Advanced Manganese Catalysis for Efficient Cyclooctatriene Indole Derivative Production
Advanced Manganese Catalysis for Efficient Cyclooctatriene Indole Derivative Production
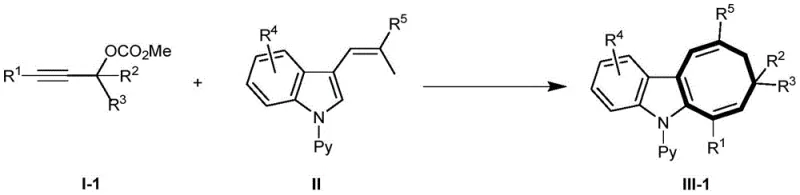
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to construct complex heterocyclic scaffolds. A groundbreaking development in this arena is detailed in patent CN109761956B, which discloses a novel method for synthesizing cyclooctatriene indole derivatives utilizing manganese catalysis. This technology represents a significant leap forward in C-H bond activation chemistry, enabling the construction of eight-membered fused ring systems through a tandem sequence of allenylation and intramolecular 1,5-hydrogen migration. By leveraging cheap and commercially available Mn(CO)5Br as the catalyst, this method overcomes the historical limitations associated with synthesizing these valuable skeletons, which traditionally required multi-step sequences and highly reactive, unstable substrates. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: high yields under mild conditions with a drastically reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of eight-membered carbocyclic rings fused with indole moieties has been a formidable challenge in organic synthesis. Traditional approaches often relied on the use of highly functionalized bis-olefins or required harsh reaction conditions that compromised atom economy and operational safety. Many existing protocols necessitated the use of expensive noble metal catalysts, such as palladium or rhodium complexes, which not only inflated the raw material costs but also introduced significant downstream processing burdens regarding heavy metal residue removal. Furthermore, the starting materials for these conventional routes were frequently unstable or difficult to prepare, requiring multi-step syntheses that lowered the overall throughput and increased waste generation. These factors combined to create a bottleneck in the supply chain for high-purity intermediates needed for drug discovery and development.
The Novel Approach
In stark contrast, the method described in patent CN109761956B utilizes a streamlined one-pot multistep tandem reaction that transforms stable and readily available propargyl alcohol carbonates and 3-alkenyl-substituted-N-pyridylindoles directly into the target cyclooctatriene indole framework. This approach eliminates the need for pre-functionalized, unstable intermediates, thereby simplifying the entire synthetic workflow. The use of manganese, an earth-abundant base metal, replaces costly noble metals, aligning perfectly with green chemistry principles and cost reduction strategies. The reaction proceeds with high selectivity and yield, constructing the complex eight-membered ring system in a single operational step. This efficiency translates directly into reduced manufacturing lead times and lower capital expenditure for production facilities.
Mechanistic Insights into Mn(CO)5Br-Catalyzed Cyclization
The core of this technological breakthrough lies in the unique ability of the Mn(CO)5Br catalyst to facilitate a cascade of transformations involving C-H bond activation, allenylation, and subsequent cyclization. The mechanism initiates with the activation of the propargyl carbonate, generating a reactive allenyl-manganese species in situ. This electrophilic intermediate then undergoes a regioselective addition to the electron-rich C-H bond of the indole ring, specifically at the position adjacent to the nitrogen atom. Following this C-C bond formation, the system undergoes a critical intramolecular 1,5-hydrogen migration, which drives the thermodynamic equilibrium towards the formation of the stable eight-membered cyclooctatriene ring. This tandem sequence is highly efficient because it avoids the isolation of reactive intermediates, minimizing side reactions and decomposition pathways that often plague stepwise syntheses.
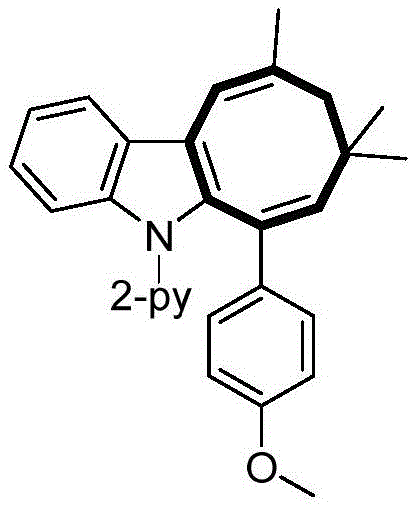
From an impurity control perspective, this mechanism offers distinct advantages. The high selectivity of the manganese catalyst ensures that the reaction proceeds primarily through the desired pathway, significantly reducing the formation of regioisomers or polymeric byproducts. The mild reaction conditions, typically ranging from 90°C to 100°C, further suppress thermal degradation of sensitive functional groups present on the substrate. For instance, substrates containing ester, ether, or halogen substituents remain intact throughout the process, as evidenced by the broad substrate scope demonstrated in the patent examples. This robustness allows chemists to introduce diverse functional handles early in the synthesis, facilitating rapid structure-activity relationship (SAR) studies without the need for extensive protecting group strategies.
