Scalable Manufacturing of 5-Trifluoromethyl-1,2,4-Triazoles via Novel Iodine-Promoted Cyclization
Scalable Manufacturing of 5-Trifluoromethyl-1,2,4-Triazoles via Novel Iodine-Promoted Cyclization
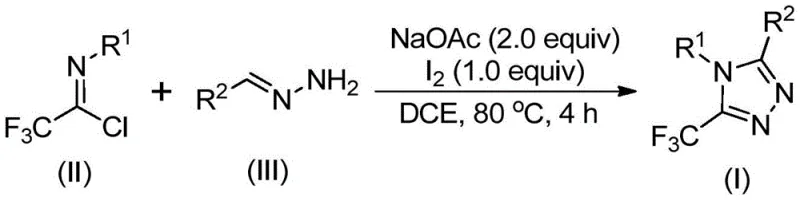
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses many historical limitations in heterocyclic synthesis. This technology leverages a non-metallic iodine-promoted strategy starting from inexpensive hydrazones and trifluoroethylimide chlorides, offering a streamlined pathway that eliminates the need for rigorous anhydrous or anaerobic environments. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for critical intermediates used in drugs like fluconazole and letrozole. The simplicity of the operation combined with the high designability of substrates makes this patent a cornerstone for modern fine chemical manufacturing strategies aiming for both efficiency and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of trifluoromethyl groups into nitrogen-containing heterocycles has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional literature reports primarily rely on two main strategies: the direct trifluoromethylation of pre-synthesized heterocycles or the coupling of trifluoromethyl synthons with suitable substrates. The former often necessitates the use of various specialized and costly trifluoromethylating reagents that can be hazardous to handle and difficult to source in bulk quantities. Furthermore, many of these conventional pathways demand strict reaction conditions, including absolute anhydrous and oxygen-free environments, which impose heavy infrastructure costs on manufacturing facilities. The reliance on transition metal catalysts in some older methods also introduces complications regarding heavy metal residue limits in final API products, requiring additional and expensive purification steps to meet stringent regulatory standards for human consumption.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in the patent utilizes a simple yet highly effective iodine-promoted cyclization strategy that fundamentally reshapes the economic landscape of triazole manufacturing. By employing readily available hydrazones and trifluoroethylimide chlorides as starting materials, the method bypasses the need for exotic reagents and complex catalytic systems. The reaction proceeds smoothly in common organic solvents like dichloroethane at moderate temperatures, eliminating the energy-intensive requirements associated with cryogenic conditions or high-pressure reactors. This shift not only drastically simplifies the operational workflow but also enhances the safety profile of the manufacturing process by avoiding toxic heavy metals. Consequently, this methodology offers a reliable agrochemical intermediate supplier pathway that is both cost-effective and environmentally friendlier, aligning perfectly with modern green chemistry initiatives while maintaining high yields and purity.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this synthesis is a sophisticated sequence of base-promoted transformations that ensure high regioselectivity and minimal by-product formation, a key concern for any R&D Director overseeing process development. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone and the trifluoroethylimide chloride, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization followed by a critical base-promoted oxidative iodination step mediated by elemental iodine. The resulting iodinated intermediate then participates in an intramolecular electrophilic substitution reaction, which drives the cyclization process forward. Finally, aromatization occurs to yield the stable 5-trifluoromethyl substituted 1,2,4-triazole core. This precise control over the reaction trajectory allows for the accommodation of various functional groups without compromising the integrity of the final heterocyclic scaffold, ensuring consistent quality across different batches.
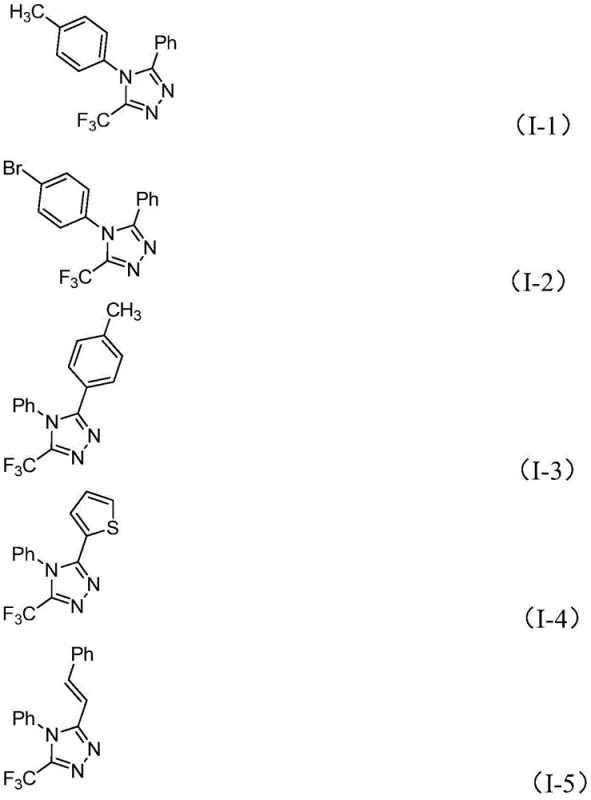
Impurity control is inherently built into this mechanism due to the mild nature of the iodine promoter and the specific reactivity of the chosen substrates. Unlike harsh fluorinating agents that might degrade sensitive functional groups on the aromatic rings, this system exhibits remarkable functional group tolerance, accommodating substituents such as methyl, methoxy, bromine, and nitro groups. The absence of transition metals means there is no risk of metal-catalyzed side reactions that often generate hard-to-remove impurities. Furthermore, the post-treatment process involves straightforward filtration and silica gel chromatography, which effectively removes inorganic salts and unreacted starting materials. This results in a high-purity OLED material or pharmaceutical intermediate profile that requires minimal downstream processing, thereby reducing the overall burden on quality control laboratories and accelerating the time to market for new drug candidates utilizing this scaffold.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis route in a production environment requires careful attention to stoichiometry and temperature control to maximize the benefits outlined in the patent documentation. The process begins with the dissolution of sodium acetate, trifluoroethylimide chloride, and the specific hydrazone derivative in a suitable aprotic organic solvent such as dichloroethane. The mixture is then heated to a temperature range of 80°C to 100°C and maintained for a period of 2 to 4 hours to ensure complete conversion of the initial intermediates. Following this initial phase, elemental iodine is introduced into the reaction system to drive the oxidative cyclization, with the reaction continuing for an additional 1 to 2 hours. Detailed standardized synthesis steps see the guide below for exact molar ratios and workup procedures tailored for commercial scale operations.
