Advanced Synthetic Route for Simvastatin Ammonium Salt Enhancing Commercial Scalability and Purity
Introduction to Advanced Simvastatin Intermediate Technology
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational efficiency, particularly for critical lipid-lowering agents. Patent CN101575286B introduces a transformative methodology for the preparation of simvastatin ammonium salt, a pivotal precursor in the manufacturing of simvastatin API. This intellectual property addresses longstanding challenges associated with traditional alcoholysis routes by introducing a novel intermediate, designated as formula IV, which fundamentally alters the reaction landscape. By shifting the synthetic strategy from a direct, non-selective alcoholysis to a protected ester pathway, the technology ensures that the structural integrity of the molecule is maintained during the initial transformation steps. This approach not only mitigates the formation of difficult-to-remove impurities but also establishes a foundation for consistent, high-yield production that meets the rigorous standards required by global regulatory bodies for cardiovascular therapeutics.
For R&D directors and process chemists, the significance of this patent lies in its ability to decouple the deprotection steps, thereby allowing for better control over the stereochemistry and functional group tolerance of the simvastatin backbone. The transition from the lactone form (Formula I) to the ammonium salt (Formula V) is achieved through a sequence that prioritizes the stability of the intermediate species. This is crucial because unstable intermediates often lead to degradation products that complicate downstream purification. The described method leverages standard organic synthesis principles—specifically selective transesterification and controlled hydrolysis—to create a process that is both chemically elegant and industrially viable. As we delve deeper into the technical specifics, it becomes evident that this route represents a substantial evolution in the manufacturing logic for HMG-CoA reductase inhibitor intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of simvastatin ammonium salt relied heavily on the methodology described by Dabora et al., which involves the direct alcoholysis of 4-acetyl simvastatin (Formula I) to produce intermediate VIII. This conventional pathway suffers from inherent kinetic and thermodynamic disadvantages that severely impact overall process efficiency. The primary bottleneck is the alcoholysis of the acetate moiety, which proceeds at an exceptionally slow rate, often requiring reaction times extending up to 16 hours to reach completion. Such prolonged exposure to reaction conditions increases the probability of side reactions, leading to a complex impurity profile that is difficult to manage. Furthermore, intermediate VIII is typically obtained as an oily substance, a physical state that precludes the use of recrystallization as a purification technique. Since simvastatin derivatives possess large, structurally similar frameworks, the inability to crystallize the intermediate means that impurities carried over from the alcoholysis step persist into the final ammonium salt formation, drastically reducing both yield and purity.
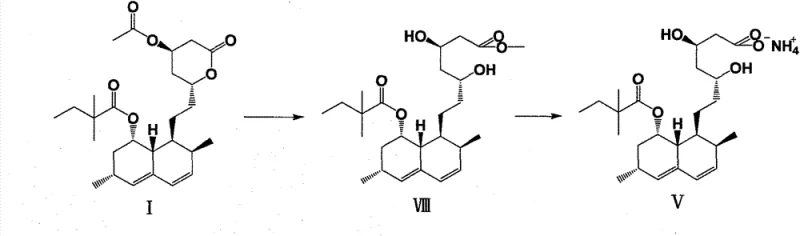
The consequences of these limitations extend beyond the laboratory bench to the commercial supply chain. The low purity of intermediate VIII necessitates extensive and costly downstream processing to meet pharmaceutical grade specifications. The difficulty in crystallizing the ammonium salt from an impure precursor results in significant material loss during isolation, driving up the cost of goods sold (COGS). For procurement managers, this translates to volatile supply availability and higher raw material costs. The reliance on a process that generates an oily, unpurifiable intermediate creates a fragile manufacturing link where small variations in reaction parameters can lead to batch failures. Consequently, the conventional method represents a significant risk factor for continuous commercial production, prompting the industry to seek alternative routes that offer better physical properties for the key intermediates involved in the synthesis.
