Advanced Chiral Copper Complexes for High-Efficiency Asymmetric Henry Reactions
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for constructing carbon-carbon bonds, particularly in the realm of asymmetric synthesis. Patent CN102010443B introduces a significant advancement in this field through the development of a novel chiral phosphonous bis-amino-oxazoline copper complex. This organometallic compound represents a breakthrough in catalytic technology, specifically designed to facilitate the Henry reaction with exceptional efficiency. The patent details a robust synthetic route that transforms simple, commercially available precursors into a sophisticated catalyst capable of driving reactions with yields reaching up to 98%. For R&D directors and process chemists, this technology offers a compelling solution for generating chiral beta-nitroalcohol intermediates, which are critical building blocks for various active pharmaceutical ingredients (APIs). The structural integrity and specific coordination geometry of this copper complex provide a stable platform for high-throughput screening and large-scale manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for catalyzing Henry reactions often suffer from significant drawbacks that hinder industrial scalability and economic viability. Many conventional catalysts lack the necessary stereocontrol to produce single-enantiomer products with high purity, necessitating costly and time-consuming downstream purification steps such as chiral resolution. Furthermore, older catalytic systems frequently require harsh reaction conditions, including extreme temperatures or pressures, which can degrade sensitive substrates and lead to the formation of unwanted byproducts. The use of non-chiral ligands or less optimized metal centers often results in poor turnover numbers, meaning that large quantities of expensive metals are required to achieve acceptable conversion rates. These inefficiencies not only drive up the cost of goods sold but also create substantial waste management challenges, complicating environmental compliance for manufacturing facilities striving for greener chemistry protocols.
The Novel Approach
The innovative approach detailed in patent CN102010443B overcomes these historical barriers by employing a meticulously designed chiral ligand system coordinated with copper. By utilizing a bis-amino-oxazoline framework linked via a phosphonous diamide bridge, the new complex creates a highly defined chiral environment around the metal center. This specific architecture ensures precise substrate orientation during the catalytic cycle, leading to superior enantioselectivity and reaction rates under relatively mild conditions. The synthesis method itself is streamlined, avoiding the need for exotic reagents or complex multi-step purifications that typically plague the production of specialized catalysts. Instead, it relies on standard reflux techniques in common solvents like chlorobenzene, toluene, and ethanol, making the transition from laboratory bench to pilot plant significantly smoother. This novel methodology not only enhances the chemical outcome but also aligns perfectly with modern principles of process intensification and cost reduction in fine chemical manufacturing.
Mechanistic Insights into Cu-N Metal-Organic Coordination
The catalytic prowess of this complex stems from the intricate interplay between the copper ion and the nitrogen/oxygen donor atoms within the bis-amino-oxazoline ligand. Mechanistically, the copper center acts as a Lewis acid, activating the nitroalkane substrate by coordinating with the nitro group, thereby increasing the acidity of the alpha-protons. Simultaneously, the chiral pocket formed by the phenyl-substituted oxazoline rings dictates the facial selectivity of the nucleophilic attack on the aldehyde carbonyl. The phosphonous diamide linker plays a crucial role in stabilizing this conformation, preventing ligand dissociation and ensuring the catalyst remains active throughout the reaction cycle. This robust coordination sphere minimizes the formation of racemic byproducts, a common issue in less rigid catalytic systems. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as solvent polarity and temperature, to maximize the turnover frequency while maintaining high optical purity in the final product.
Impurity control is another critical aspect where this mechanistic design excels. In many asymmetric catalyses, trace metals or ligand degradation products can contaminate the final API intermediate, requiring rigorous purification. However, the stability of the copper di{N-2-[(4S)-4,5-dihydro-4-phenyl-2-oxazoline]-diphenyl-phenylphosphonic diamide complex reduces the likelihood of ligand decomposition under reaction conditions. The specific choice of copper acetate monohydrate as the metal source ensures a clean initiation of the catalytic species without introducing extraneous anions that could interfere with the reaction pathway. Furthermore, the precipitation of the complex as reddish-brown crystals from ethanol indicates a high degree of crystallinity and purity, which correlates directly with consistent catalytic performance batch-to-batch. This level of control over the catalyst's physical and chemical properties is essential for meeting the stringent regulatory requirements of the pharmaceutical industry regarding impurity profiles.
