Advanced Chiral Resolution Technology for High-Purity Argatroban Intermediates and Commercial Scalability
The pharmaceutical industry continuously demands higher standards for chiral purity, particularly for potent anticoagulants like Argatroban. Patent CN109761886B introduces a groundbreaking resolution method specifically targeting the isomer impurities of Argatroban starting materials. This technology addresses the critical challenge of separating (2S,4R)-ethyl 4-methyl-2-piperidinecarboxylate from its racemic mixture with exceptional precision. By leveraging a specialized diastereomeric salt formation strategy, the process achieves an optical purity of 99.1% and a total resolution yield reaching 45.8%. For R&D directors and procurement specialists, this represents a significant leap forward in process chemistry, offering a robust pathway to secure high-quality intermediates essential for the synthesis of thrombin inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of 4-methyl-2-piperidinecarboxylic acid ethyl ester racemates has relied heavily on L-tartaric acid as the resolving agent. While functional, these legacy processes are plagued by inherent inefficiencies that impact both cost and quality. The solubility differences between the diastereomeric salts formed with L-tartaric acid are often insufficient to drive high-yield crystallization, resulting in significant material loss during mother liquor disposal. Furthermore, the chiral purity achieved through these traditional methods is frequently suboptimal, necessitating additional recrystallization steps that extend production timelines and increase solvent consumption. These limitations create bottlenecks in the supply chain for Argatroban, where strict regulatory requirements demand near-perfect stereochemical control to avoid the formation of inactive or toxic isomer impurities in the final drug product.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally shifts the resolution paradigm by employing D-(+)-dibenzoyltartaric acid (DBTA) instead of simple tartaric acid. This bulky chiral acid forms a diastereomeric salt with the target (2S,4R) isomer that exhibits vastly superior crystallization properties in a specific binary solvent system of isopropanol and acetonitrile. The process is further optimized by the addition of 1-butylpyridinium bromide and a seed crystal of D-(+)-dibenzoyltartaric acid pyridine salt, which act synergistically to control nucleation and crystal growth. This precise engineering of the crystallization environment allows for the direct isolation of the target salt with high fidelity. As illustrated in the reaction scheme below, the formation of Compound I is highly selective, setting the stage for the subsequent liberation of the pure free base.
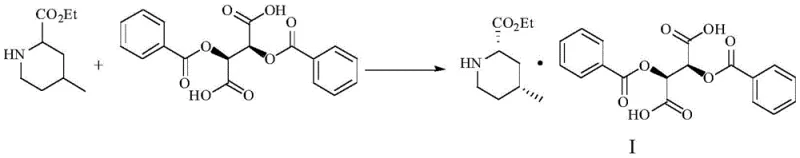
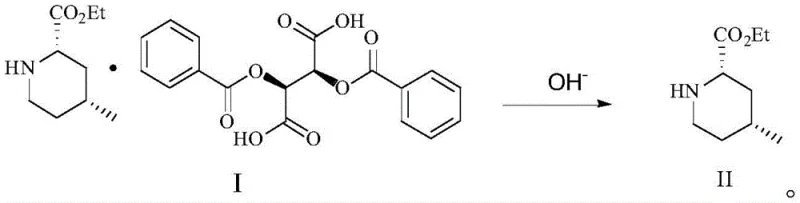
Following the isolation of the diastereomeric salt, the process employs a mild basic treatment to liberate the free amine. This step is crucial for recovering the valuable chiral intermediate without racemization. The use of aqueous sodium carbonate in methanol ensures a gentle pH adjustment that preserves the stereochemical integrity of the molecule while efficiently breaking the salt bond. The resulting free base, Compound II, is then extracted into an organic phase, yielding the high-purity (2S,4R)-ethyl 4-methyl-2-piperidinecarboxylate required for downstream Argatroban synthesis. This two-step sequence—selective salt formation followed by gentle liberation—provides a streamlined alternative to the cumbersome multi-step protections and deprotections seen in older synthetic routes.
Mechanistic Insights into Diastereomeric Salt Resolution
The success of this resolution hinges on the thermodynamic and kinetic differences in solubility between the diastereomeric salts formed in the reaction mixture. D-(+)-dibenzoyltartaric acid possesses large benzoyl groups that introduce significant steric hindrance and specific intermolecular interactions, such as pi-stacking and hydrogen bonding, which differ markedly between the (2S,4R) and (2R,4S) isomers of the piperidine derivative. In the isopropanol-acetonitrile solvent matrix, the salt formed with the desired (2S,4R) isomer reaches its saturation point much earlier than the undesired isomer salt. This differential solubility drives the preferential precipitation of the target compound. The addition of 1-butylpyridinium bromide likely serves as a crystal habit modifier, adsorbing onto specific crystal faces to inhibit the growth of impurities or to prevent oiling out, ensuring that the product precipitates as a filterable solid rather than an amorphous gum.
Impurity control is inherently built into this mechanism through the seeding strategy. By introducing D-(+)-dibenzoyltartaric acid pyridine salt during the cooling phase, the process provides a template for heterogeneous nucleation. This ensures that crystallization initiates on the correct lattice structure, effectively suppressing the spontaneous nucleation of the unwanted diastereomer which might otherwise occur under supersaturated conditions. This level of control is vital for maintaining the optical purity above 99%. Furthermore, the basic liberation step is optimized to prevent epimerization at the chiral centers adjacent to the carbonyl group. By carefully controlling the pH and temperature during the sodium carbonate treatment, the process minimizes the risk of base-catalyzed racemization, ensuring that the high enantiomeric excess achieved in the crystallization step is preserved in the final isolated intermediate.