The Novel Approach
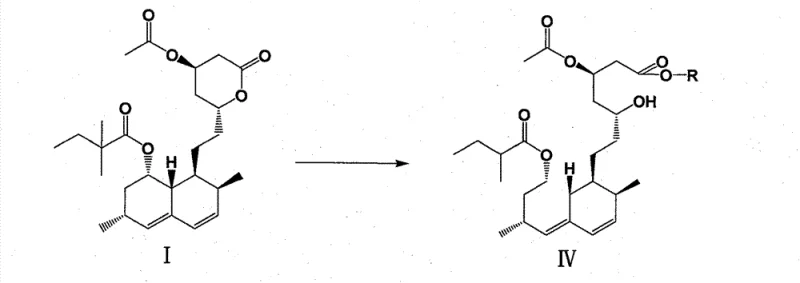
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes a selective alcoholysis strategy to generate a new intermediate, Formula IV, which serves as a superior precursor to the final ammonium salt. This method involves reacting 4-acetyl simvastatin (Formula I) with methanol or ethanol under acidic catalysis to open the lactone ring while critically retaining the acetyl protecting group at the 4-position. This selectivity is the cornerstone of the invention, as it prevents the formation of the unstable, multi-hydroxyl species seen in the old route. The resulting intermediate IV is formed rapidly, typically within 2 hours, representing a drastic reduction in processing time compared to the 16-hour requirement of the conventional method. Moreover, intermediate IV exhibits physical properties that are far more conducive to purification and handling, setting the stage for a high-yield conversion to the final product.
The strategic retention of the acetyl group during the initial alcoholysis step allows for a cleaner reaction profile. By avoiding the simultaneous deprotection of the 4-hydroxyl group, the process minimizes the generation of polar by-products that are notoriously difficult to separate from the target molecule. This chemical elegance translates directly into operational excellence; the reaction can be conducted under mild conditions, typically between 0°C and 30°C, using common solvents like methanol or ethanol which act as both reagent and solvent. The ability to drive the reaction to completion quickly, as monitored by TLC, ensures high throughput. For supply chain heads, this means a more predictable production schedule and reduced reactor occupancy time. The novel approach effectively decouples the ring-opening from the deprotection, allowing each step to be optimized independently, thereby maximizing the overall efficiency of the simvastatin ammonium salt manufacturing process.
Mechanistic Insights into Selective Alcoholysis and Hydrolysis
The core chemical innovation of this process lies in the differential reactivity of the ester functionalities present in 4-acetyl simvastatin. Under the influence of an acid catalyst such as p-toluenesulfonic acid or hydrogen chloride, the lactone ring undergoes nucleophilic attack by the alcohol solvent (methanol or ethanol). Mechanistically, the carbonyl carbon of the lactone is more susceptible to alcoholysis under these specific conditions than the acetate ester at the 4-position, or the kinetics are tuned such that the lactone opens preferentially. This results in the formation of the hydroxy-ester intermediate IV, where the side chain is now an open methyl or ethyl ester, but the 4-position remains protected as an acetate. This protection is vital; it maintains a level of lipophilicity and structural rigidity that prevents the molecule from becoming too polar or prone to intermolecular interactions that lead to oil formation. The reaction stoichiometry is carefully controlled, with the alcohol used in a large excess (30 to 50 molar equivalents) to drive the equilibrium towards the open-chain ester product.
Following the isolation of intermediate IV, the subsequent conversion to simvastatin ammonium salt (Formula V) involves a base-mediated hydrolysis. In this step, the reaction conditions are adjusted to cleave both the side-chain ester and the 4-acetyl protecting group simultaneously. Using a strong base like sodium hydroxide or potassium hydroxide in a water-miscible organic solvent facilitates the saponification of both ester linkages. The mechanism proceeds through the formation of a tetrahedral intermediate at both carbonyl centers, eventually yielding the di-hydroxy carboxylate salt. Subsequent acidification to pH 3-4 protonates the carboxylate to the free acid, which is then extracted into an organic phase. Finally, treatment with ammonia or ammonium hydroxide neutralizes the acid to form the ammonium salt. This final crystallization step is highly effective because the precursor acid is of high purity, having been derived from the clean intermediate IV. The crystallization of the ammonium salt from the organic phase allows for the exclusion of remaining inorganic salts and polar impurities, ensuring the final product meets stringent purity specifications required for API synthesis.
How to Synthesize Simvastatin Ammonium Salt Efficiently
The practical implementation of this synthetic route requires careful attention to reaction parameters to maximize the benefits of the novel intermediate strategy. The process begins with the dissolution of 4-acetyl simvastatin in a molar excess of methanol or ethanol under an inert atmosphere, typically nitrogen, to prevent oxidative degradation. An acid catalyst is introduced to initiate the transesterification, and the mixture is stirred at ambient temperature until thin-layer chromatography confirms the consumption of the starting lactone. Upon completion, the solvent is removed under reduced pressure, and the residue is partitioned between toluene and aqueous bicarbonate to remove acidic impurities and catalyst residues. The organic layer is dried and concentrated to yield intermediate IV as a light yellow oil or solid, depending on the specific ester formed. This intermediate is then subjected to hydrolysis using aqueous alkali, followed by the precise pH adjustments and ammonification steps described in the mechanistic section to isolate the final ammonium salt.
