Scalable Green Synthesis of Beta-Hydroxy Organoboron Intermediates via Chitosan-Copper Catalysis
Introduction to Advanced Organoboron Synthesis Technology
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for sustainable and efficient synthetic methodologies. A pivotal advancement in this domain is documented in patent CN110590819B, which introduces a novel preparation method for organoboron compounds and their subsequent conversion into beta-hydroxy derivatives. This technology addresses critical bottlenecks in the synthesis of Morita-Baylis-Hillman organoboron compounds, which serve as versatile building blocks for complex drug molecules and natural products. By leveraging a chitosan solid-supported copper catalyst, the process achieves high reaction activity under remarkably mild conditions, specifically utilizing water as the sole solvent. This innovation not only streamlines the synthetic pathway but also aligns perfectly with the rigorous environmental standards demanded by modern pharmaceutical and agrochemical industries, offering a robust solution for the production of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized organoboron compounds has relied heavily on homogeneous catalytic systems that present substantial operational and environmental challenges. Traditional literature methods often necessitate the use of expensive and volatile organic solvents, such as tert-butanol, which increase both the raw material costs and the complexity of solvent recovery systems. Furthermore, these conventional routes typically require strong bases like lithium tert-butoxide to drive the reaction, introducing significant safety hazards and generating corrosive waste streams that are difficult to treat. Perhaps most critically for pharmaceutical applications, homogeneous metal catalysts are notoriously difficult to remove completely from the final product, often leaving behind trace metal residues that fail to meet stringent regulatory purity specifications. This necessitates additional, costly purification steps such as scavenging or extensive chromatography, which drastically reduce overall process efficiency and yield.
The Novel Approach
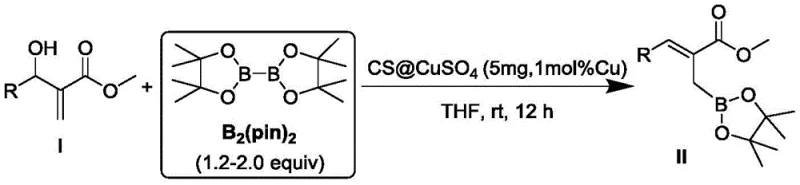
In stark contrast, the methodology disclosed in the patent utilizes a functionalized chitosan immobilized copper catalyst, representing a paradigm shift towards greener and more economical manufacturing. This heterogeneous catalytic system operates effectively in pure water at room temperature, eliminating the need for flammable organic solvents and energy-intensive heating or cooling protocols. The chitosan support provides a biocompatible and biodegradable matrix that not only stabilizes the copper species but also facilitates easy separation of the catalyst from the reaction mixture via simple filtration. This approach allows for the direct boron addition to substrates containing various substituents, including phenyl, halogen, and ester groups, with exceptional selectivity. The ability to perform this transformation under such mild conditions while avoiding strong bases significantly simplifies the downstream processing workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Chitosan-Supported Copper Catalysis
The efficacy of this synthetic route lies in the unique interaction between the chitosan support and the copper active sites, which creates a highly reactive microenvironment for the borylation reaction. Mechanistically, the process begins with the adsorption of both the alpha,beta-unsaturated carbonyl substrate and the diboron pinacol ester onto the surface of the chitosan-supported copper catalyst. The copper centers coordinate with the diboron reagent to form a transient composite metal complex, which activates the boron-boron bond for nucleophilic attack. This activation is crucial for overcoming the kinetic barriers associated with borylation under neutral conditions. The reaction proceeds through a highly organized six-membered ring transition state, where the boron group connected to the copper is selectively transferred to the substrate via a 1,4-addition mechanism. This specific pathway ensures the formation of the desired Morita-Baylis-Hillman organoboron structure with high regioselectivity, minimizing the formation of unwanted by-products that often plague less controlled homogeneous reactions.
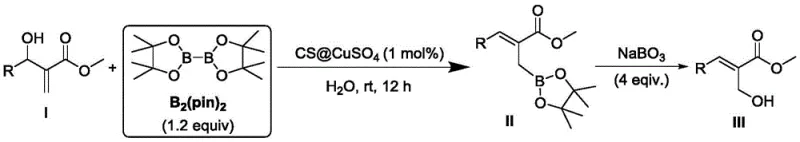
Following the initial borylation, the process offers a seamless one-pot strategy for converting the organoboron intermediate into valuable beta-hydroxy compounds, which are prevalent motifs in bioactive natural products. Without isolating the intermediate organoboron species, an oxidant such as sodium perborate tetrahydrate is introduced directly into the reaction system. This oxidative workup converts the carbon-boron bond into a carbon-oxygen bond with remarkable efficiency. The compatibility of the chitosan-copper system with this oxidative step is a testament to its robustness, as the catalyst does not interfere with the oxidation nor does it degrade under the reaction conditions. This tandem sequence effectively merges two distinct synthetic transformations into a single operational unit, thereby reducing the total number of processing steps, minimizing material handling, and maximizing the overall atom economy of the synthesis.
