Advanced Copper-Catalyzed Synthesis of Alpha,Alpha-Disubstituted Chiral Amino Acid Esters for Industrial Scale-Up
Introduction to Next-Generation Chiral Amino Acid Synthesis
The synthesis of alpha,alpha-disubstituted chiral amino acids represents a critical frontier in modern medicinal chemistry, particularly for the development of protease-resistant peptides and bioactive small molecules. Patent CN113582865A introduces a groundbreaking preparation method that leverages a sophisticated copper-catalyzed asymmetric propargylic amination strategy. This technology addresses long-standing challenges in constructing quaternary stereocenters adjacent to ester functionalities, a structural motif prevalent in numerous pharmaceutical candidates. By utilizing propargyl ester compounds and readily available arylamines as starting materials, this innovation bypasses the cumbersome multi-step sequences traditionally associated with such transformations. The core breakthrough lies in the direct formation of C-N bonds under mild conditions, utilizing a chiral copper complex that ensures high levels of stereochemical fidelity. For research and development teams seeking reliable pharmaceutical intermediate supplier partnerships, understanding the mechanistic elegance and operational simplicity of this patent is essential for integrating these high-value building blocks into complex drug discovery pipelines.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alpha,alpha-disubstituted chiral amino acids has been fraught with synthetic inefficiencies and logistical bottlenecks. Traditional strategies often rely on the formation of carbon-carbon bonds using precursors that already contain both nitrogen and ester functionalities, a approach that severely limits substrate diversity. As illustrated in prior art, such as the work by the Rong Chunjiang team, synthesizing these motifs frequently necessitates the preparation of specialized aldimine ester precursors followed by palladium and copper co-catalysis. 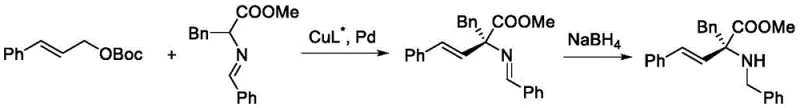 This pathway is inherently flawed for large-scale operations because the resulting products often cannot be isolated directly; they require subsequent reduction steps using reagents like sodium borohydride, which generates substantial amounts of solid waste and complicates purification. Alternatively, other methods employ electrophilic hydroxylamines as nitrogen sources, as seen in Ooi group methodologies.
This pathway is inherently flawed for large-scale operations because the resulting products often cannot be isolated directly; they require subsequent reduction steps using reagents like sodium borohydride, which generates substantial amounts of solid waste and complicates purification. Alternatively, other methods employ electrophilic hydroxylamines as nitrogen sources, as seen in Ooi group methodologies. 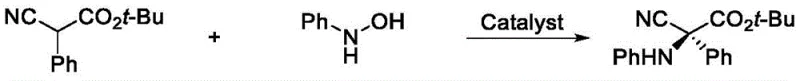 However, these electrophilic reagents are notoriously difficult to synthesize and store, posing significant safety hazards and cost barriers for procurement managers looking for cost reduction in API manufacturing. The reliance on such unstable or hard-to-access reagents restricts the chemical space that can be explored, hindering the rapid iteration required in modern drug design.
However, these electrophilic reagents are notoriously difficult to synthesize and store, posing significant safety hazards and cost barriers for procurement managers looking for cost reduction in API manufacturing. The reliance on such unstable or hard-to-access reagents restricts the chemical space that can be explored, hindering the rapid iteration required in modern drug design.
The Novel Approach
In stark contrast, the methodology disclosed in CN113582865A revolutionizes the field by enabling the direct use of commercial free amines as nucleophilic nitrogen sources. This paradigm shift eliminates the need for pre-functionalized nitrogen precursors, thereby drastically shortening the synthetic route. The reaction proceeds through a direct substitution of a leaving group on the propargyl ester by the arylamine, facilitated by a chiral copper catalyst. This approach not only improves atom economy but also significantly enhances the safety profile of the process by avoiding hazardous electrophilic nitrogen species. The versatility of this system allows for a broad substrate scope, accommodating various substituents on both the alkyne and the amine components without compromising yield or selectivity. For supply chain heads concerned with the commercial scale-up of complex pharmaceutical intermediates, this robustness translates to a more resilient supply chain less dependent on niche, custom-synthesized starting materials. The ability to access diverse chiral amino acid estors directly from commodity chemicals represents a substantial strategic advantage in competitive pharmaceutical markets.