- Perform acid-catalyzed alcoholysis on 4-acetyl simvastatin (I) using methanol or ethanol to generate the stable intermediate 4-acetyl simvastatin ester (IV).
- Conduct alkaline hydrolysis on intermediate IV in a water-soluble organic solvent to open the ester and remove the acetyl protecting group.
- Acidify the reaction mixture to pH 3-4, extract, and subsequently react with ammonia to crystallize the high-purity simvastatin ammonium salt (V).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthetic route offers profound advantages for organizations managing the supply of pharmaceutical intermediates. The primary value driver is the significant reduction in processing time and the elimination of purification bottlenecks associated with oily intermediates. By replacing a 16-hour reaction with a 2-hour process, manufacturers can dramatically increase reactor turnover rates, effectively expanding capacity without capital investment in new equipment. This efficiency gain directly correlates to cost reduction in API manufacturing, as labor, energy, and solvent usage are minimized. Furthermore, the ability to produce a high-purity intermediate that facilitates easy crystallization of the final salt reduces the need for expensive chromatographic purification or multiple recrystallization cycles, streamlining the entire production workflow.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high yield and selectivity of the alcoholysis step. Conventional methods suffer from material loss due to the inability to purify the oily intermediate VIII, often resulting in suboptimal yields in the final ammoniation step. In contrast, the new route achieves yields exceeding 90% for the intermediate and maintains high efficiency through to the final salt. This improvement in mass balance means that less raw material is required to produce the same amount of finished good. Additionally, the use of commodity chemicals like methanol, ethanol, and sodium hydroxide ensures that reagent costs remain low and stable. The elimination of extended reaction times also reduces utility costs, contributing to a leaner cost structure that enhances competitiveness in the global market for generic statin intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is critical for downstream API manufacturers, and this technology significantly de-risks the supply chain. The robustness of the reaction conditions—operating effectively at room temperature and atmospheric pressure—means that the process is less sensitive to minor fluctuations in plant operations compared to more sensitive cryogenic or high-pressure reactions. The high purity of intermediate IV ensures that the final crystallization of the ammonium salt is consistent and reproducible, reducing the incidence of batch failures or out-of-specification results. For procurement managers, this reliability translates to more accurate forecasting and the ability to maintain lower safety stock levels. The simplified workup procedure also shortens the overall cycle time from raw material intake to finished goods shipment, allowing for faster response to market demand spikes.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this route is inherently scalable due to its reliance on standard unit operations such as stirring, distillation, and filtration. The solvents used are water-soluble and easily recoverable, facilitating efficient solvent recycling programs that align with green chemistry principles. The reduction in reaction time and the avoidance of complex purification steps also lead to a decrease in waste generation, specifically reducing the volume of mother liquors and spent adsorbents typically associated with chromatography. This environmental benefit is increasingly important as regulatory scrutiny on pharmaceutical manufacturing waste intensifies. The process design supports a sustainable manufacturing model that minimizes the environmental footprint while maximizing output, making it an attractive option for companies committed to corporate social responsibility and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this simvastatin ammonium salt synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the specific advantages in terms of purity, yield, and operational simplicity that distinguish this method from prior art.
Q: What is the primary advantage of intermediate IV over intermediate VIII in simvastatin synthesis?
A: Intermediate IV retains the 4-acetyl protecting group, resulting in a stable, high-yield product that avoids the oily, impure nature of intermediate VIII found in conventional routes.
Q: How does this novel process impact the purity of the final simvastatin ammonium salt?
A: By utilizing a selective alcoholysis strategy that minimizes side reactions and allows for effective purification prior to ammonification, the process achieves significantly higher purity levels suitable for API production.
Q: Is this synthetic route scalable for industrial manufacturing?
A: Yes, the reaction conditions are mild (0-30°C) and utilize common solvents like methanol and ethanol, making the process highly adaptable for large-scale commercial production without requiring extreme parameters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Simvastatin Ammonium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful production of life-saving medications. Our technical team has thoroughly analyzed the synthetic pathway described in CN101575286B and possesses the expertise to execute this advanced chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory scale to industrial manufacturing is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and quality of every batch. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance guarantees that our simvastatin ammonium salt meets the exacting standards required by global regulatory agencies.
We invite potential partners to engage with us to explore how this optimized synthetic route can enhance your supply chain efficiency and product quality. Our team is prepared to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this novel intermediate strategy for your specific production volumes. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a reliable partner dedicated to advancing pharmaceutical manufacturing through innovation, quality, and unwavering supply chain support.
