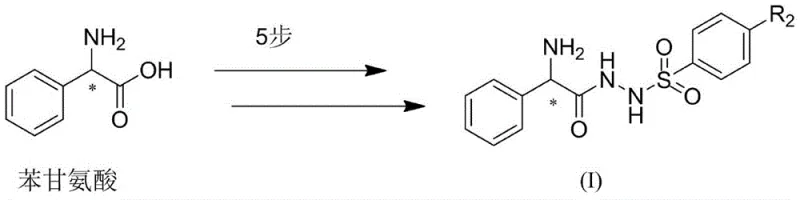
Advanced Synthesis of Chiral Phenylglycine Bifunctional Catalysts for Pharmaceutical Applications
Advanced Synthesis of Chiral Phenylglycine Bifunctional Catalysts for Pharmaceutical Applications
The landscape of asymmetric catalysis is undergoing a significant transformation with the emergence of novel organocatalysts designed to overcome the limitations of traditional systems. Patent CN109331868B discloses a groundbreaking class of phenylglycine bifunctional catalysts that offer superior chiral selectivity and reactivity. Unlike conventional proline-based catalysts, these new structures leverage the unique stereochemical properties of chiral phenylglycine combined with hydrazine functionalities. This innovation provides a robust platform for constructing complex catalytic systems capable of driving aldol condensations and other intricate organic transformations with high diastereoselectivity. For R&D directors and process chemists, this represents a pivotal shift towards more efficient and versatile catalytic tools that can streamline the synthesis of fine chemicals and biological medicines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, proline and its derivatives have been the cornerstone of organocatalysis, yet they suffer from inherent drawbacks that limit their utility in complex synthesis. The carboxylic acid functionality in proline, while useful, does not always provide the necessary balance of acidity and steric environment for optimal activity. Furthermore, existing bifunctional catalysts often struggle to achieve the expected targets for chiral selectivity and reactivity, leaving a gap in the market for high-performance alternatives. Many traditional methods require harsh conditions or expensive transition metals, which complicate downstream purification and increase the environmental footprint of the manufacturing process. The inability to effectively tune the steric hindrance and hydrogen bonding capabilities in older catalyst generations restricts their application scope, particularly in the synthesis of highly sensitive pharmaceutical intermediates where purity is paramount.
The Novel Approach
The patented methodology introduces a strategic departure from proline by utilizing chiral phenylglycine as the foundational scaffold. This approach allows for the introduction of hydrazine functional groups that significantly reduce steric hindrance while enhancing hydrogen bond activity on the nitrogen atoms. The result is a catalyst with improved chiral selection and reaction activity, capable of facilitating more complex intramolecular or intermolecular tandem reactions. The synthesis route is remarkably efficient, proceeding through five distinct steps under mild conditions without the need for heating, which simplifies operational protocols. By replacing the rigid proline ring with the flexible phenylglycine backbone, chemists gain greater control over the catalytic pocket, enabling the accommodation of diverse substrates.
Mechanistic Insights into Phenylglycine Bifunctional Catalysis
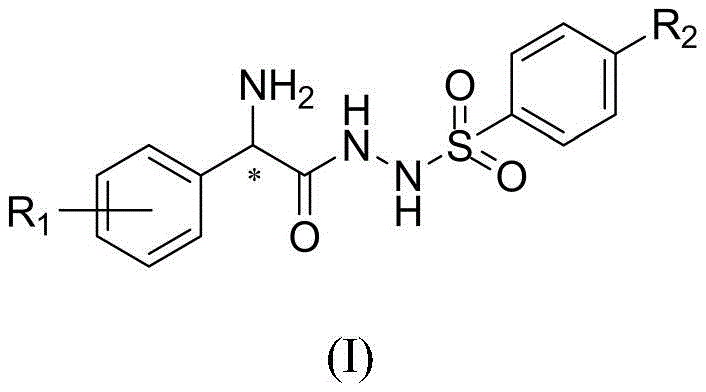
The efficacy of this catalyst class stems from its bifunctional nature, where two distinct functional groups work in concert to activate both electrophilic and nucleophilic partners simultaneously. The chiral center derived from phenylglycine ensures high enantioselectivity, while the sulfonyl hydrazide moiety provides the necessary acidic protons for hydrogen bonding activation. This dual activation mechanism lowers the energy barrier for transition states, leading to accelerated reaction rates and superior stereocontrol. The presence of the phenyl ring also offers opportunities for pi-stacking interactions, further stabilizing the transition state complex. For researchers, understanding this mechanistic nuance is critical for optimizing reaction parameters and expanding the substrate scope to include challenging electronic environments.
