Advanced Electrochemical Synthesis of Biaryl Cyclic Phosphoramides for OLED and Ligand Applications
The pharmaceutical and electronic materials industries are constantly seeking more sustainable and efficient pathways for constructing complex heterocyclic frameworks, particularly those serving as critical intermediates for high-performance applications. Patent CN113980055A introduces a groundbreaking electrochemical strategy for the synthesis of cyclic phosphoramide derivatives with a biaryl framework, addressing the longstanding challenges associated with traditional transition metal-catalyzed coupling reactions. This innovation leverages electrons as traceless reagents to drive intramolecular phosphine amination, offering a green alternative that bypasses the need for stoichiometric oxidants or precious metal catalysts. The resulting compounds are pivotal precursors for organic photoelectric materials used in OLED displays and serve as versatile chiral PN-type ligands for asymmetric catalysis. By shifting the paradigm from chemical oxidants to electrochemical oxidation, this technology not only enhances atom economy but also drastically simplifies the impurity profile of the final product, making it highly attractive for large-scale commercial manufacturing where purity and environmental compliance are paramount concerns for global supply chains.
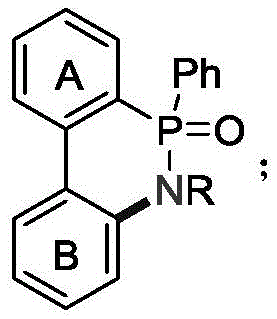
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biaryl cyclic phosphoramide skeletons has relied heavily on transition metal-catalyzed intramolecular C-N coupling or oxidative dehydrogenation reactions utilizing chemical oxidants. These conventional methodologies, while effective in laboratory settings, present significant bottlenecks when translated to industrial production scales. The reliance on transition metals such as palladium or copper necessitates rigorous and costly downstream purification steps to remove trace metal residues, which is especially critical for electronic materials where metal contamination can degrade device performance. Furthermore, the use of stoichiometric oxidants like PhI(OAc)2, NIS, or CAN generates substantial amounts of chemical waste, leading to increased disposal costs and environmental liabilities. These processes often require harsh reaction conditions, including elevated temperatures and extended reaction times, which can compromise the stability of sensitive functional groups and limit the scope of applicable substrates. The cumulative effect of these factors is a manufacturing process that is economically inefficient, environmentally burdensome, and operationally complex, failing to meet the modern standards of green chemistry and sustainable manufacturing required by top-tier multinational corporations.
The Novel Approach
In stark contrast to the drawbacks of traditional synthesis, the electrochemical method disclosed in the patent utilizes electricity as the primary driving force for the oxidative cyclization, effectively replacing chemical oxidants with electrons. This approach operates under remarkably mild conditions, typically at room temperature, which preserves the integrity of sensitive functional groups and allows for a broader substrate scope including those with electron-withdrawing or electron-donating substituents. The absence of transition metal catalysts eliminates the risk of heavy metal contamination, thereby reducing the complexity of the workup procedure and ensuring a cleaner final product profile suitable for high-purity applications. The reaction system employs a simple undivided cell configuration with inexpensive electrode materials such as graphite and platinum, which significantly lowers the capital expenditure and operational costs associated with the reactor setup. By generating hydrogen gas as the sole byproduct at the cathode, the process aligns perfectly with the principles of atom economy and environmental sustainability, offering a compelling value proposition for procurement teams focused on reducing the total cost of ownership and minimizing the environmental footprint of their supply chains.
Mechanistic Insights into Electrochemical Intramolecular Phosphine Amination
The core of this technological advancement lies in the anodic oxidation mechanism that facilitates the formation of the P-N bond within the biaryl framework without external oxidants. Upon applying a constant current, the phosphoramide substrate undergoes single-electron oxidation at the anode surface to generate a reactive radical cation intermediate. This high-energy species subsequently triggers an intramolecular nucleophilic attack by the nitrogen atom onto the adjacent aromatic ring, leading to the cyclization and formation of the stable cyclic phosphoramide structure. The use of n-butyl ammonium bromide as a supporting electrolyte plays a crucial role in mediating the electron transfer process and stabilizing the reactive intermediates, ensuring high conversion rates and selectivity. This mechanistic pathway avoids the formation of metal-ligand complexes that are typical in palladium-catalyzed cycles, thereby circumventing the issues of catalyst deactivation and metal leaching that often plague conventional methods. The robustness of this electrochemical cycle is evidenced by its tolerance to a wide range of substituents on the aromatic rings, allowing for the synthesis of diverse derivatives without the need for extensive optimization of reaction parameters for each new substrate.
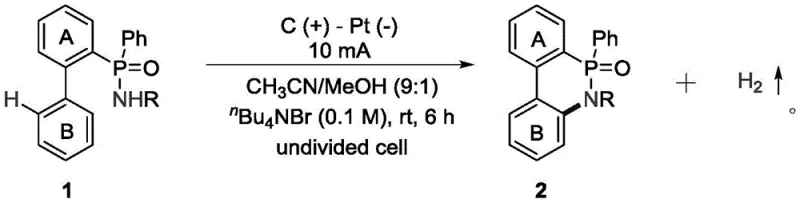
From an impurity control perspective, the electrochemical method offers distinct advantages by minimizing the generation of side products associated with chemical oxidants. In traditional oxidative couplings, the reduction products of the oxidant often remain in the reaction mixture, requiring complex chromatographic separations to achieve the desired purity levels. In the electrochemical process, the only byproducts are hydrogen gas and the oxidized form of the electrolyte, which can be easily managed or recycled. This cleanliness translates directly into higher isolated yields and reduced solvent consumption during the purification stage, as fewer passes through silica gel columns are required to remove inorganic salts or metal residues. For R&D directors, this means a more predictable and reproducible synthesis route that accelerates the timeline from laboratory discovery to pilot plant validation. The ability to fine-tune the reaction outcome by adjusting the current density or electrode potential provides an additional layer of process control, enabling the optimization of selectivity for specific isomers or derivatives that might be difficult to access through thermal or chemical activation methods.
