Advancing Pharmaceutical Intermediates: Efficient Co/Cu Catalyzed Synthesis of Pyrido[2,1-a]isoindoles
Advancing Pharmaceutical Intermediates: Efficient Co/Cu Catalyzed Synthesis of Pyrido[2,1-a]isoindoles
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the need for more sustainable and cost-effective manufacturing processes. A significant breakthrough in this domain is detailed in patent CN107629049B, which introduces a robust methodology for constructing the pyrido[2,1-a]isoindole scaffold. This privileged structure is increasingly recognized for its potential in medicinal chemistry, yet its synthesis has historically been plagued by harsh conditions and expensive catalysts. The disclosed invention leverages a dual cobalt and copper catalytic system to achieve efficient coupling and cyclization under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more accessible chemical building blocks. By utilizing earth-abundant metals instead of precious group 9 or 10 metals, the process not only lowers the barrier to entry for synthesizing these complex molecules but also aligns with modern green chemistry principles. This report delves into the technical nuances and commercial implications of this novel synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrido[2,1-a]isoindole core has relied on methodologies that present significant logistical and economic challenges for large-scale production. As illustrated in the prior art summary, early approaches utilized photocatalytic intramolecular cyclization, which often requires specialized UV equipment and suffers from limited scalability due to light penetration issues in larger reactors. ![Prior art synthesis methods for pyrido[2,1-a]isoindole scaffolds showing various catalytic systems](/insights/img/pyrido-isoindole-synthesis-pharma-supplier-20260304065844-01.webp) Furthermore, more recent developments involving rhodium-catalyzed alkynylation or iron-catalyzed couplings, while effective, introduce substantial cost burdens. Rhodium is a precious metal with volatile pricing and supply chain vulnerabilities, making it a less attractive option for commodity-level intermediate production. Additionally, many of these legacy protocols necessitate strictly inert atmospheres, such as nitrogen or argon, adding complexity to the operational setup and increasing the risk of batch failure due to oxygen sensitivity. These factors collectively hinder the widespread adoption of pyrido[2,1-a]isoindole derivatives in drug discovery pipelines where speed and cost are paramount.
Furthermore, more recent developments involving rhodium-catalyzed alkynylation or iron-catalyzed couplings, while effective, introduce substantial cost burdens. Rhodium is a precious metal with volatile pricing and supply chain vulnerabilities, making it a less attractive option for commodity-level intermediate production. Additionally, many of these legacy protocols necessitate strictly inert atmospheres, such as nitrogen or argon, adding complexity to the operational setup and increasing the risk of batch failure due to oxygen sensitivity. These factors collectively hinder the widespread adoption of pyrido[2,1-a]isoindole derivatives in drug discovery pipelines where speed and cost are paramount.
The Novel Approach
In stark contrast to the limitations of previous techniques, the method disclosed in CN107629049B offers a streamlined pathway that democratizes access to this valuable scaffold. The core innovation lies in the use of a cobalt and copper co-catalytic system, which effectively replaces expensive noble metals with abundant transition metals. ![General reaction scheme for Co/Cu catalyzed synthesis of pyrido[2,1-a]isoindoles from phenylpyridines](/insights/img/pyrido-isoindole-synthesis-pharma-supplier-20260304065844-02.webp) This approach utilizes simple starting materials, specifically phenylpyridines and bromoacetophenones, which are readily available from global chemical suppliers. Crucially, the reaction proceeds efficiently under an air atmosphere at temperatures ranging from 120°C to 140°C, eliminating the need for costly inert gas purging and specialized glovebox techniques. The operational simplicity is further enhanced by the use of common solvents like toluene and standard bases like cesium carbonate. For a reliable pharmaceutical intermediate supplier, this translates to a process that is not only chemically robust but also inherently safer and easier to manage in a multi-purpose manufacturing facility. The broad substrate scope allows for the rapid generation of diverse libraries, accelerating the lead optimization phase for medicinal chemists.
