Revolutionizing Catalytic Efficiency with Scalable Metal N-Heterocyclic Carbene Complexes
Introduction to Advanced Catalytic Precursor Technology
The landscape of modern organometallic chemistry is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective catalytic systems in the pharmaceutical and fine chemical industries. Patent CN101402644B introduces a groundbreaking preparation method for metal nitrogen heterocyclic carbene (NHC) complexes that addresses critical bottlenecks in traditional synthesis pathways. This technology leverages the unique reactivity of imidazolium salts with silver oxide followed by transmetallation with base metals, or alternatively, a direct oxidative metalation approach using active metal powders in the presence of air. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation represents a pivotal shift towards robust, scalable, and economically viable catalyst manufacturing. The ability to synthesize complexes of manganese, iron, cobalt, nickel, and copper with high yields and purity opens new avenues for developing environmentally friendly catalytic processes that do not rely on scarce and expensive precious metals like palladium or ruthenium.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of metal-NHC complexes has been fraught with challenges that hinder their widespread industrial adoption. Traditional methods often rely on the generation of free carbenes, which are notoriously unstable and highly reactive, leading to unpredictable side reactions and decomposition before they can coordinate with the metal center. Alternatively, the use of strong bases to deprotonate imidazolium salts in situ frequently results in the formation of inorganic salt byproducts that are difficult to remove, complicating downstream purification and compromising the purity profile required for sensitive pharmaceutical applications. Furthermore, many conventional protocols require stringent anhydrous conditions, inert atmospheres, and cryogenic temperatures, which drastically increase operational costs and limit the feasibility of large-scale commercial production. These technical barriers have historically restricted the utility of NHC ligands to high-value, low-volume applications, preventing their integration into bulk chemical manufacturing where cost efficiency is paramount.
The Novel Approach
The methodology disclosed in the patent data offers a sophisticated solution to these enduring problems through a versatile two-pronged strategy. The first method utilizes a silver-mediated transmetallation pathway, where an Ag-NHC intermediate is formed under mild conditions (40-60°C) in acetonitrile, serving as a stable carbene transfer agent to the target base metal. The second method simplifies the process even further by employing direct reaction of imidazolium salts with active metal powders in the presence of atmospheric oxygen, effectively bypassing the need for pre-formed silver intermediates. This dual approach ensures that even sterically hindered or electronically deactivated imidazolium salts can be successfully converted into stable metal complexes. By operating at moderate temperatures ranging from 40°C to 80°C and utilizing common solvents, this novel approach drastically reduces energy consumption and equipment requirements. 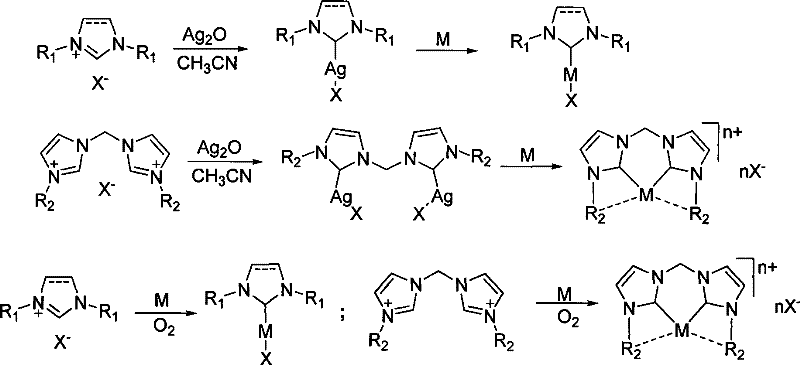
Mechanistic Insights into Silver-Mediated Transmetallation and Direct Metalation
At the heart of this technological advancement lies a deep understanding of organometallic coordination chemistry and redox processes. In the silver-mediated pathway, silver oxide acts not merely as a base but as a soft Lewis acid that facilitates the deprotonation of the C2 position of the imidazolium ring, forming a robust Ag-C bond. This Ag-NHC species is kinetically stable yet thermodynamically poised for transmetallation. When introduced to a more oxophilic or coordinatively unsaturated base metal powder, such as copper or nickel, the carbene ligand transfers efficiently due to the stronger metal-carbene bond strength of the target metal compared to silver. This mechanism effectively circumvents the instability of free carbenes by keeping the ligand coordinated throughout the entire synthetic sequence. In the direct metalation variant, the presence of oxygen plays a crucial role in activating the metal surface or oxidizing the metal to a state capable of oxidative addition into the C-H bond of the imidazolium salt. This oxidative insertion is facilitated by the polar aprotic nature of acetonitrile, which stabilizes the charged intermediates and promotes the dissolution of metal ions.
