Advanced Synthesis of Levosimendan Intermediate via Efficient Early-Stage Chiral Resolution
Advanced Synthesis of Levosimendan Intermediate via Efficient Early-Stage Chiral Resolution
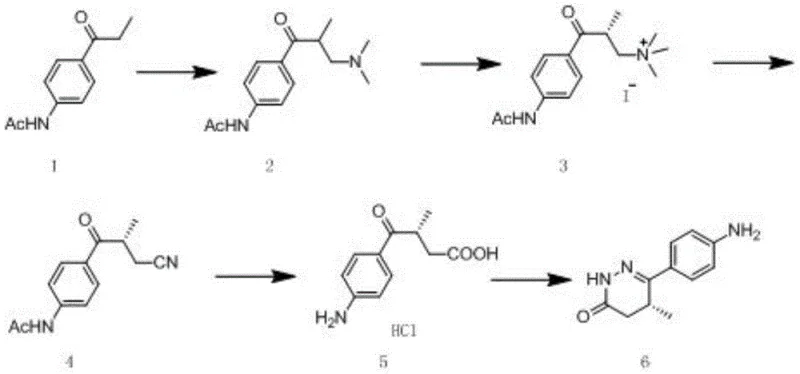
The pharmaceutical industry continuously seeks robust and scalable methodologies for producing high-value chiral intermediates, particularly for cardiovascular medications like Levosimendan. Patent CN110885315A introduces a groundbreaking preparation method for (R)-6-(4-aminophenyl)-5-methyl-4,5-dihydropyridazin-3(2H)-one, a critical building block in this therapeutic class. This technology addresses longstanding inefficiencies in chiral synthesis by shifting the resolution step to an earlier stage of the synthetic pathway. By utilizing N-(4-propionylphenyl)acetamide as a starting material, the process employs a Mannich reaction followed by a highly efficient chemical resolution. This strategic modification ensures that subsequent transformations proceed with a single enantiomer, thereby eliminating the risk of racemization and maximizing the utility of raw materials. The result is a streamlined protocol that delivers exceptional chiral purity suitable for stringent regulatory requirements.
For procurement specialists and supply chain managers, the implications of this patent are profound. Traditional methods often struggle with the physical properties of the final intermediate, making purification difficult and costly. In contrast, this novel approach targets an intermediate amine species that exhibits more favorable characteristics for salt formation and crystallization. This shift not only simplifies the operational workflow but also enhances the economic viability of large-scale manufacturing. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advantages is key to securing a stable supply of high-quality active pharmaceutical ingredients (APIs) and their precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing this specific pyridazinone derivative have been plagued by significant technical hurdles, primarily centered around the difficulty of chiral separation. Prior art, such as the methods disclosed in US6355269, typically attempts to resolve the final product or late-stage intermediates using agents like L-tartaric acid in isopropanol. However, these conventional techniques suffer from inherently low resolution efficiency, often yielding comprehensive recovery rates of merely 20 percent. This poor performance necessitates multiple crystallization cycles and complex operational steps, such as thermal filtration, which increase both energy consumption and processing time. Furthermore, the weak alkalinity of the final heterocyclic product makes it difficult to form stable salts with resolving agents, leading to substantial material loss and inflated production costs that are unsustainable for modern generic drug manufacturing.
The Novel Approach
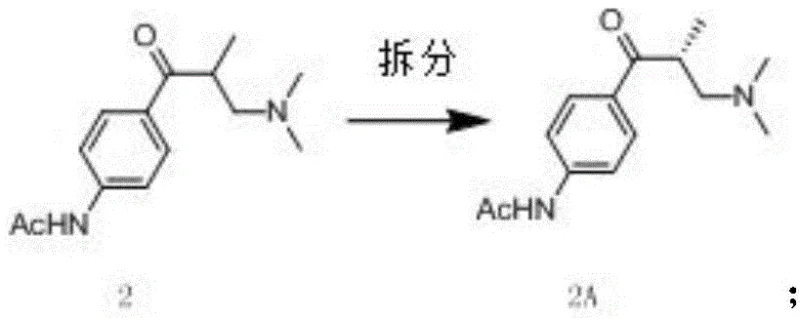
The methodology outlined in CN110885315A fundamentally reengineers the synthesis by introducing the chiral resolution step immediately after the initial Mannich reaction. Instead of struggling with the final pyridazinone ring system, the process resolves the acyclic amine intermediate, Compound 2, into its pure (R)-enantiomer, Compound 2A. This early intervention capitalizes on the distinct physicochemical properties of the amine salt, allowing for high-efficiency separation using cost-effective resolving agents like D-tartaric acid. By isolating the correct stereoisomer before constructing the sensitive heterocyclic core, the process ensures that all subsequent chemical investments are made solely on the desired enantiomer. This strategic pivot drastically reduces waste and simplifies the purification landscape, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Early-Stage Chiral Resolution and Substitution
The core innovation of this synthesis lies in the precise timing of the stereochemical control. The process begins with a Mannich reaction where N-(4-propionylphenyl)acetamide reacts with formaldehyde and dimethylamine to generate the racemic beta-amino ketone. The subsequent resolution utilizes the formation of diastereomeric salts with D-tartaric acid or its derivatives. Because the amine functionality is more basic and accessible than the final heterocyclic nitrogen, the salt formation is thermodynamically favorable and kinetically rapid. This allows for the isolation of the (R)-enantiomer with chiral purity exceeding 99 percent through simple crystallization, a significant improvement over the 93 percent purity often achieved in late-stage resolutions. This high initial purity sets a robust foundation for the remainder of the synthesis, ensuring that impurities do not propagate through the sequence.