How to Synthesize Cyclooctatriene Indole Derivatives Efficiently
Implementing this manganese-catalyzed protocol in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The process involves mixing the indole substrate and the propargyl carbonate precursor in a suitable organic solvent, such as 1,4-dioxane or toluene, under an inert nitrogen atmosphere. The addition of a base, preferably a mixture of sodium acetate and dicyclohexylamine, is crucial for neutralizing acidic byproducts and facilitating the catalytic cycle. Once the catalyst Mn(CO)5Br is introduced, the reaction mixture is heated in a sealed pressure tube to maintain the necessary temperature and pressure conditions. Detailed standardized synthesis steps see the guide below.
- Mix propargyl alcohol carbonate compounds with 3-alkenyl-substituted-N-pyridylindoles and base in an organic solvent.
- Add Mn(CO)5Br catalyst (5-15 mol%) under nitrogen protection in a closed pressure tube.
- Heat the mixture to 90-100°C for approximately 18 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this manganese-catalyzed technology offers transformative benefits that extend beyond simple yield improvements. The shift from noble metals to manganese represents a fundamental change in the cost structure of producing these complex intermediates. Manganese catalysts are orders of magnitude cheaper than their palladium or rhodium counterparts, and their abundance ensures long-term supply stability unaffected by geopolitical fluctuations in precious metal markets. Additionally, the elimination of expensive ligands and the simplification of the purification process—due to the absence of heavy metal residues—drastically reduces the cost of goods sold (COGS). This economic efficiency makes the technology particularly attractive for the commercial scale-up of complex pharmaceutical intermediates where margin compression is a constant concern.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive Mn(CO)5Br leads to substantial raw material savings. Moreover, the one-pot nature of the reaction reduces solvent consumption, energy usage, and labor costs associated with multiple isolation steps. The simplified workup procedure, which often requires only standard silica gel chromatography, further lowers operational expenses by minimizing waste disposal costs and equipment downtime. These cumulative efficiencies result in a significantly more competitive pricing structure for the final API intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically propargyl alcohol carbonates and substituted indoles, are commercially available and stable, unlike the sensitive bis-olefins required by older methods. This stability simplifies inventory management and reduces the risk of supply disruptions caused by material degradation during storage or transport. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, ensuring consistent batch-to-batch reproducibility which is critical for maintaining reliable supply chains.
- Scalability and Environmental Compliance: The mild operating temperatures and the use of common organic solvents make this process highly amenable to scale-up from gram to ton quantities without requiring specialized high-pressure or cryogenic equipment. From an environmental standpoint, the use of a non-toxic base metal catalyst and the high atom economy of the tandem reaction align with increasingly stringent global regulations on chemical manufacturing. This compliance reduces the regulatory burden and facilitates faster approval processes for new drug applications that utilize these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese-catalyzed synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of using Mn(CO)5Br over noble metal catalysts?
A: Mn(CO)5Br is significantly cheaper and more abundant than traditional noble metals like palladium or rhodium. Furthermore, it operates under milder conditions and avoids the complex heavy metal removal steps often required in pharmaceutical manufacturing, streamlining the purification process.
Q: What is the substrate scope for this manganese-catalyzed reaction?
A: The reaction demonstrates excellent versatility, accommodating various substituents on both the indole and alkyne components. It tolerates electron-donating and electron-withdrawing groups, including halogens, esters, and ethers, allowing for the synthesis of a diverse library of functionalized intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its one-pot nature, use of stable starting materials, and operation at moderate temperatures (90-100°C). The absence of sensitive reagents and the robustness of the manganese catalyst make it ideal for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclooctatriene Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this manganese-catalyzed technology to revolutionize the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of cyclooctatriene indole derivatives meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain with superior chemistry.
