Advanced Organocatalytic Synthesis of 2-Chloro Aromatic Amines for Commercial Scale-up
Advanced Organocatalytic Synthesis of 2-Chloro Aromatic Amines for Commercial Scale-up
The synthesis of 2-chloro aromatic amine compounds represents a critical challenge and opportunity within the modern pharmaceutical and fine chemical industries, serving as indispensable building blocks for a vast array of bioactive molecules including anticancer agents, B-Raf inhibitors, and insulin receptor tyrosine kinase activators. Patent CN115850119A introduces a groundbreaking synthetic methodology that fundamentally shifts the paradigm from harsh, metal-dependent processes to a mild, efficient, and environmentally benign organocatalytic approach. This innovation addresses the long-standing industry pain points regarding regioselectivity, operational complexity, and the prohibitive costs associated with traditional transition metal catalysis. By leveraging a simple organic amine catalyst in conjunction with sulfonyl chloride reagents, this technology enables the production of high-purity pharmaceutical intermediates under remarkably gentle conditions, positioning it as a superior choice for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-chloro aromatic amines has been plagued by significant technical and economic hurdles that hinder efficient large-scale production. Traditional strategies often rely on direct electrophilic chlorination, which suffers from poor regioselectivity and generates substantial amounts of unwanted poly-chlorinated byproducts, necessitating difficult and yield-reducing purification steps. Alternatively, methods involving ortho-lithiation require cryogenic conditions as low as -78°C and the use of hazardous reagents like t-BuLi, posing severe safety risks and imposing massive energy burdens on the manufacturing facility. Furthermore, transition metal-catalyzed approaches, while offering better selectivity, depend on precious metals such as palladium, which not only inflate raw material costs but also introduce stringent regulatory requirements for residual metal limits in final drug substances.
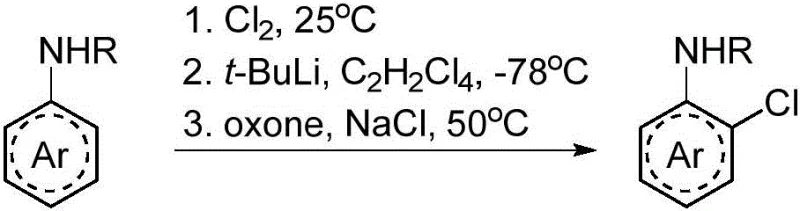
As illustrated in the comparison of prior art techniques, the reliance on complex multi-step sequences or extreme reaction parameters creates a bottleneck for supply chain continuity. The need for specialized equipment to handle cryogenic temperatures or toxic gases further complicates the operational landscape, making these conventional routes less attractive for cost reduction in API manufacturing. The environmental footprint of these methods is also considerable, often generating large volumes of hazardous waste that require expensive treatment protocols. Consequently, there is an urgent demand within the industry for a streamlined process that can deliver high yields without compromising on safety, cost, or environmental compliance.
The Novel Approach
In stark contrast to these legacy methods, the novel organocatalytic strategy disclosed in the patent offers a transformative solution by utilizing readily available organic amines as catalysts to drive the chlorination reaction with exceptional efficiency. This approach operates under mild conditions, typically between -20°C and 50°C, with a strong preference for ambient temperature at 25°C, thereby eliminating the need for energy-intensive cooling or heating systems. The use of sulfonyl chloride as the chlorinating agent ensures a controlled release of chlorine species, which, when activated by the organic amine, achieves high ortho-selectivity without the formation of significant impurities. This simplicity in reagent selection and reaction conditions translates directly into a more robust and scalable process suitable for commercial production.