Following resolution, the synthetic route proceeds through a quaternization and substitution sequence that is meticulously designed to preserve chirality. The resolved amine is methylated to form a quaternary ammonium salt, which then undergoes nucleophilic substitution with a cyanide source, such as sodium cyanide. Critically, this substitution occurs at the primary carbon atom bearing the nitrogen leaving group, while the chiral center resides at the adjacent alpha-position relative to the carbonyl. Since the chiral carbon is not directly involved in the bond-breaking or bond-forming events of this SN2-type displacement, there is no risk of inversion or racemization. This mechanistic safeguard ensures that the high optical purity established in the early resolution step is maintained throughout the hydrolysis and final cyclization with hydrazine hydrate, delivering a final product with greater than 99.5 percent enantiomeric excess.
How to Synthesize (R)-6-(4-aminophenyl)-5-methyl-4,5-dihydropyridazin-3(2H)-one Efficiently
The synthesis of this high-value chiral intermediate requires precise control over reaction parameters to maximize yield and optical purity. The process initiates with the condensation of the propionyl starting material under acidic conditions, followed by the critical resolution step using tartaric acid derivatives in a mixed solvent system. Once the pure enantiomer is secured, the pathway involves methylation, cyanide displacement, acid hydrolysis, and finally, ring closure. Each step has been optimized to operate under mild conditions, avoiding extreme temperatures or pressures that could degrade the chiral integrity of the molecule. For detailed laboratory protocols and specific stoichiometric ratios required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide below.
- Perform a Mannich reaction on N-(4-propionylphenyl)acetamide with formaldehyde and dimethylamine to form the racemic amine intermediate.
- Execute chiral resolution of the racemic amine using D-tartaric acid derivatives to isolate the high-purity (R)-enantiomer early in the sequence.
- Convert the resolved amine to a quaternary ammonium salt, substitute with cyanide, hydrolyze to the acid, and cyclize with hydrazine hydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical elegance. The primary advantage is the substantial reduction in raw material waste. By resolving the intermediate early, manufacturers avoid processing the unwanted enantiomer through expensive downstream steps like cyanide substitution and cyclization, which involve hazardous reagents and rigorous safety controls. This efficiency translates directly into lower variable costs per kilogram of the final API intermediate. Furthermore, the use of commodity chemicals like formaldehyde, dimethylamine, and sodium cyanide ensures that the supply chain remains resilient against fluctuations in the availability of exotic or proprietary catalysts. This reliance on bulk industrial chemicals enhances supply continuity and mitigates the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of complex thermal filtration steps and the reduction in crystallization cycles significantly lower the operational expenditure associated with purification. Traditional methods often require repeated recrystallization to achieve acceptable purity, consuming vast amounts of solvents and energy. In contrast, this novel route achieves high purity with fewer unit operations, reducing solvent recovery loads and waste disposal costs. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final product, driving down the cost of goods sold (COGS) and improving margin potential for generic drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable manufacturing schedule. Mild temperatures and atmospheric pressure operations reduce the likelihood of equipment failure or safety incidents that can disrupt production timelines. The process is less sensitive to minor variations in reagent quality, providing a wider operating window that ensures consistent batch-to-batch quality. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery deadlines demanded by global pharmaceutical clients, thereby strengthening the partnership between suppliers and drug developers.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates less hazardous waste due to higher atom economy and reduced solvent usage. The avoidance of heavy metal catalysts or expensive chiral ligands simplifies the effluent treatment process, making it easier to comply with increasingly stringent environmental regulations. The simplicity of the workup procedures, such as straightforward filtration and extraction, facilitates seamless scale-up from pilot plant to commercial tonnage. This scalability ensures that the supply can grow in tandem with market demand for Levosimendan, securing long-term availability for patients relying on this critical cardiac medication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this chiral intermediate. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers reflect the consensus on best practices for handling chiral amines and managing the risks associated with cyanide chemistry in a GMP environment.
Q: Why is early-stage resolution preferred over resolving the final Levosimendan intermediate?
A: Resolving the final product often suffers from low yields (around 20%) and requires complex operations like thermal filtration. Early resolution at the amine stage leverages better solubility differences, achieving over 99% chiral purity with simpler crystallization, preventing resource waste on the unwanted enantiomer in downstream steps.
Q: Does the cyanide substitution step cause racemization of the chiral center?
A: No. The process is designed so that the chiral carbon atom is not involved in the bond-breaking or bond-forming events during the substitution reaction. The quaternary ammonium group is displaced from a primary carbon, leaving the adjacent chiral center intact, ensuring the final product maintains >99% optical purity.
Q: What are the cost advantages of this specific synthetic route?
A: The route utilizes cheap and readily available starting materials like N-(4-propionylphenyl)acetamide. By resolving early, the overall yield is significantly improved compared to late-stage resolution, reducing the cost per kilogram of the active chiral intermediate. Additionally, mild reaction conditions lower energy consumption and equipment stress.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-6-(4-aminophenyl)-5-methyl-4,5-dihydropyridazin-3(2H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of life-saving cardiovascular therapies. Our technical team has extensively analyzed the pathway described in CN110885315A and possesses the expertise to implement this advanced resolution strategy effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to full-scale market launch. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee >99% ee, providing you with the confidence needed for regulatory filings.
We invite you to collaborate with us to optimize your supply chain for Levosimendan production. Our engineering team can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this early-resolution route can improve your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot batches and to discuss route feasibility assessments for your unique manufacturing constraints. Let us help you secure a sustainable and cost-effective source of this vital pharmaceutical building block.
