions. Our technical team has thoroughly analyzed the synthetic pathway described in CN114920661A and validated its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling nitration and high-pressure hydrogenation safely, and our stringent purity specifications guarantee that every batch meets the rigorous demands of global agrochemical registrars.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for integrating this intermediate into your broader manufacturing portfolio. Let us help you secure a sustainable and cost-effective supply of this vital building block.
