Advancing Pharmaceutical Intermediate Manufacturing via Green Electrocatalytic Dichlorination Strategies
Advancing Pharmaceutical Intermediate Manufacturing via Green Electrocatalytic Dichlorination Strategies
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainable and efficient synthetic methodologies, driven by the urgent need for greener processes in the pharmaceutical industry. A groundbreaking development in this sector is detailed in patent CN114411182A, which discloses a novel method for synthesizing alpha-carbonyl sulfoxide ylide dichlorides under electrocatalytic conditions. This technology represents a significant departure from traditional stoichiometric oxidation methods, leveraging electricity as a traceless reagent to drive the dichlorination of alpha-carbonyl sulfoxide ylides with remarkable precision. By utilizing a simple undivided cell setup with magnesium chloride as the chlorine source, this approach not only streamlines the synthetic route but also aligns perfectly with the principles of green chemistry, offering a robust solution for the production of high-value intermediates used in drug discovery and material science.
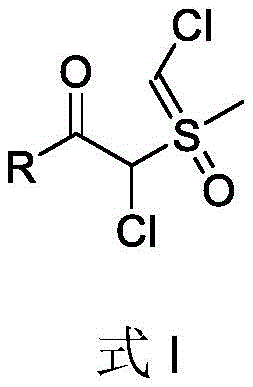
For R&D directors and process chemists, the structural versatility of the target molecules is paramount, as these dichlorinated ylides serve as critical building blocks for constructing complex molecular architectures. The general structure, as depicted in the patent, allows for a wide array of substituents on the carbonyl group, ranging from aromatic rings to heterocycles and aliphatic chains. This flexibility ensures that the methodology can be adapted to synthesize a diverse library of compounds, facilitating the rapid exploration of chemical space in medicinal chemistry programs. The ability to introduce two chlorine atoms regioselectively onto the ylide carbon skeleton opens up new avenues for downstream functionalization, making these intermediates indispensable for the synthesis of advanced therapeutic agents and specialized agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated sulfoxide derivatives has relied heavily on the use of stoichiometric amounts of hazardous chemical oxidants and chlorinating agents, which pose significant safety and environmental challenges. Traditional protocols often require harsh reaction conditions, including elevated temperatures and the use of corrosive reagents that generate substantial quantities of toxic waste streams. Furthermore, the handling of strong oxidants necessitates rigorous safety measures and specialized equipment, increasing the operational complexity and cost of manufacturing. The presence of residual heavy metals or toxic byproducts in the final product can also complicate purification processes, leading to lower overall yields and extended production timelines. These limitations have long hindered the efficient large-scale production of high-purity alpha-carbonyl sulfoxide ylide dichlorides, creating a bottleneck for supply chains dependent on these key intermediates.
The Novel Approach
In stark contrast, the electrocatalytic strategy outlined in the patent offers a transformative solution by replacing dangerous chemical oxidants with electrons, thereby fundamentally altering the reaction mechanism towards sustainability. As illustrated in the reaction scheme below, the process utilizes a constant current electrolysis setup where magnesium chloride serves as a benign chlorine source, and cesium carbonate acts as a base to facilitate the reaction. This method operates under exceptionally mild conditions, typically at room temperature (20°C), which minimizes energy consumption and reduces the risk of thermal runaway or decomposition of sensitive functional groups. The simplicity of the reaction setup, involving a graphite felt anode and a platinum cathode in a mixed solvent system of acetonitrile and water, underscores the practicality of this approach for both laboratory research and industrial application.