This approach utilizes simple starting materials, specifically phenylpyridines and bromoacetophenones, which are readily available from global chemical suppliers. Crucially, the reaction proceeds efficiently under an air atmosphere at temperatures ranging from 120°C to 140°C, eliminating the need for costly inert gas purging and specialized glovebox techniques. The operational simplicity is further enhanced by the use of common solvents like toluene and standard bases like cesium carbonate. For a reliable pharmaceutical intermediate supplier, this translates to a process that is not only chemically robust but also inherently safer and easier to manage in a multi-purpose manufacturing facility. The broad substrate scope allows for the rapid generation of diverse libraries, accelerating the lead optimization phase for medicinal chemists.
Mechanistic Insights into Co/Cu Co-catalyzed Cyclization
The success of this transformation hinges on the synergistic interaction between the cobalt and copper species, which facilitates a cascade of C-H activation and annulation events. While the precise mechanistic cycle is complex, it is hypothesized that the copper species initially activates the bromoacetophenone through oxidative addition or single-electron transfer processes, generating a reactive organometallic intermediate. Simultaneously, the cobalt catalyst likely coordinates with the nitrogen atom of the phenylpyridine, directing the activation of the proximal C-H bond on the phenyl ring. This dual activation brings the two coupling partners into close proximity, fostering the formation of the new carbon-carbon bond that bridges the pyridine and isoindole systems. The subsequent intramolecular cyclization closes the five-membered ring, establishing the rigid pyrido[2,1-a]isoindole architecture. Understanding this mechanism is vital for R&D teams aiming to further optimize the reaction or adapt it to novel substrates, as it highlights the critical role of the directing group and the electronic nature of the catalysts.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring high product purity. High-temperature reactions often lead to decomposition or polymerization of sensitive functional groups, but the 130°C threshold employed here strikes a balance between reactivity and stability. 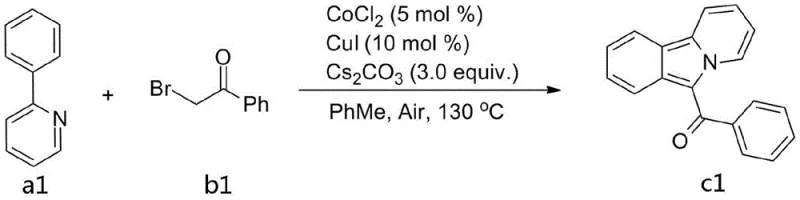 The use of air as the oxidant, rather than harsh chemical oxidants, minimizes the formation of over-oxidized byproducts that can be difficult to separate. Furthermore, the compatibility with various substituents, such as electron-donating methoxy groups and electron-withdrawing esters, suggests that the catalytic cycle is resilient to electronic perturbations. This resilience reduces the formation of regioisomers or side products that typically arise from non-selective C-H activation. For quality control laboratories, this means a cleaner crude reaction profile, which simplifies downstream purification and ensures that the final high-purity pharmaceutical intermediate meets stringent specifications with minimal processing steps.
The use of air as the oxidant, rather than harsh chemical oxidants, minimizes the formation of over-oxidized byproducts that can be difficult to separate. Furthermore, the compatibility with various substituents, such as electron-donating methoxy groups and electron-withdrawing esters, suggests that the catalytic cycle is resilient to electronic perturbations. This resilience reduces the formation of regioisomers or side products that typically arise from non-selective C-H activation. For quality control laboratories, this means a cleaner crude reaction profile, which simplifies downstream purification and ensures that the final high-purity pharmaceutical intermediate meets stringent specifications with minimal processing steps.
How to Synthesize Pyrido[2,1-a]isoindole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility. The protocol is designed to be straightforward, minimizing the need for specialized equipment while ensuring consistent results. The process begins with the precise weighing of the phenylpyridine derivative and the bromoacetophenone derivative, typically in a molar ratio favoring the ketone component to drive the reaction to completion. The catalysts, cobalt chloride and cuprous iodide, are added in catalytic amounts, alongside a stoichiometric excess of cesium carbonate to act as the base. The choice of solvent, preferably toluene, is critical for solubilizing the organic substrates while withstanding the elevated reaction temperatures. Detailed standardized synthesis steps for this procedure are outlined in the guide below.
- Combine phenylpyridine derivative, bromoacetophenone derivative, CoCl2 (5 mol%), CuI (10 mol%), and Cs2CO3 (3.0 equiv.) in toluene.
