Optimizing Lorcaserin Hydrochloride Production: A Technical Analysis of Patent CN103333111A for Global Supply Chains
Optimizing Lorcaserin Hydrochloride Production: A Technical Analysis of Patent CN103333111A for Global Supply Chains
The pharmaceutical landscape for anti-obesity therapeutics has been significantly reshaped by the introduction of selective serotonin receptor agonists, specifically Lorcaserin hydrochloride. As global demand for effective weight management solutions escalates, the efficiency of synthesizing key pharmaceutical intermediates becomes a critical bottleneck for supply chains. Patent CN103333111A, filed in 2013, introduces a transformative preparation method for (R)-8-chloro-1-methyl-2,3,4,5-tetrahydro-1H-3-benzazepine hydrochloride, the core active ingredient. This technical insight report analyzes the patent's novel synthetic pathway, which leverages p-chlorophenyl acetate as a foundational building block. By shifting away from expensive and unconventional starting materials utilized in legacy processes, this method offers a compelling value proposition for R&D directors seeking robust impurity profiles and procurement managers aiming for substantial cost optimization. The following analysis dissects the chemical ingenuity behind this route, demonstrating how it achieves high purity through a streamlined sequence of aminolysis, halogenation, reduction, and cyclization, ultimately delivering a scalable solution for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
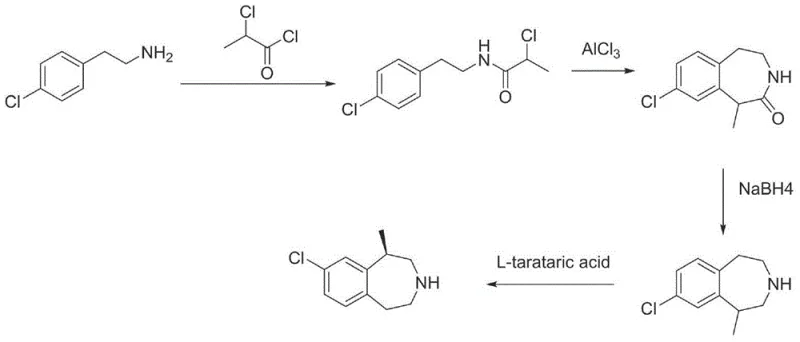
Historically, the synthesis of Lorcaserin intermediates has been plagued by significant economic and operational inefficiencies that hinder large-scale adoption. Early reported routes, such as Circuit One described in WO2005003096, rely on 4-chlorophenethylamine as the primary starting material. This compound is not only a non-conventional industrial chemical with limited market availability but also commands a prohibitively high price point, drastically inflating the cost of goods sold (COGS). Furthermore, alternative pathways like Circuit Two (US20090143576) necessitate the use of phosphorus tribromide for bromination steps. This reagent presents severe safety hazards, generates substantial amounts of corrosive hydrogen bromide waste, and complicates post-reaction workups due to the formation of polysubstituted byproducts. Additionally, Circuit Three attempts to utilize 4-chlorophenylacetic acid but often relies on expensive condensation agents or moisture-sensitive boronic acid catalysts that require prolonged water removal periods. These legacy methods collectively result in extended production cycles, complex purification protocols, and environmental compliance challenges that are untenable for modern, high-volume manufacturing facilities.
The Novel Approach
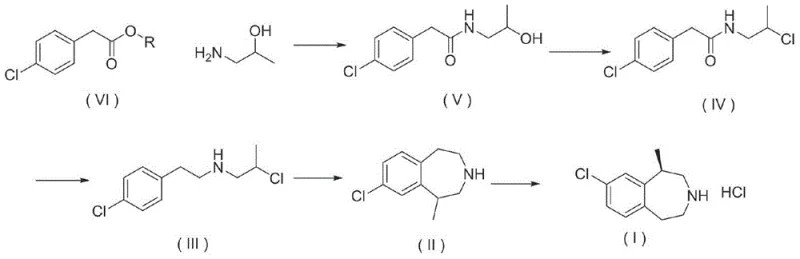
In stark contrast to these cumbersome legacy pathways, the methodology disclosed in CN103333111A establishes a streamlined, economically viable synthetic architecture centered around the readily available and inexpensive p-chlorophenyl acetate. This novel approach initiates with a direct aminolysis reaction with isopropanolamine, bypassing the need for costly activated acids or unstable amines. The subsequent transformation involves a controlled halogenation using versatile reagents like thionyl chloride or phosphorus pentachloride, followed by a highly efficient reduction step utilizing sodium borohydride. The culmination of the synthesis is a Lewis acid-catalyzed Friedel-Crafts alkylation that elegantly closes the seven-membered benzazepine ring. This route is characterized by its operational simplicity, utilizing common protic and aprotic solvents such as methanol, toluene, and dichlorobenzene. By eliminating the need for exotic catalysts and hazardous brominating agents, the process not only reduces the environmental footprint but also enables continuous operation capabilities. This structural shift in the synthetic strategy directly addresses the pain points of prior art, offering a pathway that is both chemically robust and commercially superior for industrial application.