How to Synthesize Beta-Hydroxy Organoboron Compounds Efficiently
The practical implementation of this technology involves a straightforward protocol that is easily adaptable to both laboratory and pilot-scale operations. The synthesis begins with the preparation of the aqueous catalyst suspension, followed by the sequential addition of substrates under ambient conditions. The reaction progress is monitored to ensure complete conversion before proceeding to the filtration and oxidation stages. This streamlined workflow minimizes the requirement for specialized equipment, as standard glassware or stainless steel reactors can be utilized without the need for inert atmosphere gloveboxes often required for sensitive organometallic chemistry. For detailed operational parameters, stoichiometry, and purification techniques, please refer to the standardized guide below.
- Prepare the reaction system by adding chitosan-supported copper sulfate catalyst to water and stirring at room temperature to activate the heterogeneous surface.
- Sequentially add the alpha,beta-unsaturated carbonyl substrate and pinacol diboron ester to the aqueous suspension, maintaining a molar ratio favoring the boron reagent.
- Stir the mixture at room temperature for 10-14 hours, then filter to recover the catalyst. Add sodium perborate to the filtrate for one-pot oxidation to the beta-hydroxy product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chitosan-supported copper catalysis technology offers compelling strategic advantages that extend far beyond simple chemical yield. The shift from homogeneous to heterogeneous catalysis fundamentally alters the cost structure of producing complex organoboron intermediates. By eliminating the dependency on expensive, anhydrous organic solvents and strong bases, the raw material costs are significantly reduced. Moreover, the ability to recycle the catalyst multiple times without significant loss of activity means that the effective cost of the metal catalyst per kilogram of product is drastically lowered. This reduction in consumable costs, combined with the simplification of waste treatment due to the use of water, results in substantial cost savings in fine chemical manufacturing, allowing for more competitive pricing in the global market.
- Cost Reduction in Manufacturing: The elimination of costly organic solvents and the reduction in catalyst loading directly translate to lower variable costs per batch. Since the catalyst can be recovered and reused, the expenditure on precious metal salts is amortized over multiple production runs, leading to a more favorable margin structure. Additionally, the avoidance of strong bases reduces the corrosion burden on reactor vessels and piping, extending the lifespan of capital equipment and lowering maintenance expenditures over the long term.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as chitosan and common copper salts, are abundant and readily available from diverse global suppliers, mitigating the risk of supply chain disruptions associated with specialized reagents. The robustness of the reaction conditions, which tolerate a wide range of functional groups and operate at room temperature, ensures consistent production output regardless of minor fluctuations in utility conditions. This reliability is critical for maintaining continuous supply lines to downstream pharmaceutical clients who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction system makes it inherently scalable, as the separation of the catalyst via filtration is a unit operation that translates seamlessly from grams to tons. Furthermore, the use of water as a solvent and the generation of biodegradable chitosan waste align perfectly with increasingly stringent environmental regulations. This green profile simplifies the permitting process for new manufacturing lines and reduces the liability associated with hazardous waste disposal, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the operational feasibility and performance metrics of the chitosan-copper catalytic system. Understanding these details is essential for technical teams evaluating the integration of this process into existing manufacturing workflows.
Q: Can the chitosan-supported copper catalyst be recycled without losing activity?
A: Yes, the patent data demonstrates that the heterogeneous catalyst can be recovered via simple filtration and reused for at least five consecutive cycles with negligible loss in catalytic activity or yield.
Q: What are the environmental advantages of this synthesis route compared to traditional methods?
A: This method replaces toxic organic solvents like t-butanol with pure water and eliminates the need for strong, polluting bases like lithium t-butoxide, significantly reducing hazardous waste generation.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Absolutely. The use of a heterogeneous catalyst allows for easy separation, and the mild room-temperature conditions in water make the process inherently safer and easier to scale up for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Hydroxy Organoboron Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chitosan-supported copper catalysis technology in advancing the synthesis of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry are realized at an industrial level. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of organoboron compounds we produce. We are committed to delivering products that not only meet but exceed the quality expectations of global innovator companies, supporting your drug development pipelines with reliable and sustainable supply solutions.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this technology can optimize your bill of materials. Please contact us today to request specific COA data for our catalog of beta-hydroxy organoboron compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your time-to-market with superior chemistry and supply chain excellence.