- Heat the reaction mixture at 130°C under an air atmosphere for at least 24 hours to facilitate coupling and cyclization.
- Filter the mixture, concentrate, and purify the crude product via silica gel column chromatography using petroleum ether/ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this cobalt/copper catalyzed method offers tangible benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic reduction of raw material costs associated with the catalytic system. By substituting rhodium or palladium with cobalt and copper, the direct material cost of the catalyst package is significantly lowered, which has a compounding effect on the overall cost of goods sold (COGS). Moreover, the elimination of inert atmosphere requirements simplifies the manufacturing infrastructure, allowing production to occur in standard glass-lined or stainless steel reactors without the need for extensive nitrogen blanketing systems. This operational flexibility enhances supply chain reliability by reducing dependency on specialized utility gases and minimizing the risk of production delays caused by equipment failures related to pressure or gas supply.
- Cost Reduction in Manufacturing: The economic impact of switching to earth-abundant metals cannot be overstated in the context of fine chemical manufacturing. Cobalt and copper are orders of magnitude cheaper than rhodium, and their price volatility is generally lower, allowing for more stable long-term budgeting. Additionally, the reaction utilizes cesium carbonate, a base that, while premium, is used in stoichiometric amounts that are manageable compared to the cost savings on the metal catalyst. The simplified workup procedure, involving basic filtration and column chromatography, reduces solvent consumption and labor hours compared to complex extraction protocols often required for sensitive organometallic reactions. These factors collectively contribute to a leaner manufacturing process that supports competitive pricing strategies for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of precious metals and the geopolitical complexities surrounding their sourcing. By relying on cobalt and copper, which have robust and diversified global supply chains, manufacturers can mitigate the risk of raw material shortages. Furthermore, the ability to run the reaction under air conditions removes a critical point of failure; there is no risk of batch contamination due to oxygen leaks, a common issue in large-scale inert atmosphere reactions. This robustness ensures consistent batch-to-batch quality and reliable delivery schedules, which is crucial for maintaining the production timelines of downstream API manufacturers. The use of common solvents like toluene further ensures that solvent supply remains stable and cost-effective.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden bottlenecks, but this methodology is inherently scalable due to its thermal and atmospheric simplicity. The reaction temperature of 130°C is easily achievable with standard heating media like thermal oil, avoiding the need for high-pressure autoclaves. From an environmental standpoint, the use of air as the terminal oxidant generates water as the primary byproduct, significantly reducing the toxic waste load compared to methods using stoichiometric chemical oxidants. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, facilitating smoother regulatory approvals and reducing waste disposal costs. The process exemplifies how green chemistry principles can be integrated into commercial scale-up of complex heterocycles without sacrificing efficiency.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical implementation and commercial viability of this synthesis method, we have compiled answers based on the specific data points provided in the patent literature. These questions reflect the practical concerns of technical teams evaluating this route for potential integration into their supply chains. The answers are derived directly from the experimental examples and summary data, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of the Co/Cu catalytic system over traditional noble metal catalysts?
A: The Co/Cu system significantly reduces raw material costs compared to Rhodium or Palladium catalysts while maintaining high yields and operating under mild air conditions rather than inert atmospheres.
Q: Does this synthesis method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating substituents such as methoxy, methyl, ester, and halogen groups on both the phenylpyridine and bromoacetophenone rings.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction typically proceeds in toluene at temperatures between 120°C and 140°C under an air atmosphere, requiring approximately 24 hours for completion without the need for specialized inert gas equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrido[2,1-a]isoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN107629049B for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable industrial supply. We are committed to delivering high-purity pyrido[2,1-a]isoindole derivatives that meet rigorous quality standards, supported by our state-of-the-art rigorous QC labs and analytical capabilities. Our expertise in transition metal catalysis allows us to optimize these Co/Cu systems further, tailoring the process to specific client needs while maintaining cost efficiency and environmental compliance.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced technology for their drug discovery programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific synthetic route can optimize your project budget. We encourage you to contact our technical procurement team to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Together, we can accelerate the delivery of vital medicines to the market through superior chemical manufacturing solutions.
