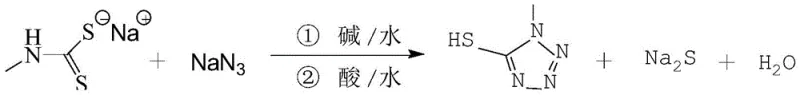
Advanced Aqueous Synthesis of 1-Methyl-5-Mercapto Tetrazole for Commercial Cephalosporin Production
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antibiotic intermediates, particularly for the vast family of cephalosporins. Patent CN112094244A introduces a transformative synthesis method for 1-methyl-5-mercapto tetrazole (1-MT), a pivotal side-chain intermediate used in the manufacture of third, fourth, and fifth-generation cephalosporin antibiotics such as cefoperazone sodium and cefmenoxime hydrochloride. This innovation addresses long-standing inefficiencies in heterocyclic chemistry by shifting from hazardous organic solvent systems to a green, aqueous-phase protocol. By utilizing sodium methylamino dithio carboxylate and sodium azide as primary feedstocks, the disclosed technology achieves exceptional conversion rates while maintaining rigorous control over impurity profiles. For global supply chain leaders, this represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering high-volume batches with consistent quality. The transition to this methodology not only enhances the environmental profile of the manufacturing process but also drastically simplifies downstream processing, thereby reducing the overall cost burden associated with solvent recovery and waste disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 1-methyl-5-mercapto tetrazole has relied heavily on the reaction between methyl isothiocyanate and sodium azide, a pathway fraught with significant technical and economic drawbacks. Conventional protocols often necessitate the use of volatile organic solvents such as ethanol or require complex biphasic systems involving diethyl ether for extraction, which introduces severe safety hazards and increases operational expenditures. Literature and prior art indicate that these traditional methods frequently struggle with poor reaction kinetics, resulting in isolated yields that hover precariously between 28% and 40%. Furthermore, the reliance on organic bases or phase transfer catalysts complicates the purification landscape, often leaving behind residual amines that are difficult to remove and can compromise the safety profile of the final antibiotic drug substance. The necessity for multiple extraction steps and concentration under vacuum not only extends the production cycle time but also amplifies the risk of product degradation due to thermal stress, making cost reduction in API intermediate manufacturing a challenging objective for procurement teams managing tight margins.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a fundamentally different starting material, sodium methylamino dithio carboxylate, to drive the cyclization reaction in a purely aqueous environment. This strategic shift eliminates the need for flammable organic solvents during the critical reaction phase, replacing them with water, which acts as both the solvent and a heat sink for the exothermic azide chemistry. The process employs inexpensive inorganic bases like sodium hydroxide or sodium carbonate to catalyze the transformation, ensuring that the reaction medium remains homogeneous and highly reactive throughout the reflux period. By avoiding the use of ether extractions and instead utilizing a controlled precipitation and filtration strategy, the new method streamlines the workflow significantly. The subsequent recrystallization step utilizes a specific toluene-water mixture, which is far more manageable and recyclable than the solvent cocktails required in older methods. This modernization of the synthetic route directly translates to enhanced supply chain reliability, as the raw materials are commodity chemicals with stable pricing, and the process itself is inherently safer and more scalable for commercial operations.
Mechanistic Insights into Aqueous Phase Heterocyclic Cyclization
The core of this technological advancement lies in the unique reactivity of the dithiocarboxylate anion towards the azide ion under basic aqueous conditions. Mechanistically, the reaction proceeds through a nucleophilic attack where the azide ion targets the electrophilic carbon center of the dithiocarboxylate group, initiating a cascade that leads to the formation of the tetrazole ring while expelling sulfide species as sodium sulfide. The presence of the inorganic base is crucial not only for maintaining the solubility of the reactants but also for stabilizing the intermediate species against premature decomposition. Operating at temperatures between 85°C and 95°C provides the necessary activation energy to overcome the kinetic barrier of ring closure without inducing the thermal runaway risks associated with organic solvents. This controlled thermal environment ensures that the reaction proceeds to near-completion, as evidenced by the high conversion rates observed in the experimental data, effectively minimizing the presence of unreacted starting materials that would otherwise act as difficult-to-remove impurities in the final crystalline lattice.
Impurity control is further refined through a precise pH manipulation strategy during the workup phase. After the reflux is complete, the reaction mixture is cooled and filtered to remove bulk inorganic salts, after which the filtrate is carefully neutralized with a protonic acid such as hydrochloric acid or sulfuric acid to a specific pH range of 6 to 7. This narrow pH window is critical; it ensures the protonation of the tetrazole thiol group to induce precipitation while keeping other ionizable impurities in the solution phase. The subsequent recrystallization from a toluene-water system acts as a final polishing step, leveraging the differential solubility of the target molecule versus organic byproducts. The toluene fraction effectively dissolves non-polar organic impurities, while the water fraction retains polar inorganic salts, resulting in a finished product that meets the stringent purity specifications required for parenteral antibiotic formulations. This dual-solvent recrystallization is a key factor in achieving the high-purity 1-methyl-5-mercapto tetrazole necessary for regulatory compliance.