Impurity control is another critical aspect where this molecular design excels. The specific arrangement of the hydrazine and sulfonyl groups minimizes side reactions that typically plague less selective catalysts. The reduced steric bulk around the active site prevents non-productive binding modes, ensuring that the reaction pathway remains focused on the desired transformation. This intrinsic selectivity translates to cleaner reaction profiles, reducing the burden on purification teams and minimizing solvent waste. The ability to tolerate various substituents on the aromatic rings (R1 and R2) means that the electronic properties of the catalyst can be fine-tuned to match specific reaction requirements, offering a level of modularity that is rare in organocatalysis.
How to Synthesize Chiral Phenylglycine Bifunctional Catalyst Efficiently
The preparation of these high-value catalysts follows a logical and scalable five-step sequence that begins with readily available chiral phenylglycine. The process involves esterification, protection, hydrazinolysis, sulfonylation, and final deprotection, each step optimized for high yield and minimal waste. The use of common reagents like thionyl chloride, di-tert-butyl dicarbonate, and hydrazine hydrate ensures that the supply chain remains robust and cost-effective. Detailed standard operating procedures for each transformation are essential for maintaining batch-to-batch consistency, particularly when scaling from gram to kilogram quantities. The following guide outlines the critical stages of this synthesis, providing a roadmap for technical teams looking to implement this technology.
- Esterify chiral phenylglycine with thionyl chloride and methanol to form the methyl ester hydrochloride.
- Protect the amine group using di-tert-butyl dicarbonate (Boc2O) and an acid-binding agent.
- Convert the ester to a hydrazide derivative using hydrazine hydrate in methanol.
- React the hydrazide with substituted sulfonyl chloride to introduce the sulfonyl functionality.
- Remove the Boc protecting group using inorganic acid followed by pH adjustment to isolate the final catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to this phenylglycine-based catalyst system offers substantial strategic benefits regarding cost stability and supply security. The reliance on commodity chemicals such as phenylglycine and basic sulfonyl chlorides mitigates the risk of price volatility associated with exotic ligands or precious metals. The synthetic route avoids the use of expensive transition metal catalysts, which not only lowers the raw material cost but also eliminates the need for costly heavy metal removal steps downstream. This simplification of the purification train results in significant operational savings and reduces the overall cycle time for production. Furthermore, the mild reaction conditions, often proceeding at room temperature or with simple ice-bath cooling, drastically reduce energy consumption compared to processes requiring high heat or cryogenic temperatures.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive starting materials directly lower the bill of materials. The high yields reported in the patent examples, often exceeding 90% for intermediate steps, minimize material loss and maximize throughput. Additionally, the simplified workup procedures, which frequently involve filtration or concentration rather than complex chromatography, reduce solvent usage and labor costs. These factors combine to create a highly economical manufacturing process that enhances profit margins for end-users.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis are widely available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source vendors. The robustness of the chemical steps means that production can be easily scaled up to meet fluctuating demand without significant re-engineering of the process. This reliability is crucial for long-term project planning and ensures that downstream API synthesis schedules are not disrupted by catalyst shortages. The environmental friendliness of the reagents also aligns with modern green chemistry initiatives, facilitating easier regulatory approval and community acceptance.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the byproducts are largely benign salts and alcohols that are easy to treat. The absence of heavy metals simplifies wastewater treatment and reduces the environmental compliance burden on manufacturing facilities. The scalability of the reaction is demonstrated by the straightforward nature of the unit operations, which can be easily transferred from laboratory glassware to industrial reactors. This ease of scale-up accelerates the time-to-market for new drug candidates that rely on this catalytic technology, providing a competitive edge in the fast-paced pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and scope of application for potential users. Understanding these details is vital for making informed decisions about integrating this catalyst into your existing synthetic workflows.
Q: Why is phenylglycine preferred over proline for this bifunctional catalyst?
A: Phenylglycine allows for the introduction of hydrazine functional groups which reduce steric hindrance and improve hydrogen bond activity on nitrogen, leading to better chiral selection and reaction activity compared to traditional proline catalysts.
Q: What are the typical reaction conditions for this synthesis?
A: The synthesis utilizes mild conditions, often requiring cooling to 0°C initially but proceeding at room temperature without the need for heating, ensuring safety and energy efficiency.
Q: Can the catalyst structure be modified for different reactions?
A: Yes, the general formula allows for various substituents (R1 and R2) including alkyl, halogen, nitro, and trifluoromethyl groups, enabling customization for specific catalytic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylglycine Bifunctional Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral phenylglycine bifunctional catalyst technology for the fine chemical and pharmaceutical sectors. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of catalyst meets the highest standards of performance and consistency. We understand that the success of your asymmetric synthesis depends on the reliability of your catalyst, and we are committed to delivering products that enable your breakthroughs.
We invite you to engage with our technical procurement team to discuss how this innovative catalyst can optimize your specific manufacturing challenges. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free organocatalytic route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency and technical excellence.