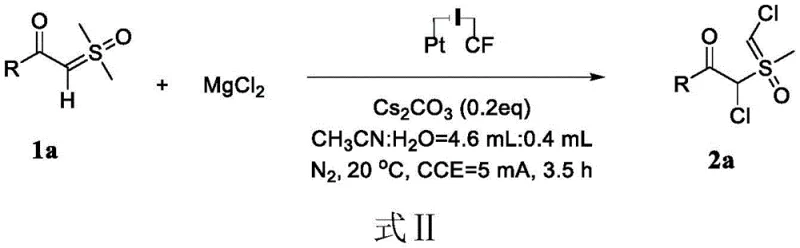
The operational simplicity extends to the workup procedure, where the crude product can be easily isolated by drying, filtration, and concentration, followed by standard silica gel column chromatography. This streamlined workflow eliminates the need for complex quenching steps associated with reactive oxidants, significantly reducing the time required to obtain pure products. Moreover, the electrochemical nature of the reaction allows for precise control over the oxidation potential, enhancing selectivity and minimizing the formation of unwanted side products. For procurement managers and supply chain heads, this translates to a more reliable and predictable manufacturing process that can be scaled up with greater confidence, ensuring a steady supply of critical intermediates without the logistical burdens associated with hazardous chemical storage and disposal.
Mechanistic Insights into Electrocatalytic Dichlorination
Understanding the mechanistic underpinnings of this electrocatalytic transformation is crucial for optimizing reaction parameters and ensuring consistent product quality in a commercial setting. The reaction proceeds through an anodic oxidation process where chloride ions from magnesium chloride are oxidized at the graphite felt anode to generate reactive chlorine species in situ. These electrogenerated chlorine species then react with the alpha-carbonyl sulfoxide ylide substrate, facilitated by the presence of cesium carbonate which likely assists in deprotonation or stabilization of intermediate species. The use of a platinum cathode ensures efficient reduction of protons or other species to complete the electrical circuit, maintaining the flow of current necessary for the continuous generation of the active chlorinating agent. This indirect electrolysis mechanism avoids direct oxidation of the organic substrate at the electrode surface, which can often lead to polymerization or over-oxidation, thereby preserving the integrity of the delicate ylide functionality.
Impurity control is another critical aspect where this electrochemical method excels, as the absence of external oxidants drastically reduces the complexity of the impurity profile. In traditional chemical oxidations, side reactions with the oxidant itself or its reduced forms can generate difficult-to-remove byproducts that compromise the purity of the final API intermediate. By contrast, the electrocatalytic route generates chlorine species in a controlled manner, limiting their concentration in the bulk solution and favoring the desired dichlorination pathway over competing reactions. The mild reaction temperature further suppresses thermal degradation pathways, ensuring that the product remains stable throughout the 3.5-hour reaction duration. For quality control teams, this means that the resulting alpha-carbonyl sulfoxide ylide dichlorides exhibit high purity levels with minimal need for extensive recrystallization or additional purification steps, directly impacting the cost of goods and time-to-market for downstream pharmaceutical products.
How to Synthesize Alpha-Carbonyl Sulfoxide Ylide Dichloride Efficiently
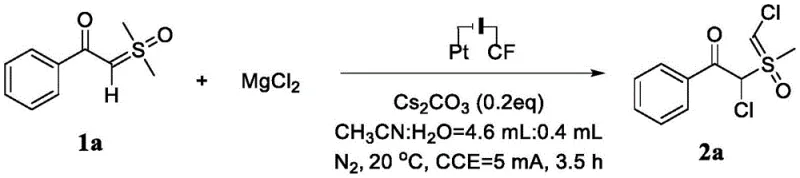
To implement this cutting-edge synthesis in a laboratory or pilot plant environment, operators must adhere to specific procedural guidelines that maximize yield and reproducibility while maintaining safety standards. The protocol involves dissolving the alpha-carbonyl sulfoxide ylide substrate along with magnesium chloride and cesium carbonate in a specific ratio of acetonitrile to water, creating a conductive medium essential for efficient electrolysis. The reaction is then driven by applying a constant current of 5 mA across the electrodes, a parameter that has been optimized to balance reaction rate with selectivity. As demonstrated in the specific example below for the synthesis of compound 2a, careful attention to the molar ratios of reagents and the composition of the solvent system is vital for achieving the reported yields of up to 78% across various substrates.
- Prepare the reaction mixture by combining alpha-carbonyl sulfoxide ylide, magnesium chloride (MgCl2), and cesium carbonate (Cs2CO3) in a molar ratio of 1: 2:0.2 within a reactor vessel.
- Add the solvent system consisting of acetonitrile and water in a volume ratio of 4.6 mL to 0.4 mL to ensure proper solubility and conductivity for the electrochemical process.
