Revolutionizing Benzofuran-3-oxocarboxylate Production: A Metal-Free Strategy for Commercial Scale-up
Revolutionizing Benzofuran-3-oxocarboxylate Production: A Metal-Free Strategy for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly benzofuran derivatives which serve as critical cores in numerous bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN113214199B, which discloses a novel synthetic method for benzofuran-3-oxocarboxylate compounds. Unlike traditional approaches that rely heavily on transition metals or harsh oxidants, this invention leverages a sophisticated acid-catalyzed rearrangement-cyclization cascade reaction. By utilizing conjugated alkenyl amidine compounds as starting materials, the process achieves the target structures in a single pot with remarkable efficiency. This technological advancement represents a paradigm shift for any reliable pharmaceutical intermediate supplier aiming to streamline their production lines while adhering to stricter environmental regulations.
The core innovation lies in the ability to construct the benzofuran ring system directly from readily available precursors without the need for precious metal catalysts such as palladium or platinum. The reaction proceeds smoothly under relatively mild thermal conditions, typically around 100°C, using common organic solvents. This eliminates the complex downstream processing associated with removing trace metal contaminants, a persistent challenge in API manufacturing. Furthermore, the method demonstrates exceptional substrate universality, tolerating a wide range of functional groups including halogens, alkyl chains, and alkoxy substituents. For R&D teams focused on library synthesis, this offers a robust platform for generating diverse analogues rapidly, thereby accelerating the drug discovery timeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 3-substituted benzofuran derivatives was predominantly governed by multi-step sequences that were both operationally cumbersome and environmentally taxing. A representative example is the method reported by the Kozikowski group (US20100004308), which served as the benchmark for years. This legacy route begins with a Friedel-Crafts acylation of phenols, followed by cyclization to form a benzofuranone intermediate. Crucially, it requires a Wittig reaction to introduce the side chain, and finally, an oxidation step using selenium dioxide (SeO2) to install the ketone functionality. 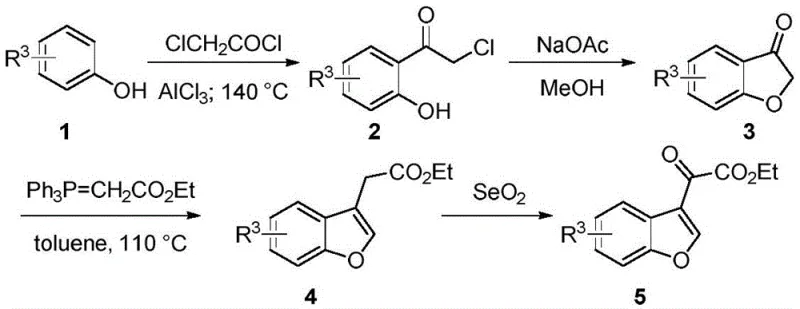
The reliance on selenium dioxide presents severe drawbacks for large-scale operations. Selenium compounds are notoriously toxic and pose significant health risks to operators, necessitating specialized containment and waste treatment facilities. Moreover, the multi-step nature of this sequence inherently leads to cumulative yield losses and increased consumption of raw materials. Each isolation and purification step adds time and cost, creating bottlenecks in the supply chain. Additionally, the use of strong Lewis acids in the initial acylation step often requires strict anhydrous conditions and low temperatures, further complicating the process engineering requirements for commercial plants.
The Novel Approach
In stark contrast, the methodology described in CN113214199B offers a streamlined, one-pot solution that bypasses these historical inefficiencies. The new strategy employs a conjugated alkenyl amidine substrate which undergoes a tandem rearrangement and cyclization upon exposure to a Brønsted acid catalyst. 
This approach fundamentally simplifies the synthetic logic by merging bond formation and functional group transformation into a single operational unit. The elimination of the selenium oxidation step alone constitutes a major victory for green chemistry initiatives, drastically reducing the hazardous waste profile of the manufacturing process. Furthermore, the reaction can be conducted under an air atmosphere, removing the need for expensive inert gas purging systems. The operational simplicity allows for higher throughput and reduced labor costs, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing. The robustness of the reaction conditions ensures consistent quality, which is paramount for maintaining supply chain continuity.
Mechanistic Insights into Acid-Catalyzed Rearrangement-Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific substrates. The reaction initiates with the protonation of the amidine nitrogen by the Brønsted acid, generating a reactive quaternary ammonium salt intermediate. This activation triggers an intramolecular nucleophilic attack by the phenolic oxygen onto the activated alkene, forming a seven-membered 1,3-benzoxazine ring intermediate. 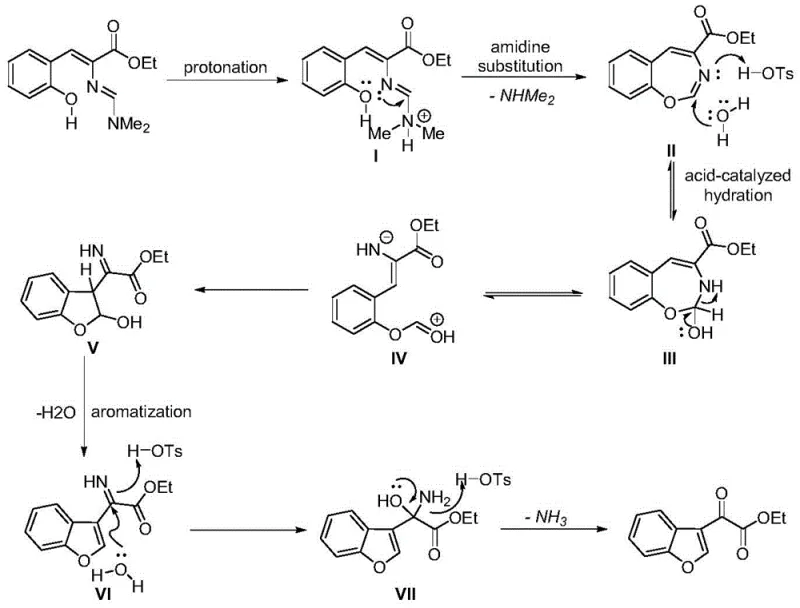
Subsequently, the system undergoes a fascinating hydration and ring-opening rearrangement. Water molecules, either from the hydrated acid catalyst or the solvent environment, facilitate the hydrolysis of the imine bond, leading to a zwitterionic species. This intermediate then collapses via a second cyclization event to form the dihydrobenzofuran five-membered ring. The final driving force of the reaction is aromatization; the loss of a water molecule and subsequent elimination of dimethylamine restores the aromaticity of the furan ring, yielding the stable benzofuran-3-oxocarboxylate product. This intricate cascade highlights the elegance of organocatalysis, where simple protons orchestrate complex bond reorganizations without the need for external redox agents.
