Scalable Metal-Free Electrochemical Synthesis of Indole Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, sustainable methodologies for constructing privileged scaffolds like the indole nucleus. Patent CN112708902B introduces a groundbreaking electrochemical approach that addresses critical pain points in traditional synthesis. This technology leverages iodine-induced electrocatalytic intramolecular C(sp2)-H oxidative cyclization to transform 2-vinylaniline derivatives into valuable indole compounds. By replacing stoichiometric chemical oxidants and expensive transition metals with electricity and catalytic iodide salts, this method offers a paradigm shift towards greener manufacturing. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for bioactive molecules and functional materials. The process operates under mild conditions, utilizing simple undivided cells and readily available reagents, ensuring that the transition from laboratory discovery to commercial production is seamless and economically viable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole derivatives has relied heavily on transition metal catalysis, particularly palladium-catalyzed oxidative cyclization, or the use of stoichiometric hypervalent iodine reagents. While effective, these traditional pathways suffer from inherent drawbacks that impact both cost and environmental compliance. Transition metal catalysts are not only expensive but also pose significant challenges regarding residual metal contamination in final API intermediates, necessitating costly purification steps to meet stringent regulatory limits. Furthermore, methods employing stoichiometric oxidants often exhibit poor atom economy, generating substantial quantities of waste byproducts such as iodobenzene. This accumulation of chemical waste increases disposal costs and complicates the environmental footprint of the manufacturing process, making it less attractive for modern green chemistry initiatives.
The Novel Approach
The electrochemical method disclosed in CN112708902B circumvents these issues by utilizing electrons as the primary oxidant. In this novel system, an iodide electrolyte serves as a redox mediator, facilitating the oxidative cyclization without being consumed in stoichiometric amounts. The addition of thiocyanate further enhances the reaction efficiency, likely acting as a nucleophilic mediator or stabilizing agent. This metal-free strategy eliminates the risk of heavy metal contamination entirely, simplifying the downstream purification workflow. Moreover, because the oxidant is generated in situ at the anode, the process avoids the generation of stoichiometric iodobenzene waste, resulting in superior atom economy. This approach aligns perfectly with the industry's demand for high-purity pharmaceutical intermediates produced via sustainable, cost-effective routes.

Mechanistic Insights into Iodine-Induced Electrocatalytic Cyclization
The core of this technology lies in the anodic oxidation of iodide ions to generate reactive iodine species, such as molecular iodine or cationic iodine intermediates, which activate the alkene moiety of the 2-vinylaniline substrate. This activation renders the double bond susceptible to intramolecular nucleophilic attack by the adjacent nitrogen atom, initiating the ring-closing event. The presence of thiocyanate ions in the electrolyte mixture plays a crucial role, potentially intercepting reactive intermediates to prevent side reactions or facilitating the regeneration of the active iodine species. This synergistic interaction between the iodide electrolyte and the thiocyanate additive ensures a smooth catalytic cycle driven by the applied electric current. The result is a highly selective formation of the indole core with minimal formation of over-oxidized byproducts or polymeric tars.
From an impurity control perspective, the electrochemical nature of the reaction allows for precise tuning of the oxidation potential. By controlling the current density and electrode material, operators can minimize the formation of undesired oxidation products that typically plague chemical oxidant methods. The absence of transition metals means there are no metal-ligand complexes to separate, leading to a cleaner crude reaction profile. This purity advantage is critical for pharmaceutical applications where impurity profiles must be tightly controlled. The method demonstrates excellent functional group tolerance, accommodating various substituents on the aromatic ring, which suggests a robust mechanism that is not easily perturbed by electronic effects of the substrate.
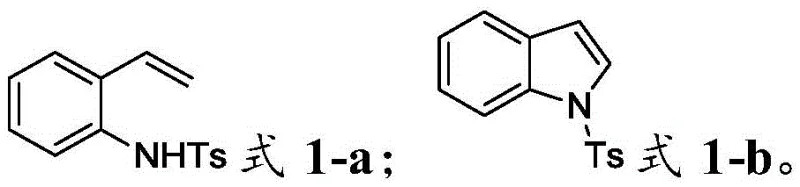
How to Synthesize N-Protected Indoles Efficiently
The synthesis protocol outlined in the patent provides a straightforward recipe for accessing high-purity indole derivatives. The process begins with the preparation of the reaction mixture in an undivided electrolytic cell, combining the 2-vinylaniline precursor with catalytic amounts of iodide salt and thiocyanate in a polar aprotic solvent. The detailed standardized synthesis steps below outline the specific molar ratios, electrode choices, and workup procedures required to replicate the high yields reported in the patent examples. This section serves as a practical guide for process chemists looking to implement this technology in their own laboratories or pilot plants.