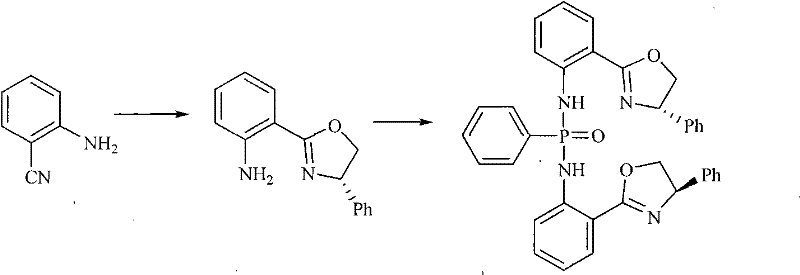
How to Synthesize Chiral Phosphonous Bis-Amino-Oxazoline Copper Complex Efficiently
The synthesis of this high-value catalyst is divided into distinct stages that prioritize yield and purity at every step. The process begins with the formation of the chiral oxazoline intermediate, followed by phosphorylation to create the bidentate ligand, and concludes with metal coordination. Each stage utilizes standard unit operations familiar to chemical engineers, such as reflux, extraction, and crystallization, ensuring that the technology is readily transferable to existing manufacturing infrastructure. The use of anhydrous and oxygen-free conditions during the initial ligand synthesis is critical to prevent oxidation of the phosphorus center, which would deactivate the catalyst. Detailed standardized operating procedures for these steps are essential for maintaining reproducibility and safety in a commercial setting.
- React 2-cyano-aniline with L-phenylglycinol using anhydrous ZnCl2 catalyst in chlorobenzene under reflux to form the oxazoline intermediate.
- Condense the intermediate with phenylphosphonyl chloride in toluene and triethylamine to generate the chiral phosphonous diamide ligand.
- Coordinate the ligand with copper acetate monohydrate in absolute ethanol under reflux to precipitate the final reddish-brown copper complex crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology presents a strategic opportunity to optimize sourcing strategies and reduce overall production costs. The reliance on commodity chemicals like 2-cyano-aniline and L-phenylglycinol as starting materials means that the supply chain is not dependent on scarce or geopolitically sensitive resources. This abundance of raw materials ensures a stable supply base, mitigating the risks associated with price volatility and supplier disruptions that often affect specialty chemical markets. Additionally, the high catalytic efficiency reported in the patent implies that lower loading levels may be sufficient to achieve desired conversion rates, further reducing the consumption of copper salts and ligands per kilogram of product. This efficiency translates directly into improved margin structures for the final pharmaceutical intermediates produced using this method.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals like palladium or rhodium in favor of abundant copper significantly lowers the raw material cost basis for the catalyst itself. Moreover, the high yield of 98% in the model Henry reaction suggests minimal waste generation and reduced need for recycling unreacted starting materials. By streamlining the synthesis into fewer steps with high atom economy, manufacturers can achieve substantial cost savings in both material usage and energy consumption. The ability to operate under atmospheric pressure and moderate reflux temperatures also reduces the capital expenditure required for specialized high-pressure reactors, making the process accessible to a wider range of contract manufacturing organizations.
- Enhanced Supply Chain Reliability: The synthetic route described avoids the use of highly unstable or hazardous reagents that often complicate logistics and storage. Solvents such as chlorobenzene, toluene, and ethanol are widely available globally, ensuring that production can be sustained even during regional supply shortages. The robustness of the catalyst synthesis, characterized by simple filtration and crystallization steps, reduces the dependency on highly skilled labor for complex chromatographic separations. This operational simplicity enhances the reliability of the supply chain, allowing for consistent delivery schedules and reducing the lead time for high-purity pharmaceutical intermediates needed for clinical trials and commercial launches.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the use of homogeneous reaction conditions that are easily modeled and controlled in large vessels. The precipitation of the final product as crystals simplifies isolation, removing the need for complex distillation or extraction workflows that generate large volumes of solvent waste. From an environmental perspective, the use of copper, a less toxic metal compared to many alternatives, aligns with green chemistry initiatives and simplifies wastewater treatment protocols. This compliance with environmental standards reduces the regulatory burden on manufacturing sites and supports corporate sustainability goals, making the technology attractive for long-term investment in eco-friendly chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral copper complex technology. These insights are derived directly from the experimental data and claims presented in patent CN102010443B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this catalyst into their existing process pipelines.
Q: What is the primary application of this chiral copper complex?
A: This complex is specifically designed as a highly efficient catalyst for asymmetric Henry reactions, achieving yields up to 98% in the synthesis of beta-nitroalcohols from benzaldehyde.
Q: What are the key starting materials for this synthesis?
A: The synthesis relies on readily available precursors including 2-cyano-aniline (2-aminobenzonitrile), L-phenylglycinol, phenylphosphonyl chloride, and copper acetate monohydrate.
Q: How does this catalyst improve upon traditional methods?
A: By utilizing a unique bis-amino-oxazoline phosphonous ligand structure, this copper complex offers superior enantioselectivity and catalytic activity compared to conventional non-chiral or less optimized metal complexes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphonous Bis-Amino-Oxazoline Copper Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN102010443B for the synthesis of high-value chiral intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the enantiomeric excess and metal content of every batch. Our commitment to quality assurance guarantees that the catalysts and intermediates we supply meet the exacting standards required by global regulatory bodies, providing you with peace of mind and consistency in your manufacturing operations.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current production volumes and target specifications. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral catalysis can drive efficiency and profitability in your supply chain.