From an impurity control perspective, this mechanism offers distinct advantages over base-mediated deprotonation. Since the process does not generate stoichiometric amounts of inorganic salts (such as lithium halides or potassium carbonates) as byproducts, the crude reaction mixture is significantly cleaner. The primary impurities are typically unreacted starting materials or homocoupled ligand species, which can be easily removed through standard crystallization techniques involving diethyl ether precipitation. The use of acetonitrile as a solvent ensures that the metal complexes remain in solution during the reaction but can be selectively precipitated upon the addition of a non-polar anti-solvent. This solubility switch is critical for achieving the high purity specifications demanded by regulatory bodies for pharmaceutical intermediates. Furthermore, the avoidance of strong bases prevents the hydrolysis of sensitive functional groups on the ligand periphery, such as esters or nitriles, preserving the structural integrity and catalytic activity of the final complex.
How to Synthesize Metal N-Heterocyclic Carbene Complexes Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing pilot and production facilities. The process begins with the precise weighing of imidazolium salt precursors and silver oxide, which are suspended in dry acetonitrile under light-protected conditions to prevent photodecomposition of the silver species. Following the formation of the intermediate, the target metal powder is added, and the mixture is heated to promote the transmetallation event. Alternatively, for the direct method, the imidazolium salt and metal powder are combined in acetonitrile and heated in an open vessel to allow air circulation. Detailed standardized synthesis steps see the guide below.
- Dissolve the imidazolium salt precursor in acetonitrile and react with silver oxide under light-protected conditions to form the Ag-NHC intermediate.
- Introduce active metal powder (such as copper, nickel, or iron) to the reaction mixture and heat to facilitate transmetallation or direct coordination.
- Filter the reaction solution, concentrate the filtrate, and induce crystallization using diethyl ether to isolate the high-purity metal complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology translates into tangible strategic benefits that extend beyond simple unit cost savings. The shift from precious metal catalysts to base metal analogues fundamentally alters the cost structure of catalytic processes, reducing exposure to the volatile pricing of commodities like palladium and platinum. Moreover, the simplified workup procedures eliminate the need for expensive scavenging resins or complex chromatographic purification steps that are typically required to remove trace metal contaminants from API intermediates. This streamlining of the downstream processing workflow significantly shortens the overall manufacturing cycle time, allowing for faster turnaround on customer orders and improved inventory turnover rates. The robustness of the reaction conditions also implies a lower risk of batch failure due to environmental fluctuations, thereby enhancing supply continuity.
- Cost Reduction in Manufacturing: The substitution of expensive noble metals with abundant base metals such as iron, copper, and nickel results in a drastic reduction in raw material costs. Additionally, the elimination of strong bases and the associated salt waste reduces the burden on waste treatment facilities, leading to substantial operational expenditure savings. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted per unit of product produced. By avoiding complex multi-step ligand modifications often required to stabilize free carbenes, the overall synthesis tree is shortened, further compounding the cost benefits.
- Enhanced Supply Chain Reliability: Base metals like copper and iron are globally abundant and subject to far less geopolitical supply risk compared to platinum group metals. This abundance ensures a stable and predictable supply chain, mitigating the risk of production stoppages due to raw material shortages. The use of common solvents like acetonitrile and diethyl ether, which are commodity chemicals with established global supply networks, further secures the production pipeline. The mild reaction conditions reduce the dependency on specialized high-pressure or cryogenic reactors, allowing production to be distributed across a wider network of contract manufacturing organizations without requiring significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and moderate temperatures, making it inherently safer and easier to scale from gram to ton quantities. The absence of pyrophoric reagents or toxic gases simplifies the safety protocols required for large-scale operations. From an environmental standpoint, the reduction in heavy metal usage aligns with green chemistry principles and increasingly stringent regulatory requirements regarding metal residues in pharmaceutical products. The simplified purification process generates less solvent waste and hazardous byproducts, facilitating easier compliance with environmental discharge standards and reducing the overall carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-NHC synthesis technology. These answers are derived directly from the experimental data and mechanistic understanding provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this method into your specific production workflows.
Q: What are the primary advantages of using silver oxide transmetallation over direct deprotonation?
A: Silver oxide transmetallation avoids the use of strong bases which can degrade sensitive functional groups on the ligand, ensuring higher purity and structural integrity of the final catalyst.
Q: Which base metals are compatible with this synthesis protocol?
A: The protocol demonstrates high compatibility with a wide range of active metal powders including manganese, iron, cobalt, nickel, and copper, expanding the scope beyond traditional precious metals.
Q: How does this method impact the scalability of catalyst production?
A: By utilizing mild reaction temperatures between 40°C and 80°C and common solvents like acetonitrile, the process eliminates the need for cryogenic conditions, significantly enhancing industrial scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metal N-Heterocyclic Carbene Complexes Supplier
As the global demand for efficient and sustainable catalytic solutions continues to rise, NINGBO INNO PHARMCHEM stands at the forefront of delivering high-performance chemical intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify the structural integrity and metal content of every batch. We understand that in the pharmaceutical and fine chemical sectors, consistency is key, and our robust quality management systems are designed to deliver that reliability consistently.
We invite you to collaborate with us to explore how this advanced synthesis technology can optimize your specific catalytic applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current process pain points. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and expert insight. Together, we can drive innovation and efficiency in the next generation of chemical manufacturing.