Mechanistic Insights into Copper-Catalyzed Asymmetric Propargylic Amination
The success of this transformation hinges on the precise orchestration of a chiral copper catalytic cycle that effectively differentiates between the enantiotopic faces of the reactive intermediate. The catalytic system typically comprises a copper salt, such as copper(II) acetate, and a specialized chiral ligand, often a bis(oxazoline) or Pybox derivative. 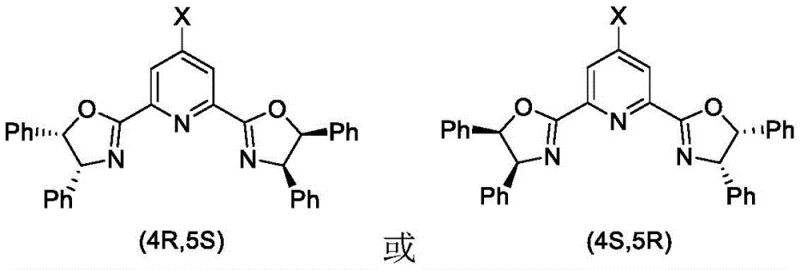 The ligand, specifically the (4R,5S)-L1 variant shown in the patent, coordinates with the copper center to create a rigid chiral environment. This steric confinement is crucial for directing the approach of the arylamine nucleophile to the propargylic cation or pi-allyl copper intermediate generated upon leaving group departure. The electronic properties of the ligand, modulated by substituents like methoxy groups on the pyridine backbone, fine-tune the Lewis acidity of the copper center, optimizing the activation of the propargyl ester. Mechanistically, the reaction likely proceeds via an SN2' or SN2-type displacement where the chirality of the ligand dictates the absolute configuration of the newly formed quaternary center. This high degree of control is evidenced by the consistently high enantiomeric excess values observed across different examples, demonstrating the catalyst's ability to suppress racemic background reactions. Understanding these mechanistic nuances allows process chemists to troubleshoot potential issues related to catalyst loading or solvent effects, ensuring consistent quality in the final product.
The ligand, specifically the (4R,5S)-L1 variant shown in the patent, coordinates with the copper center to create a rigid chiral environment. This steric confinement is crucial for directing the approach of the arylamine nucleophile to the propargylic cation or pi-allyl copper intermediate generated upon leaving group departure. The electronic properties of the ligand, modulated by substituents like methoxy groups on the pyridine backbone, fine-tune the Lewis acidity of the copper center, optimizing the activation of the propargyl ester. Mechanistically, the reaction likely proceeds via an SN2' or SN2-type displacement where the chirality of the ligand dictates the absolute configuration of the newly formed quaternary center. This high degree of control is evidenced by the consistently high enantiomeric excess values observed across different examples, demonstrating the catalyst's ability to suppress racemic background reactions. Understanding these mechanistic nuances allows process chemists to troubleshoot potential issues related to catalyst loading or solvent effects, ensuring consistent quality in the final product.
Furthermore, the choice of solvent and base plays a pivotal role in stabilizing the transition state and facilitating proton transfer steps. The patent highlights the use of a mixed solvent system comprising trifluoroethanol (TFE) and acetonitrile. TFE, being a fluorinated alcohol, possesses unique hydrogen-bonding capabilities that can stabilize charged intermediates and enhance the nucleophilicity of the amine without promoting side reactions. The base, such as quinuclidine, serves to deprotonate the ammonium species formed after nucleophilic attack, regenerating the active catalyst and driving the equilibrium towards product formation. This synergistic effect between the solvent, base, and chiral catalyst creates a highly efficient reaction manifold. Impurity control is inherently built into this mechanism; the high selectivity minimizes the formation of regioisomers or racemates, simplifying downstream purification. For R&D directors focused on purity and impurity profiles, this inherent selectivity reduces the burden on analytical teams and lowers the risk of genotoxic impurities often associated with harsher synthetic conditions.
How to Synthesize Alpha,Alpha-Disubstituted Chiral Amino Acid Ester Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions outlined in the patent to ensure reproducibility and high optical purity. The process begins with the preparation of the active catalyst species under an inert atmosphere to prevent oxidation of the copper center. Substrates are then introduced in a controlled manner to manage the exotherm and maintain the low temperature required for high enantioselectivity. The following guide summarizes the critical operational parameters derived from the exemplary embodiments, providing a roadmap for laboratory validation and pilot scale trials. Detailed standardized synthesis steps follow below.
- Prepare the catalytic system by mixing copper salt (e.g., Cu(OAc)2), chiral Pybox ligand, and organic solvent under nitrogen, then cool to -40°C.