Mechanistic Insights into Friedel-Crafts Cyclization and Chiral Resolution
The cornerstone of this synthetic success lies in the precise execution of the intramolecular Friedel-Crafts alkylation and the subsequent chiral resolution. In the cyclization step, the precursor 2-chloro-N-(4-chlorobenzyl)propane-1-amine undergoes ring closure in the presence of potent Lewis acids such as anhydrous aluminum chloride or zinc chloride. The reaction mechanism involves the coordination of the Lewis acid with the chloro substituent, generating a reactive carbocation or complex that facilitates electrophilic aromatic substitution on the adjacent phenyl ring. This step is critically optimized by controlling the reaction temperature between 60°C and 160°C in high-boiling solvents like dichlorobenzene, ensuring complete conversion while minimizing polymerization or degradation side reactions. The choice of Lewis acid and the molar ratio (0.8 to 2 equivalents) are pivotal in driving the equilibrium towards the desired 1-methyl-2,3,4,5-tetrahydro-1H-3-benzazepine scaffold. Following cyclization, the crude racemic mixture is subjected to a sophisticated resolution process using L-tartaric acid. This chiral resolving agent selectively forms a crystalline salt with the (R)-enantiomer, leveraging differences in solubility to separate it from the (S)-isomer. Recrystallization from aqueous or alcoholic media further enhances the enantiomeric excess to greater than 99%, ensuring that the final hydrochloride salt meets the rigorous stereochemical purity standards required for FDA-approved pharmaceutical ingredients.
Impurity control is inherently built into this mechanistic design through the selection of mild reaction conditions and specific reagent stoichiometry. For instance, the initial aminolysis is conducted at moderate temperatures (50°C to 70°C) in methanol or ethanol, which suppresses the formation of di-substituted amides often seen in harsher acylation conditions. During the halogenation phase, the use of thionyl chloride at low temperatures (0°C to 5°C) followed by triethylamine neutralization prevents the over-chlorination of the propyl chain. Furthermore, the reduction step employs sodium borohydride in combination with boron trifluoride or iodine, a system that selectively reduces the amide bond without affecting the aryl chloride moiety, a common vulnerability in catalytic hydrogenation methods. The final salt formation with hydrochloric acid gas in ethyl acetate ensures the precipitation of the product as a stable hydrochloride salt, effectively trapping any remaining basic impurities in the mother liquor. This multi-layered approach to impurity management results in a final product with purity exceeding 99.5%, significantly reducing the burden on downstream purification units and enhancing the overall yield of the manufacturing process.
How to Synthesize Lorcaserin Hydrochloride Efficiently
The implementation of this synthesis requires strict adherence to the optimized parameters outlined in the patent embodiments to ensure reproducibility and safety. The process begins with the dissolution of p-chlorophenyl acetate in a protic solvent, followed by the addition of isopropanolamine under stirring. Temperature control is vital during the exothermic aminolysis to prevent solvent loss and byproduct formation. Subsequent steps involve careful handling of halogenating agents and reducing agents, necessitating appropriate ventilation and quenching protocols. The cyclization step demands anhydrous conditions to maintain the activity of the Lewis acid catalyst. Finally, the resolution step requires precise pH adjustment and temperature cycling to maximize the recovery of the desired enantiomer. For a detailed, step-by-step breakdown of the exact reagent quantities, reaction times, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Perform aminolysis of p-chlorophenyl acetate with isopropanolamine to form the hydroxypropyl acetamide intermediate.
- Convert the hydroxyl group to a chloro or bromo group using halide reagents like thionyl chloride or phosphorus pentachloride.
- Reduce the amide bond using sodium borohydride to generate the chloro-N-(4-chlorobenzyl)propane-1-amine precursor.
- Execute intramolecular Friedel-Crafts alkylation using Lewis acids such as aluminum chloride to close the benzazepine ring.