How to Synthesize Biaryl Cyclic Phosphoramides Efficiently
The practical implementation of this electrochemical synthesis route is designed to be straightforward and adaptable to existing chemical manufacturing infrastructure, requiring minimal specialized equipment beyond a standard DC power supply and electrochemical cell. The process begins with the dissolution of the phosphoramide precursor and the supporting electrolyte in a mixed solvent system of acetonitrile and methanol, which provides the optimal conductivity and solubility for the reaction to proceed efficiently. Once the electrodes are immersed and the current is applied, the reaction progresses autonomously at ambient temperature, eliminating the need for energy-intensive heating or cooling systems that are common in traditional batch reactors. Detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification protocols, are outlined in the structured guide below to ensure reproducibility and safety during scale-up operations. This streamlined workflow not only reduces the operational burden on technical teams but also enhances the overall safety profile of the manufacturing process by avoiding the handling of hazardous chemical oxidants and high-temperature conditions.
- Prepare the reaction mixture by adding 1 equivalent of phosphoramide substrate and 0.1 mol/L of n-butyl ammonium bromide into a reaction vessel with acetonitrile/methanol mixed solvent.
- Insert a graphite anode and a platinum cathode into the undivided cell and connect to a power supply to introduce a constant current of 10 milliampere.
- Stir the reaction at room temperature for 6 hours, then concentrate under reduced pressure and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology represents a strategic opportunity to optimize cost structures and enhance supply reliability for critical electronic chemical intermediates. The elimination of expensive transition metal catalysts and stoichiometric oxidants directly reduces the raw material costs, while the simplified purification process lowers the consumption of solvents and chromatography media, contributing to significant overall cost savings in the manufacturing budget. Furthermore, the mild reaction conditions and robust substrate tolerance reduce the risk of batch failures and production delays, ensuring a more consistent and reliable supply of high-purity materials to meet the demanding schedules of downstream clients in the OLED and pharmaceutical sectors. The green nature of the process also aligns with increasingly stringent environmental regulations, mitigating the risk of compliance-related disruptions and enhancing the corporate sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts and chemical oxidants from the process workflow eliminates a major cost driver associated with traditional synthesis methods, leading to substantial reductions in direct material expenses. Additionally, the simplified workup procedure reduces the labor and utility costs associated with extensive purification steps, allowing for a more lean and efficient production model that maximizes resource utilization. The use of inexpensive electrode materials and common solvents further contributes to the economic viability of the process, making it a highly competitive option for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The robustness of the electrochemical method against variations in substrate structure ensures a stable and consistent production output, minimizing the risk of supply interruptions caused by process sensitivity or raw material quality fluctuations. The availability of simple and scalable equipment configurations allows for rapid capacity expansion to meet surging demand, providing a flexible supply base that can adapt to the dynamic needs of the global electronics and pharmaceutical markets. This reliability is further bolstered by the reduced dependence on specialized reagents that may be subject to supply chain volatility, ensuring a secure and continuous flow of materials to end users.
- Scalability and Environmental Compliance: The inherent safety and cleanliness of the electrochemical process facilitate seamless scale-up from laboratory to industrial production without the need for complex engineering controls or hazardous waste management systems. The generation of minimal waste and the absence of toxic heavy metals simplify the environmental compliance landscape, reducing the regulatory burden and associated costs of waste disposal and treatment. This alignment with green chemistry principles not only future-proofs the manufacturing operation against tightening environmental regulations but also enhances the brand value of the final products in markets that prioritize sustainability and ethical sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of biaryl cyclic phosphoramides, providing clarity on the process capabilities and benefits based on the patented technology. These insights are derived from the experimental data and comparative analysis presented in the patent documentation, offering a transparent view of the method's performance and applicability for industrial partners. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains or product development pipelines.
Q: What are the primary advantages of this electrochemical method over traditional transition metal catalysis?
A: The electrochemical method eliminates the need for expensive transition metal catalysts and stoichiometric oxidants, significantly reducing heavy metal impurities and waste generation while simplifying the downstream purification process.
Q: Does this synthesis method support diverse functional groups on the biaryl skeleton?
A: Yes, the method demonstrates excellent substrate universality, tolerating various electron-withdrawing and electron-donating groups such as halogens, alkyls, alkoxy, and trifluoromethyl groups on both aromatic rings without significant yield loss.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction proceeds under mild conditions at room temperature using a constant current of 10 mA in an undivided cell with a graphite anode and platinum cathode, typically completing within 6 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biaryl Cyclic Phosphoramide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this electrochemical synthesis technology in delivering high-performance materials for the next generation of electronic and pharmaceutical applications. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-scale manufacturing environments. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of biaryl cyclic phosphoramide meets the exacting standards required for OLED materials and chiral ligand applications. We are committed to leveraging our technical expertise to optimize this green synthesis route, providing our clients with a sustainable and cost-effective supply solution that drives innovation and competitiveness in their respective markets.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing capability can address your specific material requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits associated with switching to this electrochemical process for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance the efficiency and sustainability of your production operations while securing a reliable source of high-quality electronic chemical intermediates.