- Perform electrolysis using a graphite felt anode and platinum sheet cathode at a constant current of 5 mA and 20°C for 3.5 hours, followed by drying, filtration, and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrocatalytic technology offers profound benefits that extend beyond mere technical feasibility, addressing key pain points in cost management and supply chain resilience. The elimination of expensive and hazardous chemical oxidants represents a direct reduction in raw material costs, while the simplified workup procedure lowers labor and waste disposal expenses. Furthermore, the use of electricity as the primary driving force decouples the process from the volatility of chemical reagent markets, providing a more stable cost structure for long-term production contracts. For supply chain leaders, the ability to source simpler, commodity-grade reagents like magnesium chloride and cesium carbonate enhances supply security, reducing the risk of disruptions caused by the scarcity of specialized fine chemicals.
- Cost Reduction in Manufacturing: The transition to an electrocatalytic process inherently lowers the cost of goods sold by removing the need for stoichiometric oxidants, which are often among the most expensive components in a synthetic recipe. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to overall operational efficiency. The simplified purification workflow means less solvent usage and shorter processing times, which cumulatively result in substantial cost savings without compromising product quality. These economic advantages make the technology highly attractive for large-scale manufacturing where margin optimization is critical.
- Enhanced Supply Chain Reliability: By relying on abundant and commercially available starting materials such as magnesium chloride and common solvents, manufacturers can mitigate the risks associated with supply chain bottlenecks. The robustness of the electrochemical setup allows for flexible production scheduling, as the reaction can be easily started or stopped by controlling the power supply. This flexibility is invaluable for meeting fluctuating demand in the pharmaceutical sector, ensuring that customers receive their orders on time without the delays often caused by complex multi-step syntheses or reliance on single-source reagents.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with increasingly stringent environmental regulations, reducing the burden of waste treatment and permitting. The absence of heavy metal catalysts and toxic oxidants simplifies the effluent profile, making it easier to meet discharge standards and maintain a sustainable manufacturing footprint. Scalability is further supported by the modular nature of electrochemical reactors, which can be expanded by adding more cells or increasing electrode surface area, facilitating a smooth transition from gram-scale research to ton-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of this electrocatalytic synthesis method, providing clarity for potential partners and stakeholders. These insights are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the fit of this technology within existing manufacturing frameworks and for assessing its potential impact on product development pipelines.
Q: What are the primary advantages of using electrocatalysis for synthesizing alpha-carbonyl sulfoxide ylide dichlorides compared to traditional chemical oxidants?
A: The electrocatalytic method described in patent CN114411182A eliminates the need for external chemical oxidants and additives, resulting in a greener process with reduced waste generation. It operates under mild conditions (20°C) and utilizes electricity as a clean reagent, significantly simplifying the workup procedure and enhancing safety profiles for industrial scale-up.
Q: What is the substrate scope for this electrochemical dichlorination reaction?
A: The methodology demonstrates excellent functional group tolerance, successfully accommodating various R substituents including phenyl, substituted phenyls (methyl, methoxy, chloro), heterocycles like furyl and thienyl, as well as aliphatic groups such as cyclohexyl and adamantyl. This broad versatility makes it highly suitable for generating diverse libraries of pharmaceutical intermediates.
Q: Is this electrochemical protocol scalable for commercial production of fine chemicals?
A: Yes, the process utilizes standard electrolytic cell configurations with graphite felt and platinum electrodes, which are amenable to flow chemistry and larger batch reactors. The use of inexpensive magnesium chloride as the chlorine source and the absence of hazardous oxidants facilitate safer commercial scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Carbonyl Sulfoxide Ylide Dichloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic synthesis in modernizing the production of complex pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114411182A can be seamlessly translated into robust industrial processes. We are committed to delivering high-purity alpha-carbonyl sulfoxide ylide dichlorides that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to green chemistry and process efficiency positions us as a strategic partner for global pharmaceutical companies seeking to optimize their supply chains and reduce their environmental footprint.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this electrocatalytic method for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and efficiency in your drug development programs.