- Dissolve the propargyl ester substrate, arylamine nucleophile, and base (e.g., quinuclidine) in the solvent mixture and add slowly to the catalyst.
- Stir the reaction at low temperature (-40°C to 0°C) for 24-72 hours, then concentrate and purify via silica gel chromatography.
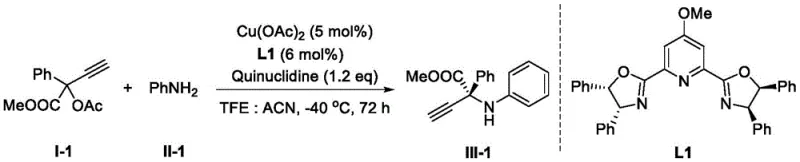
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this copper-catalyzed protocol offers profound commercial benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational agility of chemical manufacturing enterprises. By shifting the synthetic strategy to utilize commodity arylamines and propargyl esters, companies can decouple their production from volatile supply chains associated with specialized nitrogen precursors. This strategic pivot enhances supply chain reliability, ensuring that production schedules are not disrupted by the unavailability of niche reagents. Moreover, the elimination of additional reduction steps and the associated workup procedures leads to a drastically simplified workflow. This streamlining reduces the consumption of solvents, reagents, and energy, contributing to substantial cost savings in pharmaceutical intermediate manufacturing. The high yields and selectivity reported in the patent further imply that less raw material is wasted, improving the overall mass balance of the process. For procurement managers, this translates to a more predictable cost structure and the ability to negotiate better terms for bulk raw materials that are widely available in the global market.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive copper salts compared to precious metals like palladium or rhodium often found in alternative methodologies. Additionally, the avoidance of stoichiometric reducing agents and the reduction in purification steps significantly lower the operational expenditure per kilogram of product. The high atom economy ensures that a greater proportion of the input mass ends up in the final product, minimizing waste disposal costs which are increasingly stringent in modern regulatory environments. These factors collectively drive down the cost of goods sold (COGS), allowing for more competitive pricing in the global marketplace without sacrificing margin.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for supply chain heads, and this method excels by relying on feedstocks that are produced at massive scales for other industries. Arylamines and simple alkynes are foundational chemicals with robust, multi-vendor supply networks, mitigating the risk of single-source dependency. The robustness of the catalytic system also means that the process is less sensitive to minor variations in raw material quality, further insulating production from supply fluctuations. This resilience is vital for maintaining continuous manufacturing operations, especially in the face of global logistical challenges. By securing a synthesis route based on stable, commoditized inputs, organizations can guarantee longer-term supply continuity for their key therapeutic programs.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this protocol aligns well with green chemistry principles. The reduction in solid waste generation, particularly from avoiding borohydride reductions, simplifies effluent treatment and lowers the environmental footprint of the facility. The reaction conditions, while requiring low temperatures, do not demand extreme pressures or hazardous reagents that would complicate scale-up engineering. The use of standard organic solvents facilitates easy recovery and recycling, further enhancing the sustainability profile. This ease of scale-up ensures that the transition from gram-scale laboratory synthesis to ton-scale commercial production can be achieved with minimal process re-engineering, accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity for stakeholders evaluating this synthesis route for their specific applications. Understanding these details is crucial for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this copper-catalyzed method over traditional palladium-catalyzed routes?
A: This method utilizes commercially available arylamines directly as nitrogen sources, eliminating the need for complex pre-synthesis of aldimine esters or electrophilic hydroxylamines required in older protocols. Furthermore, it avoids additional reduction steps, significantly simplifying the workflow and reducing solid waste generation.
Q: What level of enantioselectivity can be achieved with this protocol?
A: The protocol demonstrates excellent stereocontrol, achieving enantiomeric excess (ee) values typically ranging from 90% to 95% across various substrates, including those with electron-donating or electron-withdrawing groups on the aromatic ring.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for industrial feasibility. It employs robust copper catalysts and standard organic solvents like trifluoroethanol and acetonitrile, operating at manageable low temperatures without requiring exotic reagents, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Alpha-Disubstituted Chiral Amino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed asymmetric synthesis described in CN113582865A for advancing your pharmaceutical pipeline. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral amino acid ester delivered meets the highest industry standards for enantiomeric excess and chemical purity. We understand that consistency is key in drug development, and our robust quality management systems are designed to provide the reliability you need to move forward with confidence.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this technology offers for your specific molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in delivering high-quality chemical solutions. Let us help you accelerate your development timeline with our proven expertise in complex chiral synthesis.