How to Synthesize 1-Methyl-5-Mercapto Tetrazole Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize the benefits of the aqueous system. The process begins with the dissolution of sodium methylamino dithio carboxylate in water, followed by the sequential addition of the inorganic base catalyst and sodium azide under vigorous stirring to ensure homogeneity. The reaction mixture is then heated to reflux conditions, typically maintained between 85°C and 95°C for a duration of 14 to 16 hours, allowing sufficient time for the cyclization to reach equilibrium. Upon completion, the protocol dictates a controlled cooling sequence to below 10°C to facilitate the initial separation of solids, followed by a reheating step to 30-40°C before neutralization, a nuanced operation designed to optimize crystal growth and filtration characteristics. For a comprehensive breakdown of the specific operational parameters and safety checks required for this procedure, please refer to the standardized synthesis guide below.
- React sodium methylamino dithio carboxylate with sodium azide in water using an inorganic base catalyst at 85-95°C for 14-16 hours.
- Cool the reaction mixture, filter, heat the filtrate, and neutralize with protonic acid to pH 6-7 to precipitate the crude product.
- Recrystallize the crude product using a mixed solvent of toluene and water (65-75% toluene) to obtain high-purity finished product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers compelling economic and logistical benefits that extend beyond simple yield improvements. The transition to a water-based reaction system fundamentally alters the cost structure of production by eliminating the massive volumes of organic solvents previously required for reaction media and extraction. This reduction in solvent consumption directly lowers raw material costs and significantly decreases the expense associated with solvent recovery distillation and hazardous waste incineration. Furthermore, the use of commodity inorganic chemicals like sodium hydroxide and sodium azide ensures a stable supply chain that is less susceptible to the price volatility often seen with specialized organic reagents. The simplified workup procedure, which relies on filtration and crystallization rather than complex liquid-liquid extractions, reduces the labor hours and equipment occupancy time per batch, thereby increasing the overall throughput capacity of the manufacturing facility without requiring capital investment in new hardware.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents like ethanol and diethyl ether from the primary reaction and extraction steps results in substantial cost savings. By shifting to water as the primary medium, the process removes the need for explosion-proof reactors and complex solvent recovery trains, leading to lower utility costs and reduced maintenance overhead. Additionally, the high yield achieved minimizes the loss of valuable starting materials, ensuring that the cost per kilogram of the final active intermediate is significantly optimized compared to legacy methods that suffered from poor mass balance.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as sodium methylamino dithio carboxylate and sodium azide are widely available bulk chemicals with established global supply networks. This availability reduces the risk of production stoppages due to raw material shortages, a common issue with niche organic precursors. The robustness of the aqueous chemistry also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain fluctuations and ensuring consistent delivery schedules for downstream antibiotic manufacturers.
- Scalability and Environmental Compliance: The inherent safety of the water-phase reaction makes this process ideal for commercial scale-up of complex heterocyclic intermediates, as it mitigates the fire and explosion risks associated with large-scale azide chemistry in organic solvents. From an environmental perspective, the generation of biodegradable inorganic salts as byproducts simplifies wastewater treatment, helping manufacturers meet increasingly strict environmental regulations without investing in advanced oxidation or specialized treatment facilities. This alignment with green chemistry principles enhances the corporate sustainability profile of the manufacturer, a growing requirement for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method outperforms traditional routes in terms of efficiency and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines or for procurement specialists negotiating supply agreements based on these improved specifications.
Q: How does the new aqueous method improve yield compared to traditional methyl isothiocyanate routes?
A: Traditional methods using methyl isothiocyanate often suffer from yields as low as 28-40% due to side reactions and difficult extraction processes. The patented aqueous method utilizing sodium methylamino dithio carboxylate achieves yields exceeding 93% by optimizing reaction kinetics in a homogeneous water phase and simplifying isolation.
Q: What are the safety advantages of using inorganic bases over organic amines in this synthesis?
A: The process replaces volatile and potentially toxic organic bases like triethylamine with stable inorganic bases such as sodium hydroxide or sodium carbonate. This eliminates the risk of amine contamination in the final API and simplifies waste treatment, as the byproducts are biodegradable inorganic salts rather than persistent organic pollutants.
Q: Why is the toluene-water recrystallization system critical for pharmaceutical grade purity?
A: The specific binary solvent system allows for the simultaneous removal of both organic impurities and inorganic salts. By tuning the toluene-to-water ratio between 65:35 and 75:25, the process ensures that the final 1-methyl-5-mercapto tetrazole meets the stringent purity specifications required for cephalosporin antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Methyl-5-Mercapto Tetrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the consistency and purity of key intermediates like 1-methyl-5-mercapto tetrazole are paramount to the success of your cephalosporin antibiotic production lines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify that every batch meets stringent purity specifications, adhering to the highest international standards for pharmaceutical intermediates. We understand the critical nature of your supply chain and are committed to providing a stable, high-quality source of this essential building block.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this greener, high-yield methodology. Please contact us today to request specific COA data from our recent pilot runs and to receive detailed route feasibility assessments that demonstrate our capability to support your long-term production goals with reliability and excellence.