How to Synthesize (2S,4R)-Ethyl 4-methyl-2-piperidinecarboxylate Efficiently
The synthesis protocol outlined in the patent offers a reproducible route for manufacturing this critical chiral building block. It combines standard unit operations—heating, cooling, filtration, and extraction—with precise chemical controls to maximize yield and purity. The key to success lies in the strict adherence to the solvent ratios and the timing of additive introduction. Operators must ensure the reaction mixture is heated sufficiently to dissolve all components before the controlled cooling begins, as this thermal cycle is essential for establishing the correct supersaturation profile. The following guide summarizes the operational workflow derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- React racemic ethyl 4-methyl-2-piperidinecarboxylate with D-(+)-dibenzoyltartaric acid in an isopropanol and acetonitrile solvent system at 65-68°C, utilizing 1-butylpyridinium bromide as an additive to facilitate crystallization of the target diastereomeric salt.
- Cool the reaction mixture to room temperature and add D-(+)-dibenzoyltartaric acid pyridine salt to induce static crystallization over 12 hours, followed by filtration to isolate the white crystalline salt intermediate.
- Dissolve the isolated salt in methanol, adjust pH to approximately 8.0 using 10% sodium carbonate solution to liberate the free base, then extract with dichloromethane and purify to obtain the high-purity (2S,4R) isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution technology translates directly into operational resilience and cost optimization. Traditional methods often require multiple recrystallizations to meet purity specifications, which drastically increases solvent usage, waste generation, and processing time. By achieving high optical purity in a single crystallization step, this new method significantly reduces the manufacturing footprint. The elimination of complex protection-deprotection sequences found in alternative synthetic routes further simplifies the supply chain, reducing the number of raw materials that need to be sourced and qualified. This simplification mitigates the risk of supply disruptions caused by the shortage of specialized reagents or catalysts.
- Cost Reduction in Manufacturing: The primary driver for cost savings is the substantial improvement in total yield and the reduction of processing steps. Higher yields mean less raw material is required per kilogram of final product, directly lowering the cost of goods sold. Additionally, the use of commodity solvents like isopropanol and acetonitrile, rather than exotic or hazardous chemicals, keeps material costs stable and predictable. The avoidance of expensive transition metal catalysts, which are often required in asymmetric hydrogenation routes, removes the need for costly metal scavenging and validation steps, further streamlining the production budget and reducing environmental compliance costs associated with heavy metal waste.
- Enhanced Supply Chain Reliability: The robustness of the crystallization process ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance in pharmaceutical manufacturing. A reliable process with high success rates reduces the likelihood of batch failures, which can cause significant delays in delivery schedules. Furthermore, the raw materials required, such as D-(+)-dibenzoyltartaric acid and the piperidine racemate, are commercially available from multiple global suppliers. This diversification of the supply base prevents single-source bottlenecks and ensures that production can continue uninterrupted even if one supplier faces logistical challenges, thereby securing the continuity of supply for downstream Argatroban production.
- Scalability and Environmental Compliance: The process is inherently scalable because it relies on crystallization, a unit operation that scales linearly from grams to tons with minimal re-optimization. Unlike chromatographic separations which are difficult and expensive to scale, filtration and crystallization are standard industrial practices. From an environmental perspective, the method generates less waste due to higher efficiency and the use of recyclable solvents. The absence of heavy metals simplifies wastewater treatment protocols, making it easier for manufacturing sites to meet stringent environmental regulations. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines. The answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is D-(+)-dibenzoyltartaric acid preferred over L-tartaric acid for this resolution?
A: Prior art methods utilizing L-tartaric acid often suffer from lower resolution yields and insufficient chiral purity. The use of D-(+)-dibenzoyltartaric acid in this specific solvent system creates a diastereomeric salt with significantly different solubility characteristics, allowing for more efficient separation of the (2S,4R) isomer and achieving optical purity up to 99.1%.
Q: What is the role of 1-butylpyridinium bromide in the crystallization process?
A: 1-butylpyridinium bromide acts as a critical additive during the salt formation step. It functions to modify crystal growth habits and potentially suppress the nucleation of unwanted isomers, thereby enhancing the selectivity of the crystallization process and contributing to the high overall yield and purity of the final product.
Q: Can this resolution method be scaled for industrial production of Argatroban intermediates?
A: Yes, the method is designed for scalability. It utilizes common organic solvents like isopropanol and acetonitrile and avoids complex chromatographic separations. The reliance on crystallization and standard extraction techniques makes it highly suitable for commercial scale-up, ensuring consistent supply chain continuity for anticoagulant manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 4-methyl-2-piperidinecarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development and commercialization of life-saving anticoagulants. Our technical team has extensively analyzed advanced resolution technologies like the one described in CN109761886B to ensure our manufacturing capabilities align with the highest industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of chiral intermediate meets the exacting demands of global regulatory agencies.
We invite you to collaborate with us to optimize your supply chain for Argatroban and related thrombin inhibitors. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our advanced resolution processes can reduce your overall manufacturing costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to deliver reliable, high-quality pharmaceutical intermediates that drive your success in the competitive global market.
