Advanced One-Step Synthesis of Alpha,beta-Diamino Acid Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks efficient pathways to construct complex chiral scaffolds, particularly those found in bioactive natural products and potent therapeutics. Patent CN102432485B introduces a groundbreaking methodology for the synthesis of alpha,beta-diamino acid derivatives, a critical structural motif prevalent in anticancer agents like bleomycins and various antibiotics. This innovation leverages a sophisticated four-component coupling reaction that merges diazo compounds, anilines, and aldehyde esters in a single operational step. By employing a dual catalytic system comprising a metal Lewis acid and a chiral phosphoric acid, the process achieves exceptional levels of stereocontrol without the need for harsh conditions or extensive purification sequences. This represents a paradigm shift from traditional multi-step syntheses, offering a robust platform for generating diverse libraries of chiral intermediates with high atom economy.
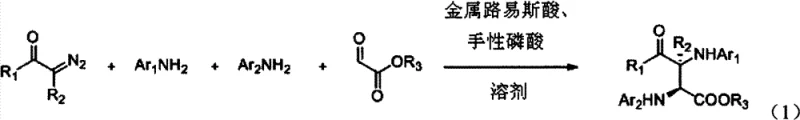
For procurement managers and supply chain directors, the implications of this technology are profound. The ability to access high-purity pharmaceutical intermediates through a convergent synthetic route translates directly into reduced lead times and lower manufacturing costs. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such advanced catalytic processes allows us to optimize production schedules and ensure consistent quality for our global partners. The reaction's tolerance for a wide range of substrates, including various substituted anilines and diazo ketones, ensures flexibility in sourcing raw materials, thereby mitigating supply chain risks associated with specialized reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alpha,beta-diamino acid skeletons has been fraught with synthetic challenges that hinder efficient commercial production. Traditional routes often rely on linear sequences involving multiple protection and deprotection steps to manage the reactivity of the amine and carboxylic acid functionalities. These lengthy protocols not only increase the consumption of solvents and reagents but also result in significant material loss at each stage, drastically lowering the overall yield. Furthermore, achieving high stereoselectivity in these classical approaches frequently requires stoichiometric amounts of chiral auxiliaries or resolution steps, which are both cost-prohibitive and environmentally burdensome. The accumulation of impurities from side reactions in multi-step processes complicates downstream purification, often necessitating expensive chromatographic separations that are difficult to scale industrially.
The Novel Approach
In stark contrast, the methodology disclosed in CN102432485B utilizes a direct, one-pot multicomponent reaction that bypasses the inefficiencies of linear synthesis. By combining four distinct components—diazo compound, two equivalents of aniline (or one aniline and one imine intermediate), and an aldehyde ester—the reaction constructs two new carbon-nitrogen bonds and establishes two adjacent chiral centers simultaneously. This convergent strategy maximizes atom economy, ensuring that the majority of the starting material mass is incorporated into the final product. The use of a cooperative catalytic system allows the reaction to proceed under mild temperatures ranging from 0°C to 40°C, eliminating the need for energy-intensive heating or cryogenic cooling. This streamlined approach not only simplifies the operational workflow but also significantly reduces the environmental footprint by minimizing waste generation and solvent usage.
Mechanistic Insights into Rh-Catalyzed Multicomponent Coupling
The success of this transformation hinges on the intricate interplay between the metal Lewis acid and the chiral phosphoric acid catalyst. The reaction initiates with the decomposition of the diazo compound by the rhodium catalyst to generate a reactive metal carbene species. This electrophilic intermediate is then intercepted by the first equivalent of aniline to form an ammonium ylide. Simultaneously, the chiral phosphoric acid activates the aldehyde ester and the second aniline molecule, facilitating the formation of a reactive imine or hemiaminal intermediate. The ammonium ylide subsequently attacks this activated species in a highly stereocontrolled manner, dictated by the chiral environment provided by the phosphoric acid ligand. This dual activation mode ensures that the nucleophilic attack occurs with precise facial selectivity, leading to the observed high diastereoselectivity and enantioselectivity.