- Mix the 2-vinylaniline substrate with an iodide electrolyte (e.g., tetramethylammonium iodide) and a thiocyanate salt (e.g., potassium thiocyanate) in a suitable solvent like acetonitrile.
- Perform electrolysis in an undivided cell using inert electrodes (Pt or C) at a constant current (e.g., 5 mA) and elevated temperature (e.g., 80°C) until reaction completion.
- Concentrate the reaction mixture via rotary evaporation and purify the crude indole product using column chromatography with a petroleum ether/ethyl acetate system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical methodology offers tangible strategic benefits beyond mere technical novelty. The elimination of precious metal catalysts directly translates to reduced raw material costs and mitigates supply risks associated with volatile metal markets. Furthermore, the simplified purification process reduces solvent consumption and processing time, leading to significant operational expenditure savings. The use of commodity chemicals like potassium thiocyanate and ammonium iodide ensures a stable and reliable supply chain, free from the geopolitical constraints often affecting specialized catalysts.
- Cost Reduction in Manufacturing: The removal of expensive palladium catalysts and stoichiometric oxidants drastically lowers the bill of materials. Additionally, the avoidance of heavy metal scavenging resins and complex filtration steps reduces downstream processing costs. The high atom economy means less waste to treat, lowering environmental compliance fees and disposal costs associated with hazardous byproducts like iodobenzene.
- Enhanced Supply Chain Reliability: The reagents required for this process, including iodide salts and thiocyanates, are bulk commodities with stable global availability. This contrasts sharply with specialized ligands or organometallic catalysts that may face supply bottlenecks. By relying on electricity as the primary reagent, the process decouples production capacity from the logistics of chemical oxidant delivery, ensuring consistent manufacturing throughput.
- Scalability and Environmental Compliance: The use of undivided cells simplifies reactor design, making scale-up from gram to tonnage more straightforward compared to complex divided cell setups. The metal-free nature of the reaction inherently satisfies strict environmental regulations regarding heavy metal discharge. This facilitates faster regulatory approval for new drug filings and reduces the administrative burden of environmental reporting for manufacturing sites.
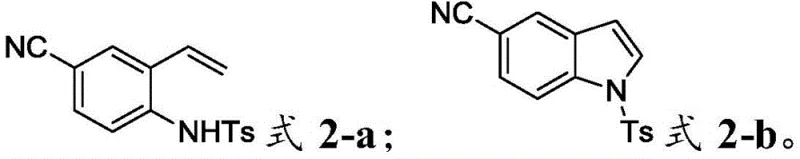
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical indole synthesis. These answers are derived directly from the experimental data and technical disclosures within patent CN112708902B, providing clarity on reaction scope, safety, and scalability for potential adopters of this technology.
Q: Does this electrochemical method require expensive transition metal catalysts?
A: No, the method described in patent CN112708902B is explicitly metal-free. It utilizes an iodide electrolyte and electricity to drive the oxidative cyclization, eliminating the need for palladium or other costly transition metals often used in traditional indole synthesis.
Q: What are the environmental advantages of this synthesis route?
A: This process offers high atom economy by avoiding stoichiometric chemical oxidants that generate heavy waste. Unlike traditional hypervalent iodine methods, it does not produce equivalent amounts of iodobenzene byproducts, significantly reducing downstream purification burdens and environmental impact.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the protocol uses an undivided electrolytic cell and common reagents like potassium thiocyanate and ammonium iodide. The simplicity of the setup and the absence of sensitive metal catalysts make it highly amenable to commercial scale-up for producing drug intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and efficiently, ensuring that the benefits of this metal-free technology are fully realized at scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of indole intermediate meets the highest global standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain with sustainable, high-quality indole intermediates.