The versatility of this new method is evidenced by its broad substrate scope, successfully accommodating various aromatic amines including those with electron-donating and electron-withdrawing substituents, as well as heterocyclic systems like thiophene and naphthalene derivatives. The reaction proceeds smoothly in common aprotic solvents such as toluene, which are inexpensive and easy to recover, further enhancing the economic viability of the process. By avoiding the use of heavy metals entirely, this route simplifies the downstream processing workflow, removing the need for expensive scavengers or complex filtration steps required to meet strict pharmacopeial standards for metal residues. This represents a significant leap forward in the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Organocatalytic Chlorination
The core of this technological breakthrough lies in the unique activation mechanism facilitated by the organic amine catalyst, which acts as a Lewis base to activate the sulfonyl chloride reagent. Upon interaction with the amine catalyst, such as diisopropylamine, the sulfonyl chloride forms a reactive intermediate species that is capable of delivering the chlorine atom specifically to the ortho-position of the aromatic ring. This activation lowers the energy barrier for the electrophilic substitution, allowing the reaction to proceed rapidly even at room temperature. The steric and electronic properties of the amine catalyst play a crucial role in directing the regioselectivity, ensuring that the chlorination occurs exclusively at the desired position adjacent to the amino group, thus minimizing the formation of meta- or para-chlorinated isomers.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed pathways. The controlled nature of the amine-sulfonyl chloride interaction prevents the generation of free chlorine radicals that often lead to non-selective poly-chlorination. Furthermore, the absence of transition metals eliminates the risk of metal-induced side reactions or catalyst deactivation, which can be problematic in substrates containing coordinating functional groups. The reaction kinetics are highly favorable, with completion times ranging from 0.5 to 72 hours depending on the specific substrate, allowing for flexible batch scheduling. This precise control over the reaction pathway ensures a clean impurity profile, reducing the burden on purification units and increasing the overall yield of the high-purity OLED material or pharmaceutical intermediate.
How to Synthesize 2-Chloro Aromatic Amines Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the molar ratios and addition rates to maximize efficiency and safety. The process begins with the dissolution of the aromatic amine substrate and the organic amine catalyst in a suitable solvent like toluene, followed by the controlled dropwise addition of the sulfonyl chloride reagent to manage exotherms. Detailed standard operating procedures regarding temperature control and workup protocols are essential to reproduce the high yields reported in the patent examples. For a comprehensive guide on the specific execution of this protocol, please refer to the standardized synthesis steps outlined below.
- Prepare the reaction mixture by dissolving the aromatic amine compound and the organic amine catalyst in an aprotic solvent such as toluene.
- Maintain the reaction temperature between -20°C and 50°C, preferably at 25°C, while slowly adding the sulfonyl chloride reagent.
- Allow the reaction to proceed for 0.5 to 72 hours, followed by aqueous workup and purification to isolate the high-purity 2-chloro aromatic amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic technology presents a compelling value proposition centered around cost stability and operational resilience. By shifting away from precious metal catalysts, manufacturers can insulate their production costs from the volatile pricing of commodities like palladium, leading to substantial cost savings over the lifecycle of the product. The simplified reaction conditions also mean that existing general-purpose reactors can be utilized without the need for specialized cryogenic infrastructure, thereby optimizing capital expenditure and increasing facility utilization rates. This flexibility allows for faster response times to market demands and reduces the lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like organic amines and sulfonyl chlorides drastically lower the raw material bill of materials. Additionally, the removal of heavy metal scavenging steps reduces the consumption of auxiliary materials and simplifies waste disposal, contributing to significant operational expense reductions without compromising product quality.
- Enhanced Supply Chain Reliability: Since the reagents involved are widely available bulk chemicals rather than specialized proprietary catalysts, the risk of supply disruption is minimized. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production failures or delays. This reliability is critical for maintaining continuous supply lines for critical drug substances and avoiding costly stockouts.
- Scalability and Environmental Compliance: The mild reaction temperatures and lack of toxic heavy metals make this process inherently safer and easier to scale from kilogram to tonne quantities. The reduced environmental footprint aligns with increasingly stringent global regulations on industrial emissions and waste, facilitating smoother regulatory approvals and enhancing the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These insights are derived directly from the patent data and are intended to clarify the practical benefits and operational parameters for potential partners. Understanding these details is key to evaluating the feasibility of integrating this technology into your existing supply chain.
Q: What are the primary advantages of this organocatalytic method over traditional metal-catalyzed routes?
A: The primary advantage is the elimination of expensive transition metal catalysts like palladium, which significantly reduces raw material costs and removes the need for complex heavy metal removal steps, ensuring higher purity standards for pharmaceutical applications.
Q: Does this method support a wide range of substrate functional groups?
A: Yes, the method demonstrates excellent substrate compatibility, tolerating both electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluorine and esters, making it versatile for diverse drug intermediate synthesis.
Q: How does the reaction condition impact scalability and safety?
A: Operating at mild temperatures ranging from -20°C to 50°C, preferably at ambient 25°C, drastically reduces energy consumption and thermal hazards, facilitating safer and more straightforward commercial scale-up compared to cryogenic lithiation processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro Aromatic Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in reshaping the landscape of fine chemical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, industrial settings. Our state-of-the-art facilities are equipped to handle the specific requirements of this mild chlorination process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemistry and reliable supply.