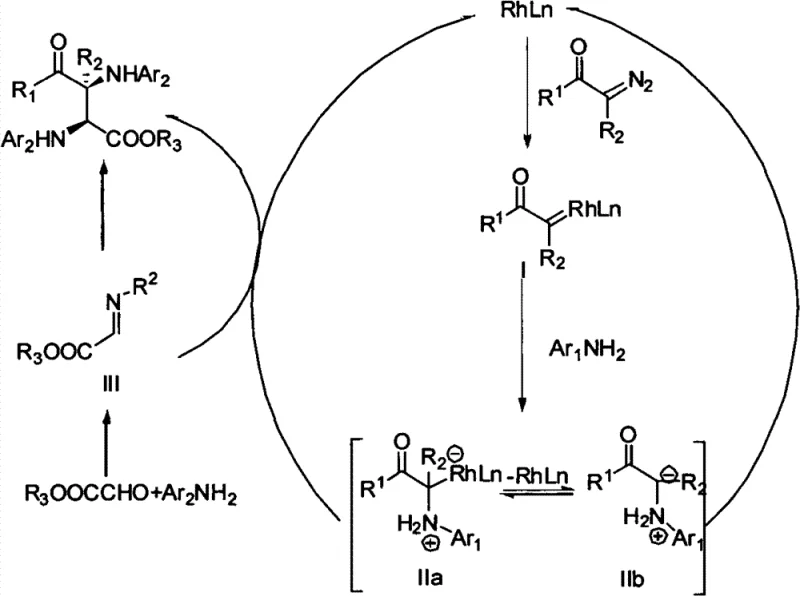
Understanding the mechanistic pathway is crucial for R&D directors focused on impurity control and process optimization. The specific choice of the chiral phosphoric acid, derived from BINOL with bulky substituents such as triphenylsilyl or 3,5-bis(trifluoromethyl)phenyl groups, plays a pivotal role in shielding one face of the reacting species. This steric hindrance prevents the formation of unwanted diastereomers and enantiomers, thereby simplifying the impurity profile of the crude reaction mixture. Moreover, the use of molecular sieves as a water scavenger is essential to drive the equilibrium towards product formation by removing water generated during the imine formation step. This attention to mechanistic detail ensures that the process remains robust and reproducible, even when scaling up to kilogram quantities, providing a reliable foundation for the synthesis of complex API intermediates.
How to Synthesize Alpha,beta-Diamino Acid Derivatives Efficiently
The practical execution of this synthesis is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The protocol involves dissolving the aldehyde ester, aniline, molecular sieves, metal Lewis acid, and chiral phosphoric acid in a suitable organic solvent such as toluene or dichloromethane. After a brief stirring period to ensure homogeneity and catalyst activation, the diazo compound solution is added dropwise over the course of one hour to control the exotherm and concentration of the reactive carbene species. Following the addition, the reaction is allowed to stir for a short period to reach completion. The simplicity of the workup, typically involving filtration of the molecular sieves followed by column chromatography, makes this method highly attractive for rapid process development and scale-up activities.
- Prepare the reaction mixture by dissolving ethyl glyoxylate, aniline, molecular sieves, metal Lewis acid (e.g., Rh2(OAc)4), and chiral phosphoric acid in an organic solvent like toluene at 0-40°C.
- Slowly add the solution of the diazo compound (e.g., diazoacetophenone) in organic solvent to the reaction system over a period of 1 hour while maintaining stirring.
- Continue stirring the reaction mixture for an additional 20 minutes after the addition is complete, then purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers tangible benefits that align with the strategic goals of cost reduction and supply chain resilience. The consolidation of multiple synthetic steps into a single operation drastically reduces the man-hours required for production, leading to significant labor cost savings. Additionally, the high yields and selectivity reported in the patent minimize the need for extensive recycling of unreacted materials or complex purification trains, further enhancing the overall process efficiency. For procurement teams, the reliance on commodity chemicals like anilines and glyoxylates, rather than exotic custom synthons, ensures a stable and cost-effective supply of raw materials. This stability is critical for maintaining consistent pricing and avoiding disruptions in the manufacturing schedule.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps results in a substantial decrease in solvent consumption and waste disposal costs. By avoiding the use of stoichiometric chiral auxiliaries and replacing them with catalytic amounts of chiral phosphoric acid, the material cost per kilogram of product is significantly lowered. This economic efficiency allows for more competitive pricing strategies in the global market for pharmaceutical intermediates, providing a distinct advantage in tender negotiations and long-term supply agreements.
- Enhanced Supply Chain Reliability: The broad substrate scope of this reaction means that alternative starting materials can be easily sourced if primary suppliers face shortages. The mild reaction conditions reduce the risk of equipment failure or safety incidents associated with high-pressure or high-temperature processes, ensuring uninterrupted production cycles. Furthermore, the robustness of the catalytic system allows for flexibility in batch sizing, enabling manufacturers to respond quickly to fluctuating demand without compromising product quality or delivery timelines.
- Scalability and Environmental Compliance: The one-pot nature of the reaction simplifies the engineering requirements for scale-up, as there is no need for complex transfer lines or multiple reactor vessels for intermediate steps. The reduced solvent usage and higher atom economy contribute to a greener manufacturing process, helping companies meet increasingly stringent environmental regulations and sustainability goals. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible and forward-thinking partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this four-component reaction over traditional methods?
A: This method offers high atom economy and eliminates the need for multiple protection-deprotection steps, significantly reducing waste and processing time compared to conventional linear syntheses.
Q: What level of stereoselectivity can be achieved with this catalytic system?
A: The dual catalytic system utilizing rhodium and chiral phosphoric acid achieves excellent diastereoselectivity (dr > 90:10) and enantioselectivity (ee up to 95%), ensuring high optical purity for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (0-40°C) with readily available starting materials and simple workup procedures, making it highly amenable to commercial scale-up and continuous manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,beta-Diamino Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN102432485B for accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into viable industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality and consistency makes us a trusted partner for multinational pharmaceutical companies seeking reliable sources for complex chiral building blocks.
We invite you to collaborate with us to explore the full potential of this synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and innovation in your supply chain. Together, we can overcome the challenges of complex synthesis and bring life-saving therapies to market faster and more economically.