- Resolve the racemic mixture using L-tartaric acid followed by salt formation with hydrochloric acid to obtain the final API intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN103333111A offers transformative advantages that extend far beyond simple chemical yield improvements. The strategic shift to p-chlorophenyl acetate as the starting material fundamentally alters the cost structure of the supply chain. Unlike the specialized amines required in previous methods, p-chlorophenyl acetate is a bulk commodity chemical produced by numerous manufacturers globally, ensuring a stable and competitive pricing environment. This abundance mitigates the risk of supply disruptions caused by single-source dependencies. Furthermore, the elimination of phosphorus tribromide removes a significant logistical and safety burden; this hazardous material requires special handling, storage, and disposal protocols that inflate operational expenditures. By replacing it with safer, more manageable reagents like thionyl chloride, facilities can reduce their environmental compliance costs and insurance premiums. The simplified post-treatment procedures, which avoid complex chromatographic separations or extensive aqueous washes, translate directly into reduced utility consumption and shorter batch cycle times. These cumulative efficiencies allow for a drastic simplification of the manufacturing workflow, enabling producers to respond more agilely to market demand fluctuations without compromising on quality or margin.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of high-cost inputs with commoditized alternatives. The use of p-chlorophenyl acetate eliminates the premium associated with custom-synthesized amines, while the avoidance of expensive condensation agents (such as those used in boronic acid catalysis) removes a major variable cost component. Additionally, the high atom economy of the aminolysis and cyclization steps minimizes raw material waste. The ability to perform the reduction and cyclization without requiring ultra-dry conditions or exotic catalysts further lowers the barrier to entry for contract manufacturing organizations, fostering a more competitive supplier landscape that drives down overall acquisition costs for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of the chemical transformations involved. The reactions utilize standard industrial solvents like toluene, methanol, and dichlorobenzene, which are widely stocked and less prone to regulatory restrictions compared to specialized reagents. The process tolerance to minor variations in temperature and stoichiometry, as evidenced by the multiple successful embodiments in the patent, ensures consistent output even in large-scale reactors where heat transfer dynamics differ from lab-scale flasks. This reliability reduces the incidence of failed batches and off-spec material, ensuring a steady flow of high-purity intermediates to downstream formulation partners. The shortened production cycle, achieved by removing time-consuming water removal steps, allows for increased throughput per annum, effectively expanding capacity without the need for capital-intensive infrastructure expansion.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route represents a significant advancement in green chemistry principles applicable to fine chemical manufacturing. The reduction in hazardous waste generation, particularly the avoidance of heavy metal catalysts and corrosive bromine waste, simplifies effluent treatment and aligns with increasingly stringent global environmental regulations. The process is inherently scalable; the exothermic nature of the aminolysis and cyclization steps can be managed effectively in continuous flow reactors or large batch vessels with appropriate cooling systems. This scalability ensures that the method remains viable as production volumes ramp up from pilot plant scales to multi-ton commercial campaigns. The straightforward isolation of the product via crystallization rather than distillation or column chromatography further enhances the energy efficiency of the process, contributing to a lower carbon footprint for the manufactured pharmaceutical intermediate.
Frequently Asked Questions (FAQ)
The technical nuances of this synthesis often raise questions regarding its practical implementation and regulatory alignment. Stakeholders frequently inquire about the specific impurity profiles generated during the Friedel-Crafts cyclization and the efficacy of the chiral resolution in removing the (S)-enantiomer. Others seek clarification on the solvent recovery rates and the potential for recycling mother liquors to further enhance process economics. Understanding these details is crucial for technology transfer teams evaluating the feasibility of adopting this route. The following answers address the most critical technical and commercial queries based on the data provided in the patent documentation, offering clarity for decision-makers assessing this technology for their supply networks.
Q: What are the primary cost advantages of the synthesis method in CN103333111A compared to prior art?
A: The method utilizes p-chlorophenyl acetate, a significantly cheaper and more commercially available starting material compared to the expensive 4-chlorophenethylamine required in older routes. Additionally, it avoids costly condensation agents and hazardous reagents like phosphorus tribromide, simplifying post-treatment and reducing waste disposal costs.
Q: How does this process ensure high enantiomeric purity for the final Lorcaserin product?
A: The process incorporates a robust chiral resolution step using L-tartaric acid. The crude benzazepine base is treated with the resolving agent to form a tartrate salt, which is then recrystallized. This effectively isolates the desired (R)-enantiomer with an enantiomeric excess greater than 99%, meeting stringent pharmaceutical specifications.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights the suitability for industrial production. The reactions utilize common solvents like methanol, toluene, and dichlorobenzene, and the operations are designed for continuity. The avoidance of moisture-sensitive steps found in other methods (like boronic acid catalysis requiring long water removal) significantly shortens the production cycle, facilitating scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lorcaserin Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of CN103333111A are fully realized in practice. We have invested heavily in state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to monitor every critical process parameter, guaranteeing stringent purity specifications for every batch of Lorcaserin hydrochloride intermediate we produce. Our facility is designed to handle the specific solvent systems and reaction conditions required by this novel route, including the safe management of Lewis acids and chlorinating agents, providing our partners with a secure and compliant source of supply.
We invite global pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cost-effective synthesis technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and comprehensive route feasibility assessments. Let us help you optimize your supply chain for Lorcaserin production, ensuring you stay competitive in the rapidly evolving anti-obesity therapeutic market while maintaining the highest standards of product integrity and delivery reliability.