- Mix sodium acetate, trifluoroethylimide chloride, and hydrazone in an organic solvent such as dichloroethane.
- Heat the reaction mixture to 80°C and maintain for 2 to 4 hours to facilitate initial bond formation.
- Add elemental iodine to the system and continue reacting for 1 to 2 hours before post-treatment purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into tangible operational efficiencies and risk mitigation strategies that are crucial for maintaining competitive advantage. The elimination of expensive and toxic heavy metal catalysts removes a significant cost center associated with catalyst procurement, recovery, and the extensive testing required to certify low metal residues in final products. Additionally, the use of cheap and easily obtainable starting materials like aldehydes and aromatic amines ensures a stable supply chain that is less susceptible to the volatility often seen with specialized fluorinating reagents. The ability to operate without strict anhydrous conditions further reduces infrastructure costs, as facilities do not need to invest heavily in specialized drying equipment or inert gas manifolds. These factors collectively contribute to substantial cost savings and enhanced supply chain reliability for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The strategic avoidance of transition metal catalysts and specialized trifluoromethylating reagents leads to a significant reduction in raw material expenditure and waste disposal costs. By utilizing elemental iodine and sodium acetate, which are commodity chemicals, the process minimizes the financial burden associated with sourcing niche reagents that often carry high price premiums and long lead times. Furthermore, the simplified workup procedure reduces the consumption of solvents and stationary phases during purification, driving down the overall cost of goods sold. This economic efficiency makes the commercial scale-up of complex polymer additives or drug intermediates much more viable for mid-sized manufacturers looking to optimize their margins without sacrificing quality.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as substituted benzaldehydes and aromatic amines ensures that production schedules are not disrupted by raw material shortages. Since these precursors are produced globally in large volumes for various chemical industries, the risk of supply chain bottlenecks is drastically minimized compared to methods relying on proprietary or single-source reagents. This stability allows for more accurate forecasting and inventory management, enabling procurement teams to negotiate better terms with vendors. Reducing lead time for high-purity electronic chemical becomes feasible when the upstream supply chain is robust and diversified, ensuring continuous flow of materials to the production line.
- Scalability and Environmental Compliance: The process is explicitly designed to be easily expanded from gram-level laboratory synthesis to industrial large-scale production, offering a clear path for capacity growth as market demand increases. The absence of heavy metals simplifies environmental compliance, as there is no need for complex wastewater treatment systems dedicated to removing toxic metal ions. This aligns with increasingly stringent global environmental regulations, reducing the risk of fines or production halts due to non-compliance. The straightforward post-treatment involving filtration and chromatography is easily adaptable to continuous flow chemistry or large batch reactors, supporting cost reduction in fine chemical manufacturing through economies of scale.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and beneficial effects. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers provided reflect the objective capabilities of the method as described in the intellectual property documentation, ensuring transparency for all stakeholders involved in the decision-making process. Please review the specific technical responses below to gain deeper insights into the operational parameters and advantages.
Q: Does this synthesis method require anhydrous or anaerobic conditions?
A: No, unlike many conventional trifluoromethylation methods, this patented process operates effectively without strict anhydrous or oxygen-free conditions, significantly simplifying operational requirements.
Q: What are the advantages regarding catalyst usage in this protocol?
A: The method utilizes elemental iodine as a promoter rather than expensive or toxic heavy metal catalysts, which streamlines downstream purification and reduces environmental compliance burdens.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be easily expanded from gram-level laboratory synthesis to industrial large-scale production due to its simple operation and accessible raw materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-promoted cyclization technology for the production of high-value heterocyclic intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole meets the exacting standards required by the global pharmaceutical industry. We are committed to leveraging this patent's efficiency to deliver superior products that enhance your supply chain resilience.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to be your trusted partner in delivering high-quality chemical solutions efficiently and reliably.