From an impurity control perspective, this mechanism is highly favorable. The pathway avoids the generation of radical species often associated with metal-catalyzed oxidations, which can lead to unpredictable side products. The primary by-products are dimethylamine and water, both of which are volatile and easily removed during the concentration phase. This clean reaction profile simplifies the purification process, often allowing for straightforward crystallization or minimal chromatography. For quality control teams, this translates to a cleaner crude profile and easier validation of the final API intermediate, ensuring compliance with stringent purity specifications required by regulatory bodies.
How to Synthesize Benzofuran-3-oxocarboxylate Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility. The standard protocol involves charging the conjugated alkenyl amidine precursor and p-Toluenesulfonic acid monohydrate into a reaction vessel, typically a Schlenk tube for small scale or a glass-lined reactor for larger batches. Acetonitrile is the preferred solvent due to its ability to stabilize the polar transition states involved in the rearrangement. The mixture is then heated to reflux, approximately 100°C, and maintained for 8 hours. Monitoring via TLC ensures complete conversion of the starting material before workup.
- Load conjugated alkenyl amidine substrate and p-TsOH·H2O (2.0 equiv) into a reaction vessel.
- Add anhydrous acetonitrile as the solvent and heat the mixture to 100°C under air atmosphere.
- Stir for 8 hours until TLC indicates complete conversion, then concentrate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical curiosity. The most significant advantage is the drastic simplification of the supply chain for raw materials. By eliminating the need for selenium dioxide, triphenylphosphine, and expensive transition metal catalysts, the bill of materials becomes leaner and less susceptible to geopolitical supply shocks. These traditional reagents often face fluctuating prices and availability issues, whereas p-Toluenesulfonic acid and acetonitrile are commodity chemicals produced at massive scales globally. This stability ensures enhanced supply chain reliability and predictable budgeting for long-term production contracts.
Furthermore, the environmental and safety profile of this process directly correlates to substantial cost savings in waste management. Hazardous waste disposal, particularly for heavy metals and selenium compounds, is a major cost center in chemical manufacturing. By switching to a metal-free, organocatalytic process, facilities can significantly reduce their hazardous waste volume. This not only lowers disposal fees but also minimizes the regulatory burden and insurance premiums associated with handling toxic substances. The ability to run the reaction under air atmosphere further reduces utility costs related to nitrogen consumption and specialized equipment maintenance. These cumulative efficiencies contribute to a more competitive pricing structure for the final intermediates.
Scalability is another critical factor where this method excels. The one-pot nature of the reaction reduces the number of unit operations, meaning fewer transfers between reactors and less hold-up volume. This intensification of the process allows existing infrastructure to produce higher volumes without capital expenditure on new equipment. The robustness of the acid catalysis means the reaction is less sensitive to minor variations in temperature or mixing, a common challenge when scaling up from gram to ton quantities. Consequently, this technology facilitates the commercial scale-up of complex pharmaceutical intermediates with lower risk and faster time-to-market, enabling companies to respond agilely to market demands.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation of this synthesis, we have compiled answers based on the experimental data provided in the patent documentation. These insights cover catalyst selection, solvent effects, and substrate tolerance, providing a clear roadmap for technical teams evaluating this route.
Q: Why is this new method superior to the Kozikowski route?
A: The conventional Kozikowski route requires toxic selenium dioxide oxidation and multiple steps including Wittig reactions. This new patent utilizes a direct one-pot acid-catalyzed rearrangement, eliminating heavy metal waste and significantly simplifying the workflow.
Q: What catalysts are compatible with this rearrangement reaction?
A: While various Brønsted acids were tested, p-Toluenesulfonic acid monohydrate (p-TsOH·H2O) and Camphorsulfonic acid demonstrated the highest efficacy, achieving yields over 70%, whereas Lewis acids like TfOH showed negligible activity.
Q: Is this process scalable for industrial production?
A: Yes, the reaction operates under air atmosphere without inert gas protection and uses common solvents like acetonitrile. The absence of sensitive transition metal catalysts and the robust nature of the acid catalysis make it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran-3-oxocarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN113214199B for the production of high-value benzofuran derivatives. As a leading CDMO partner, we possess the technical expertise to translate this academic innovation into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this metal-free route are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzofuran-3-oxocarboxylate meets the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis can optimize your specific supply chain. Whether you require a Customized Cost-Saving Analysis for your current portfolio or need specific COA data and route feasibility assessments for new projects, we are equipped to provide comprehensive support. By leveraging this cutting-edge technology, we can collectively drive down costs and enhance the sustainability of your drug development pipeline, securing a competitive edge in the marketplace.
